 Organovo (NYSE MKT:ONVO) is one of those companies which seems to be almost too ahead of their time to really be taken seriously from an investment standpoint. That’s at least the feeling I get when discussing the company’s work with various individuals within the investing community. With that said, the company, who is building a business around the sale of 3D printed human tissue — first liver, then kidney and perhaps skin — may have just made believers out of some.
Organovo (NYSE MKT:ONVO) is one of those companies which seems to be almost too ahead of their time to really be taken seriously from an investment standpoint. That’s at least the feeling I get when discussing the company’s work with various individuals within the investing community. With that said, the company, who is building a business around the sale of 3D printed human tissue — first liver, then kidney and perhaps skin — may have just made believers out of some.
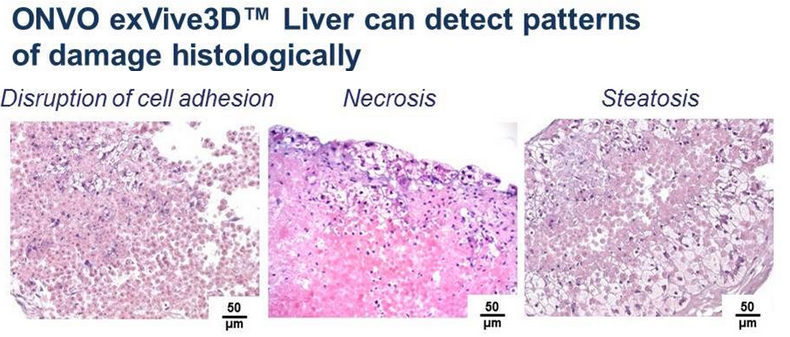
Organovo’s exVive3D Human Liver Tissue, which they commercially launched in November of 2014, apparently has begun generating revenue for the company, as they divulged this following their fiscal 2015 year-end report (which ended March 31 2015). The tissue, which is being purchased by multiple top 25 global pharma companies as a method for preclinical safety testing, has generated $1.94 million in sales between April 1 and June 9. Of that, $0.29 million is reflected in the fiscal report released after hours yesterday.
“Response to the exVive3D Human Liver Tissue has been strong, and in line with our expectations,” commented Keith Murphy, Organovo’s chief executive officer. “We continue to expect this tissue to grow into the tens of millions in annual revenue, and that it has $100M+ revenue potential. The fact that recent data shows that the exVive3D Human Liver Tissue clearly demonstrated the toxicity of another drug missed during preclinical studies, troglitazone (Rezulin), which exhibited a clear response by causing a statistically significant drop in liver function in our liver tissue at 7 days, provides the potential for our commercial penetration to accelerate even faster.”
The majority of this revenue, to date, comes by way of pre-lease availability of their exVive3D tissue, and the company notes that it won’t be until the August quarter that full-scale availability of the product/service will be realized within a quarterly report.
“Fiscal 2015 represented a successful step forward for Organovo with the launch of our commercial liver product and service,” stated Barry Michaels, Organovo’s Chief Financial Officer. “We expect continued adoption from the global pharmaceutical industry in the coming year. We are moving forward with confidence and momentum, both in the expansion of our toxicology tissues and in our efforts to advance other programs.”
A few highlights of this fiscal year-end report are as follows (note that most of the revenue from their exVive3D tissue has been realized after the fiscal year had ended on March 31st):
- Cash and cash equivalents on hand: $50.1 million
- Negative cash flow from operations: $19.6 million
- Accumulated deficit: $122.3 million
- Total current assets: $51.3 million
- Total liabilities: $4.8 million
- Operating expenses increased approximately $9.9 million, or 47% to $30.9 million for the year
- R&D expenses increased 61% to approximately $12.9 million for the year
- Selling, general and administrative expenses increased approximately 38% to approximately $18.0 million for the year
 In his conference call yesterday evening, Murphy seemed rather bullish on their recent research partnerships with L’Oreal and Merck, stressing that such deals could eventually lead to royalty revenue streams. He also made it a point to mention that Organovo is expecting additional partnerships like these in the future:
In his conference call yesterday evening, Murphy seemed rather bullish on their recent research partnerships with L’Oreal and Merck, stressing that such deals could eventually lead to royalty revenue streams. He also made it a point to mention that Organovo is expecting additional partnerships like these in the future:
“Together, these deals represent the type of partnering that Organovo can continue to achieve as we move forward and we have an active discussion pipeline with a number of other companies very interested in 3D bioprinting. We do expect additional agreements to be signed over time though it is not possible for us to get exact timelines due to the nature of the discussions and negotiations.”
Are you an Organovo shareholder? What do you think of their latest report and sneak peek at next quarter’s earnings? Discuss in the Organovo Earnings forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Precision at the Microscale: UK Researchers Advance Medical Devices with BMF’s 3D Printing Tech
University of Nottingham researchers are using Boston Micro Fabrication‘s (BMF) 3D printing technology to develop medical devices that improve compatibility with human tissue. Funded by a UK grant, this project...
3D Printing Webinar and Event Roundup: April 21, 2024
It’s another busy week of webinars and events, starting with Hannover Messe in Germany and continuing with Metalcasting Congress, Chinaplas, TechBlick’s Innovation Festival, and more. Stratasys continues its advanced training...
3D Printing Webinar and Event Roundup: March 17, 2024
It’s another busy week of webinars and events, including SALMED 2024 and AM Forum in Berlin. Stratasys continues its in-person training and is offering two webinars, ASTM is holding a...
3D Printed Micro Antenna is 15% Smaller and 6X Lighter
Horizon Microtechnologies has achieved success in creating a high-frequency D-Band horn antenna through micro 3D printing. However, this achievement did not rely solely on 3D printing; it involved a combination...































