Materialise Partners with University of Michigan and Tissue Regeneration Services for Clinical Trials of 3D Printed Tracheal Splint
 New parents, while overjoyed at the life they have created, also tend to be filled with fear: What if I’m a terrible parent? What if I do something wrong? And, perhaps the most pervasive fear: What if there’s something wrong with my baby? While medical resources, at least in developed countries, are better than they’ve ever been, there are still a lot of conditions that can affect infants and keep their parents up all night with worry. One of these conditions is tracheobronchomalacia (TBM), a life-threatening, congenital condition that affects one in 2,200 babies. TBM causes the airway to periodically collapse, dangerously affecting breathing.
New parents, while overjoyed at the life they have created, also tend to be filled with fear: What if I’m a terrible parent? What if I do something wrong? And, perhaps the most pervasive fear: What if there’s something wrong with my baby? While medical resources, at least in developed countries, are better than they’ve ever been, there are still a lot of conditions that can affect infants and keep their parents up all night with worry. One of these conditions is tracheobronchomalacia (TBM), a life-threatening, congenital condition that affects one in 2,200 babies. TBM causes the airway to periodically collapse, dangerously affecting breathing.
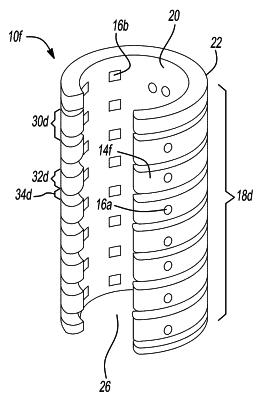
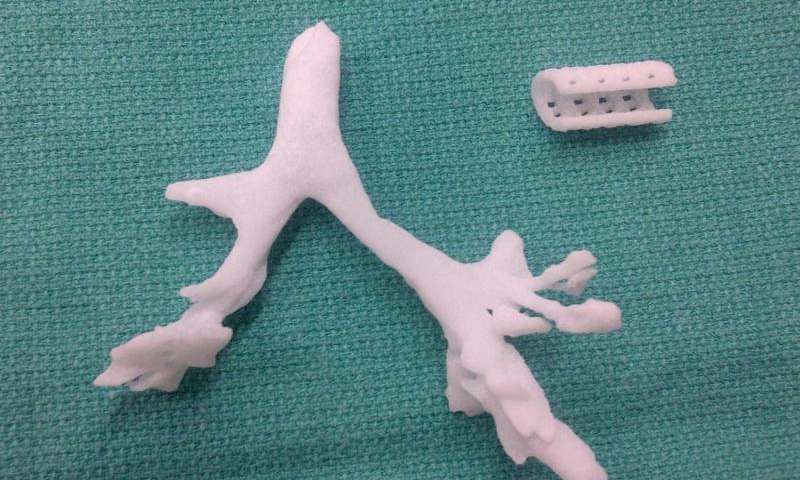
While the thought of TBM might make parents break out into a cold sweat, there is hope. Dr. Scott Hollister, a biomedical engineering professor at the University of Michigan, and Dr. Glenn Green, an otolaryngologist at C.S. Mott Children’s Hospital, invented a tracheal splint using 3D printing in 2012. The splint, implanted in the trachea of a baby with TBM, keeps the airway open until it can grow into a healthy state and stay open on its own. This process takes about three years, at which point the splint will dissolve and be absorbed by the body. We’ve already seen examples of the lifesaving potential of this device, but it’s been continually under development and hasn’t been widely available yet.
That should change thanks to two new partnerships: one between Materialise and the University of Michigan, the other between Materialise and Tissue Regeneration Systems (TRS). The University of Michigan has signed a licensing agreement with Materialise, whose Mimics Innovation Suite was used to design the tracheal splint. The agreement will allow for the commercialization of the device, which will begin with a clinical trial involving 30 patients at Mott Children’s Hospital sometime next year.
“This agreement is a critical step in our goal to make this treatment readily available for other children who suffer from this debilitating condition,” said Dr. Green. “We have continued to evolve and automate the design process for the splints, allowing us to achieve in two days what used to take us up to five days to accomplish. I feel incredibly privileged to be building products that surgeons can use to save lives.”

L – R: Dr. Francesco Migneco, Lead Biomedical Engineer, TRS; Jim Adox, Chairman of the Board, TRS; and Materialise CEO Fried Vancraen
The bioresorbable splints will be manufactured by Plymouth, Michigan startup Tissue Regeneration Systems, which received its first commercial product clearance from the FDA in 2013 after several years of product development.
TRS has also worked with the University of Michigan in the past; in 2007, the company licensed a bioresorbable technology platform used to design the splint from the university. The splints are custom-designed based on patients’ CT scans and manufactured with a biodegradable biopolymer called polycaprolactone.
“The collaboration between TRS and Materialise will provide production capacity for the tracheal splint, which will allow the splint to be available to a larger number of infants who are affected with TBM,” said Bryan Crutchfield, Managing Director, Materialise USA.
Regarding the partnership with U of M, he added: “Here at Materialise, we strongly believe in the transformative power of great collaborations, such as the one we have enjoyed these past two years with Dr. Green, Dr. Hollister, and all others who have contributed to this live-saving application of 3D printing. This collaboration is proof that when the right skill sets and technologies are combined, solutions can be found for problems once thought impossible.”

L- R, Standing: Maarten Zandbergen, Global Services Manager, Materialise; Dr. Kevin Ward, Professor of Emergency Medicine, University of Michigan Medical School; Bryan Crutchfield, Managing Director, Materialise USA; Dr. Glenn E. Green, Associate Professor, Otolaryngology, University of Michigan Medical School; Steven L. Kunkel, Pd.D., Senior Associate Dean of Research, University of Michigan Medical School; Kenneth Nisbet, Associate Vice President for Research, Tech Transfer, University of Michigan Seated: Fried Vancraen, CEO, Materialise
Materialise, which serves a wide array of industries including automotive, aerospace, and consumer products, recently announced that they will be expanding their production of medical models and focusing more on the healthcare field.
“It was the possibility to realize 3D-printing enabled medical applications that, in part, motivated me to start Materialise 25 years ago,” said Materialise founder and CEO Fried Vancraen. “Today, we are proud to offer an entire range of software and services that can help universities, research centers, medical device companies, and hospitals develop solutions that result in a better and healthier world, such as the treatment developed for children diagnosed with TBM.”
Discuss this story in the Materialise 3D Printed Tracheal forum on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
The Hidden Cost of Lost Engineering Intent
Recent reporting on the UPS MD-11 engine separation on Flight 2976 has understandably focused on the immediate question: what happened? The investigation will determine that, as it should. Serious engineering...
3D Printing News Briefs, May 28, 2026: Continuous Fiber Reinforcement, Bioprinted Trachea, & More
In today’s 3D Printing News Briefs, America Makes announced the winners of its JAQS-SQ Project Call. Axtra3D is partnering with Keystone Industries to expand its dental material ecosystem, while BigRep...
Stratasys Acquires Markforged, Analysis of AM’s Latest Consolidation Move
A very long time ago, in 2023, the additive manufacturing (AM) industry was enraptured over the attempts by a large chunk of its publicly traded original equipment manufacturers (OEMs) to...
Why Additive Manufacturing Has Finally Earned Its Place on the Production Line
For years, the conversation around additive manufacturing followed a predictable script. Engineers would acknowledge its usefulness for prototyping – faster iterations, cheaper design validation, no tooling to worry about –...