Additive Orthopaedics Develops 3D Printed Implant to Successfully Treat Hammertoes
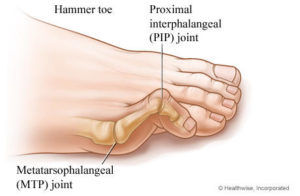 A hammertoe is a deformity of the foot that typically presents in the second, third or fourth toe as an overly bent toe that looks similar in shape to a hammer. It is caused by the joints becoming deformed by being bent for very long periods of time, which causes the muscles to shorten. It is usually caused by wearing shoes that are too small or too narrow, or by wearing extremely high heels that force the foot into a very unnatural shape, though hammertoe can also be caused by conditions as varied as diabetes, osteoarthritis, rheumatoid arthritis or even a stroke. It can be an extremely painful condition that can inhibit a patient’s ability to walk, and lead to other painful foot issues.
A hammertoe is a deformity of the foot that typically presents in the second, third or fourth toe as an overly bent toe that looks similar in shape to a hammer. It is caused by the joints becoming deformed by being bent for very long periods of time, which causes the muscles to shorten. It is usually caused by wearing shoes that are too small or too narrow, or by wearing extremely high heels that force the foot into a very unnatural shape, though hammertoe can also be caused by conditions as varied as diabetes, osteoarthritis, rheumatoid arthritis or even a stroke. It can be an extremely painful condition that can inhibit a patient’s ability to walk, and lead to other painful foot issues.
For very mild cases of hammertoe, doctors usually send the patient to physical therapy and suggest replacing any tight shoes with new shoes that have very soft insoles and are roomy enough for the toes to be able to fully extend. Any prescribed physical therapy usually consists of a series of foot and joint exercises that can help reshape the toe and prevent the condition from worsening. Doctors and physical therapists have also developed special braces that force the toe back into the correct shape, but these can be very painful to wear depending on the severity of the deformity, and they don’t always work.
 Many cases of hammertoe need to be corrected with podiatric surgery that involves surgically reshaping the afflicted toes. In fact more than 1 million people in the United States end up having to have a surgical procedure to correct their hammertoe each year. As with any surgery on the foot, this can often cause a great deal of pain, take weeks of recuperation in order to be fully mobile again and is no guarantee of it permanently solving the problem. But Additive Orthopaedics, a Little Silver, New Jersey-based early stage orthopedic extremity device manufacturer and developer has announced that they have successfully treated a series of patients who were diagnosed with hammertoe using a new 3D printed hammertoe implant.
Many cases of hammertoe need to be corrected with podiatric surgery that involves surgically reshaping the afflicted toes. In fact more than 1 million people in the United States end up having to have a surgical procedure to correct their hammertoe each year. As with any surgery on the foot, this can often cause a great deal of pain, take weeks of recuperation in order to be fully mobile again and is no guarantee of it permanently solving the problem. But Additive Orthopaedics, a Little Silver, New Jersey-based early stage orthopedic extremity device manufacturer and developer has announced that they have successfully treated a series of patients who were diagnosed with hammertoe using a new 3D printed hammertoe implant.
“Our implant has properties and features only possible through the additive manufacturing process. We are confident that the market will respond well to this, our patient specific products in development, and our other devices currently being reviewed by the FDA,” said the President and Founder of Additive Orthopaedics, Greg Kowalczyk.
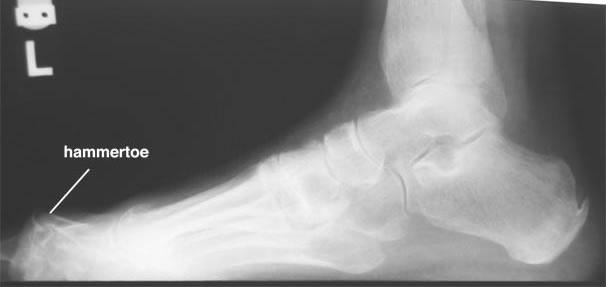 Additive Orthopaedics developed their 3D printed corrective implant to be faster to manufacture, custom made for the patient’s foot and to improve the chances of the condition being successfully corrected. After the successful testing of the implant, the FDA cleared the hammertoe implant, making it available to doctors all over the country. The young startup has already raised enough seed money to close their first round and develop the hammertoe implant. Among their investors is Asimov Ventures. After the successful tests, Additive Orthopaedics is readying themselves to open up a second round of financing to help them take their new product to market. They are also in the process of developing and gaining approval for new corrective implants for similar conditions like mallet toe or claw toe. Do you know anyone who would benefit from one of these implants? Discuss over in the 3D Printed Hammertoe forum at 3DPB.com.
Additive Orthopaedics developed their 3D printed corrective implant to be faster to manufacture, custom made for the patient’s foot and to improve the chances of the condition being successfully corrected. After the successful testing of the implant, the FDA cleared the hammertoe implant, making it available to doctors all over the country. The young startup has already raised enough seed money to close their first round and develop the hammertoe implant. Among their investors is Asimov Ventures. After the successful tests, Additive Orthopaedics is readying themselves to open up a second round of financing to help them take their new product to market. They are also in the process of developing and gaining approval for new corrective implants for similar conditions like mallet toe or claw toe. Do you know anyone who would benefit from one of these implants? Discuss over in the 3D Printed Hammertoe forum at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
nScrypt’s Ken Church on Why Additive Electronics Is Finally Finding Its Fit
For years, additive manufacturing (AM) has promised to reshape electronics. The idea has always been to print circuits directly where they are needed, add them into parts, and move beyond...
Harvard’s Jennifer Lewis Lab Is 3D Printing Artificial Muscles That Twist and Bend on Demand
Researchers at Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS) have developed a new way to 3D print materials that can move on their own, bending, twisting,...
3D Printing News Briefs, May 2, 2026: Soft Robots, Agricultural Waste, & More
In this weekend’s 3D Printing News Briefs, we’ll start off with a multi-laser metal powder bed fusion 3D printer and post-processing news. We’ll end with research into soft robotics and...
Harvard SEAS Engineers Develop 3D Printing Method for Soft Robotic Components with Programmable Shapes
The world of soft robotics is still largely in its pure research phase, but the R&D landscape has started to produce examples of early-stage commercialization. Researchers have started to refine...















































