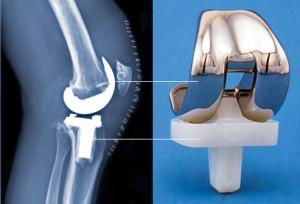
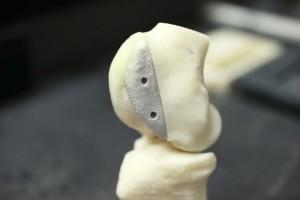
Over the years many different materials have been used to make bone and joint implants, including a wide range of plastics, ceramics and metals like titanium, with varying results. The one thing that all of these implants have in common is the fact that they simply won’t last. Typically a medical implant will need to be replaced in about 10 to 20 years, which can cause more pain and trauma to the patient, especially as they age and surgery becomes more of a risk. Additionally, these implants are mass produced, making them, essentially, one-size fits all. Unfortunately not every patient is the same size, so either the implants need to be modified to fit during surgery, or the bone needs to be shaped to fit the implant, which often doubles the length of the procedure.
University of Toronto professor emeritus Bob Pilliar has been researching synthetic bone substitutes for more than twenty years, and he and his team of researchers finally found their secret ingredient. Calcium polyphosphate is the mineral that makes up more than seventy percent human bones, and Pilliar believed that it was the ideal material to make a bone substitute. Not only was it biodegradable, but it could actually be used to connect to the patient’s existing bone cells to encourage regrowth and create a longer-lasting replacement. Pillar’s team only needed a way to manufacture the bone implants quickly and accurately, which is where mechatronics engineer Mihaela Vlasea comes in.
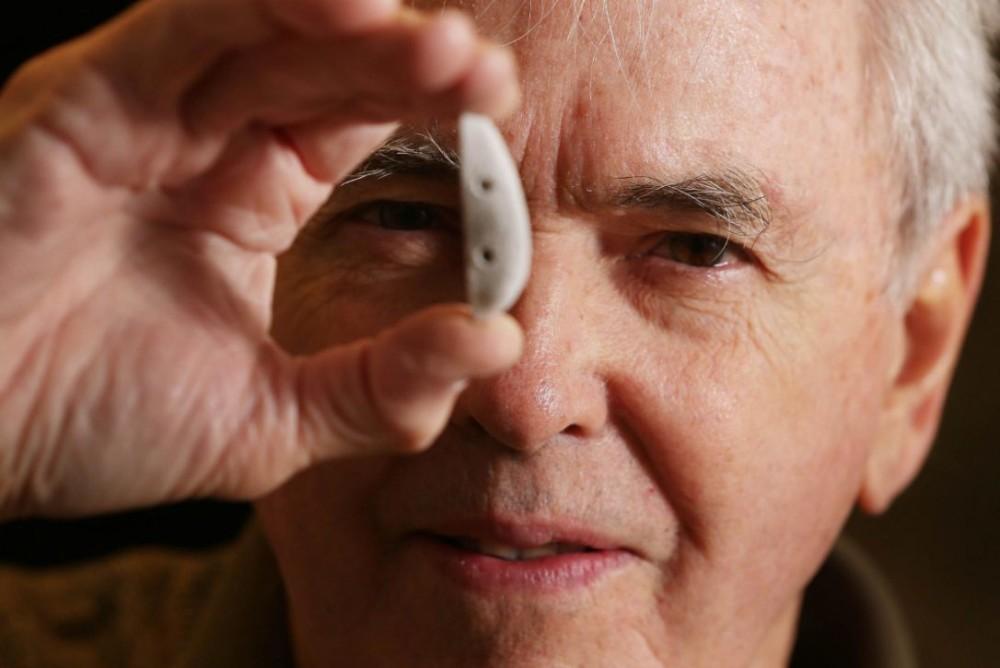
University of Toronto’s Bob Pilliar and a 3D printed bone implant. [Image: Steve Russell/Toronto Star]
The massive, refrigerator-sized 3D printer uses ultraviolet light and a light-reactive binding agent to fuse the powdered calcium polyphosphate into a biodegradable bone scaffold that has an intricate internal network of porous features. The physical structure of the 3D printed implant will encourage natural bone cells to migrate, and grow, into the scaffold implant. Slowly, as the implant begins to be broken down by the patient’s body, real tissue will begin to replace it. The surface of the implant will also encourage cartilage cells to naturally attach to them, ultimately making the implant more mechanically compatible than traditional implants.
One of the more exciting applications for this 3D printed bone material is in creating replacement joints and cartilage for people living with arthritis. While most people believe that arthritis primarily afflicts the elderly, in fact many types of the degenerative joint disorder are caused by trauma or infections in patients of almost any age. Unfortunately cartilage cells do not regenerate, so the available treatment options capable of stimulating joint repair are limited to implants made of plastic or metal.“It’s a little short of Star Trek where you zap a person, and they’re fixed, but it’s along the same lines. It is nice to think of replacing joints and getting rid of all these metals. But in the short term, we are looking at substituting small regions of bone. If we’re successful, you can imagine trying to re-generate a larger region of a joint,” Pillar told the Toronto Star.
Dr. Rita Kandel is the chief of pathology and laboratory medicine at Mount Sinai Hospital, and she has been working with Pillar and his fellow University of Toronto researcher Marc Grynpas for twenty years. She is developing a method of using Pillar’s calcium polyphosphate bone material and Vlasea’s 3D printer to create biological joint replacements made from the patient’s own tissue and cellular material. The process starts by extracting stem cells from a patient’s bone marrow, which are then reprogrammed into immature cartilage cells and then matured over a period of several weeks by Dr. Kandel.
Once the cells have matured, the patient’s custom 3D printed calcium polyphosphate implant is immersed in a bioactive material containing the stem cells. When these replacement joints are implanted into the patient, the new cells begin to function naturally and they will even start to heal themselves. This process is called “biological resurfacing”, and it will reduce the chance of the body rejecting the implant. It could eventually lead to more natural, longer-lasting joint replacements, especially for chronic conditions like osteoarthritis.
“If you are using the individual’s own cells, they are not recognized by the body as foreign. It will help people so they don’t experience as much pain. For me, that’s a major contribution,” Dr. Kandel says.
Currently the 3D printed bone replacements have only been tested on animals, and those trials are expected to continue for a few years. Dr. Kandel and her joint implant replacement process is closer to being available, however there is currently no time table for availability, or a known price point. According to Dr. Kandel, these new bio-replacement articulated joints could be used to repair damage to knees, hips and fingers, and they will likely be available within a decade. Discuss in the 3D Printed Bone Scaffolds forum over at 3DPB.com.
[Source: Toronto Star]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printed Bone Grafts From Georgetown Researchers Could Replace Traditional Implants
Researchers at Georgetown University are developing a new type of 3D printed bone graft designed to work more like real human bone. Instead of relying on metal implants or donor...
TV’s Scarpetta Suggests We Can 3D Print Full Human Organs. Reality Is… Not Yet
Having spent a good part of my journalistic career covering crime stories, it’s hard for me to stay away from any crime or medical forensic drama. So when Scarpetta premiered...
At RAPID + TCT 2026, Executive Keynotes Break Down What’s Next for AM
While AI is expected to be a major focus at RAPID + TCT 2026, the event is also putting the spotlight on something just as important: leadership. This year’s Executive...
3D Printing Moves Deeper Into Production as Parts Near $110B by 2034
A new report takes a closer look at how much 3D printing is actually being used in real production. The numbers point to a market that is already growing at...