 For medicine, bioprinting is a necessity and an inevitability. The demand for organs will undoubtedly continue to climb with the global median age. Yet what hurdles lie in the way of creating a renewable supply? One major engineering obstacle is properly seeding and feeding the culture in order to create a functional organ. This is because complex tissues are composed of many different types of cells precisely arranged in 3 dimensions. The approaches can be broadly divided between those using lasers and those that do not, those with scaffolds and those without. The number of possibilities multiplies when one takes into account choices in materials, growth and differentiation factors, and the types of cells one is using. Bioprinting will be a boon to transplant patients and pharmaceutical testing.
For medicine, bioprinting is a necessity and an inevitability. The demand for organs will undoubtedly continue to climb with the global median age. Yet what hurdles lie in the way of creating a renewable supply? One major engineering obstacle is properly seeding and feeding the culture in order to create a functional organ. This is because complex tissues are composed of many different types of cells precisely arranged in 3 dimensions. The approaches can be broadly divided between those using lasers and those that do not, those with scaffolds and those without. The number of possibilities multiplies when one takes into account choices in materials, growth and differentiation factors, and the types of cells one is using. Bioprinting will be a boon to transplant patients and pharmaceutical testing.
Laser assisted bioprinters were invented first. Later, Laser Induced Forward Transfer (LIFT), a technique initially invented for inorganic molecules, was appropriated by the biological sciences. After refining the technique, researchers reported a cell survival rate of 95%. This is because, unlike its predecessor, the beam does not come into direct contact with its cargo. Living cells are, not unexpectedly, quite sensitive to elevated temperatures. Laser pulses cause an absorbing layer to vaporize, resulting in the creation of droplets. The size of the droplet can be adjusted by altering the energy behind the pulse. Laser-free methods, inspired by household inkjet printers, came later. In 2002 Makoto 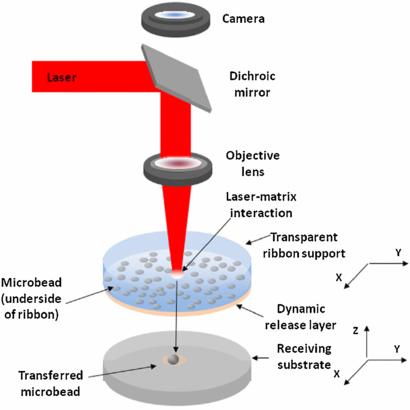 Nakamura had an epiphany: the ink droplets ejected by standard household printers are approximately the size of human cells. Thermal DoD (Drop on Demand), used by the Canon in your home office, differs from piezoelectric DoD. In thermal DoD a current is passed through the heating units, resulting in the formation of a bubble, which propels ink onto the paper. Piezoelectric printers are more common in commercial and industrial settings, probably because residue accumulation is not an issue and a wider variety of inks can be used. This is quite interesting in and of itself, but the medium in which the cells replicate and the substrate onto which they are printed is equally vital.
Nakamura had an epiphany: the ink droplets ejected by standard household printers are approximately the size of human cells. Thermal DoD (Drop on Demand), used by the Canon in your home office, differs from piezoelectric DoD. In thermal DoD a current is passed through the heating units, resulting in the formation of a bubble, which propels ink onto the paper. Piezoelectric printers are more common in commercial and industrial settings, probably because residue accumulation is not an issue and a wider variety of inks can be used. This is quite interesting in and of itself, but the medium in which the cells replicate and the substrate onto which they are printed is equally vital.
We are not amoebas; the contents of our bodies are not suspended in a gooey ectoplasm. They are anchored by the extracellular matrix (ECM) and, developmentally speaking, are dependent on a dizzying array of chemical cues. The ECM supplies bodily structures with their shape and with the correct set of messenger molecules to grow, maintain, and remodel the tissue. Because animals need to take in nutrients, excrete waste, and receive sufficient oxygenation, vascularization and neovascularization are both essential, as is initial porosity of the chosen scaffold. Biocompatibility is key from a clinical perspective and the correct bioactivity is needed from an engineering one. The scaffold releases biomolecules to “influence cell morphology and alignment.” This can be achieved in a few different ways. Hydrogels, for instance, release the desired proteins at the right times by swelling. Hydrogels have been made from a variety of substances familiar to molecular biologists: alginate, chitosan, collagen, matrigel, gelatin, and agarose. All of these substrates are noncovalently crosslinked. Covalently linked synthetic hydrogels like polyethylene glycol can be fine tuned for responsiveness, but problems with biodegradability and biocompatibility persist.
For curious minds and antsy hands there are a number of easy-to-follow DIY tutorials for modifying old (or new) inkjets. These guides are only a Google search away. Far from being the pointless playthings of wet lab lovers, the technology has gained the attention of two household names: L’Oréal and Procter & Gamble. Both companies have a seemingly inexhaustible thirst for skin cells to test their products. Currently it takes a week for a skin sample to form. Organovo is also working with the pharmaceutical titan Merck to produce liver and kidney tissues, a significantly more complex undertaking. Spheroids containing parenchymal (fundamental) cells are loaded into a syringe. In the other nonparenchymal cells are placed along with the hydrogel, which creates the mould. The fundamental portions are poured in and after a period of incubation liver tissue emerges. Organovo is also pioneering the production of tumors on demand, which allow for rapid drug testing. These feats, which may seem small to the uninitiated, involve a profound grasp of the organ’s geometry and the way the various factors come together to form it. Ergo, someone serious about contributing to this field would be advised to brush up on, among many other areas, their 3D design skills, topology, and statistics.
the other nonparenchymal cells are placed along with the hydrogel, which creates the mould. The fundamental portions are poured in and after a period of incubation liver tissue emerges. Organovo is also pioneering the production of tumors on demand, which allow for rapid drug testing. These feats, which may seem small to the uninitiated, involve a profound grasp of the organ’s geometry and the way the various factors come together to form it. Ergo, someone serious about contributing to this field would be advised to brush up on, among many other areas, their 3D design skills, topology, and statistics.
Computers play a substantial and growing role in all areas of modern biology. In order to create a decent program one must first become familiar with the Differential Adhesion Hypothesis (DAH). Put simply, cells move towards other cells with a similar adhesiveness in order create stable structures. As it is with so many other seemingly simple phenomena, the models needed to simulate DAH require a little more than basic arithmetic. Although the mathematical barrier to entry is not dreadfully high. Monte Carlo Methods are useful in creating simulations and solving optimization problems. This entails generating many possible pathways and evaluating their outcomes. Predictive modeling, even if it does not give perfect answers, is an excellent tool in facilitating discovery. One day clever researchers may design superlivers capable of attacking an even wider variety of toxins. Maybe they will make pancreases immune to diabetes and cancer. Before these wonders are wrought researchers must continue to wrestle with the rules written by billions of years of evolution. In the words of Francis Bacon, a life extension luminary before the term existed: “nature to be commanded, must be obeyed.”
About Author
Adam Alonzi is a writer, biotechnologist, documentary maker, futurist, inventor, and programmer. He is the author of A Plank in Reason and Praying for Death: A Zombie Apocalypse. His personal blog can be found at adamalonzi.wordpress.com. And his podcast can be found at adamalonzi.libsyn.com.
Works Cited
- Bacon, Francis, Francis Bacon, and Francis Bacon. Advancement of Learning ; And, Novum Organum ; And, New Atlantis. Chicago: Encyclopædia Britannica, 1955. Print.
- Barnatt, Christopher. “ExplainingTheFuture.com : Bioprinting.” ExplainingTheFuture.com : Bioprinting. Explaing The Future, n.d. Web. 17 Aug. 2015.
- Chan, B. P., and K. W. Leong. “Scaffolding in tissue engineering: general approaches and tissue-specific considerations.” European spine journal 17.4 (2008): 467-479.
- Chua, Chee Kai, and Wai Yee Yeong. Bioprinting: Principles and Applications. N.p.: n.p., n.d. Print.
- Gilpin, Lyndsey. “3D ‘bioprinting’: 10 Things You Should Know about How It Works.” TechRepublic. TechRepublic, n.d. Web. 17 Aug. 2015.
- Jakab, Karoly, et al. “Three-dimensional tissue constructs built by bioprinting.”Biorheology 43.3 (2006): 509-516.
- Li, Chuang, Alan Faulkner-Jones, Alison R. Dun, Juan Jin, Ping Chen, Yongzheng Xing, Zhongqiang Yang, Zhibo Li, Wenmiao Shu, Dongsheng Liu, and Rory R. Duncan. “Rapid Formation of a Supramolecular Polypeptide–DNA Hydrogel for In Situ Three-Dimensional Multilayer Bioprinting”
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Inside The Barnes Global Advisors’ Vision for a Stronger AM Ecosystem
As additive manufacturing (AM) continues to revolutionize the industrial landscape, Pittsburgh-based consultancy The Barnes Global Advisors (TBGA) is helping shape what that future looks like. As the largest independent AM...
Ruggedized: How USMC Innovation Officer Matt Pine Navigates 3D Printing in the Military
Disclaimer: Matt Pine’s views are not the views of the Department of Defense nor the U.S. Marine Corps Throughout this decade thus far, the military’s adoption of additive manufacturing (AM)...
U.S. Congress Calls Out 3D Printing in Proposal for Commercial Reserve Manufacturing Network
Last week, the U.S. House of Representatives’ Appropriations Committee moved the FY 2026 defense bill forward to the House floor. Included in the legislation is a $131 million proposal for...


































