FDA Gives 510(k) Clearance to 3D Printed Titanium Digital Fusion Implant – First Product from Additive Orthopaedics
 Some of the most delicate bones in the body are those in the extremities. While a broken finger or toe is obviously not as debilitating as, say, a broken arm or leg, they’re still painful and, in some cases, can have complications. The bones in the toes are particularly tricky – there’s a lot that can go wrong with them, breaks or not. Those delicate little bones do a lot of work and carry a lot of weight, so it’s important to make sure they heal correctly, and sometimes a little extra help is required.
Some of the most delicate bones in the body are those in the extremities. While a broken finger or toe is obviously not as debilitating as, say, a broken arm or leg, they’re still painful and, in some cases, can have complications. The bones in the toes are particularly tricky – there’s a lot that can go wrong with them, breaks or not. Those delicate little bones do a lot of work and carry a lot of weight, so it’s important to make sure they heal correctly, and sometimes a little extra help is required.
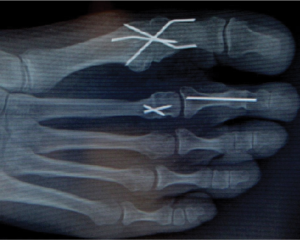
Digital fusion refers to a procedure in which the bones of the toe are fused together after a surgery, and occasionally they require stabilization from a pin or implant while they grow together and heal. New Jersey-based Additive Orthopaedics, LLC is a brand new company dedicated to the development of 3D printed orthopedic devices specifically for the extremities, and their first product, a 3D printed titanium digital fusion implant, has just been given 510(k) clearance from the FDA.
3D printing has made a major impact on the orthopedics industry lately, largely thanks to the development of metal printing technologies. The devices that get the most attention tend to be the big, potentially lifesaving implants like those for the neck and spine, but there are a lot of great things being done for bones elsewhere in the body – like the feet and hands – that shouldn’t be ignored. One of the biggest benefits of additive manufacturing in orthopedics is its ability to customize. Traditionally manufactured implants tend to be “one size fits all,” but the human body isn’t one size fits all, and an ill-fitting implant can lead to complications in the healing process. That’s what Additive Orthopaedics is trying to change with their customized, patient-specific implants.“Additive manufacturing allows us to develop complex geometries at reasonable manufacturing costs not possible before now with traditional manufacturing,” said Greg Kowalczyk, President and Founder of Additive Orthopaedics. “Design teams really need to think outside the box when considering this innovative manufacturing process. And where better to do that then the Global US Extremity Market valued at $1.3 billion and growing at over 10% per year. We are confident that the market will respond well to this, our patient specific products in development, and our other devices currently being reviewed by the FDA.”
 Additive Orthopaedics is still in the earliest stages; they just moved past their initial seed funding rounds and have just announced their second round of funding. One of the company’s investors is Asimov Ventures, the venture capital firm launched last year to support early-stage startups in 3D printing and robotics. They’ve already invested in several promising companies that have really taken off, like Wiiv and Metamason, and with their flagship device already FDA approved, I expect Additive Orthopaedics is soon to take off as well. They’re certainly in the right market.
Additive Orthopaedics is still in the earliest stages; they just moved past their initial seed funding rounds and have just announced their second round of funding. One of the company’s investors is Asimov Ventures, the venture capital firm launched last year to support early-stage startups in 3D printing and robotics. They’ve already invested in several promising companies that have really taken off, like Wiiv and Metamason, and with their flagship device already FDA approved, I expect Additive Orthopaedics is soon to take off as well. They’re certainly in the right market.
“3D printing has a major role in the future of orthopaedics,” said Dr. Selene Parekh, Professor of Surgery in the Department of Orthopaedic Surgery at Duke Orthopaedics and North Carolina Orthopaedic Clinic. “In today’s cost sensitive health care environment, 3D printing allows us to bring highly complex, lower cost technologies to the market very quickly, with patient specific features that will potentially improve patient outcomes.”
Discuss further in the Additive Orthopaedics Gains FDA Clearance for 3D Printed Implants forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
nScrypt’s Ken Church on Why Additive Electronics Is Finally Finding Its Fit
For years, additive manufacturing (AM) has promised to reshape electronics. The idea has always been to print circuits directly where they are needed, add them into parts, and move beyond...
Harvard’s Jennifer Lewis Lab Is 3D Printing Artificial Muscles That Twist and Bend on Demand
Researchers at Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS) have developed a new way to 3D print materials that can move on their own, bending, twisting,...
3D Printing News Briefs, May 2, 2026: Soft Robots, Agricultural Waste, & More
In this weekend’s 3D Printing News Briefs, we’ll start off with a multi-laser metal powder bed fusion 3D printer and post-processing news. We’ll end with research into soft robotics and...
Harvard SEAS Engineers Develop 3D Printing Method for Soft Robotic Components with Programmable Shapes
The world of soft robotics is still largely in its pure research phase, but the R&D landscape has started to produce examples of early-stage commercialization. Researchers have started to refine...