Affecting one to five of every 100,000 children born, Microtia is a condition in which an individual has an undeveloped ear or no external ear features at all. Treatment may consist of creating an ear prosthetic or surgical reconstruction via rib cartilage graft. This latter case involves several operations in which rib cartilage is formed into an ear shape, growing it in place on the scalp, and then raising the ear into place. 3DBio Therapeutics hopes that it has found an easier path with bioprinting.

3DBio is a very credible company that includes Cornell professors Larry Bonassar and Hod Lipson. The latter is one of the sharpest minds in 3D printing and has done a lot of groundbreaking work for many years. The company is running a human Phase 1/2a clinical trial with 11 patients to test the efficacy and safety its AuriNovo product. Designed to mirror the patients’ intact ear, the AuriNovo is a bioprinted collagen ear implant made out of a hydrogel and the patients’ own chondrocytes (cartilage generating cells). Conducted by Cedars-Sinai Pediatric Plastic Surgery, the Microtia-Congenital Ear Deformity Institute, and 3DBio, the clinical trial started in 2021 and is set to run at the earliest until 2023, with the study ending in 2028.
“As a physician who has treated thousands of children with microtia from across the country and around the world, I am inspired by what this technology may mean for microtia patients and their families. This study will allow us to investigate the safety and aesthetic properties of this new procedure for ear reconstruction using the patient’s own cartilage cells. My hope is that AuriNovo will one day become the standard-of-care replacing the current surgical methods for ear reconstruction requiring the harvesting of rib cartilage or the use of porous polyethylene (PPE) implants. The AuriNovo implant requires a less invasive surgical procedure than the use of rib cartilage for reconstruction. We also expect it to result in a more flexible ear than reconstruction with a PPE implant. The AuriNovo living tissue implant is designed to provide a better solution for patients born with microtia by transforming their appearance and building their confidence and self-esteem,” said Pedriatric Microtia surgeon Dr. Arturo Bonilla.

“This is a truly historic moment for patients with microtia, and more broadly, for the regenerative medicine field as we are beginning to demonstrate the real-world application of next-generation tissue engineering technology. It is the culmination of more than seven years of our company’s focused efforts to develop a uniquely differentiated technology platform meeting the FDA’s requirements for therapeutic manufacturing of reconstructive implants. We believe that the microtia clinical trial can provide us not only with robust evidence about the value of this innovative product and the positive impact it can have for microtia patients, but also demonstrate the potential for the technology to provide living tissue implants in other therapeutic areas in the future. Our initial indications focus on cartilage in the reconstructive and orthopedic fields including treating complex nasal defects and spinal degeneration. We look forward to leveraging our platform to solve other high impact, unmet medical needs like lumpectomy reconstruction and eventually expand to organs,” said 3DBio CEO Daniel Cohen.
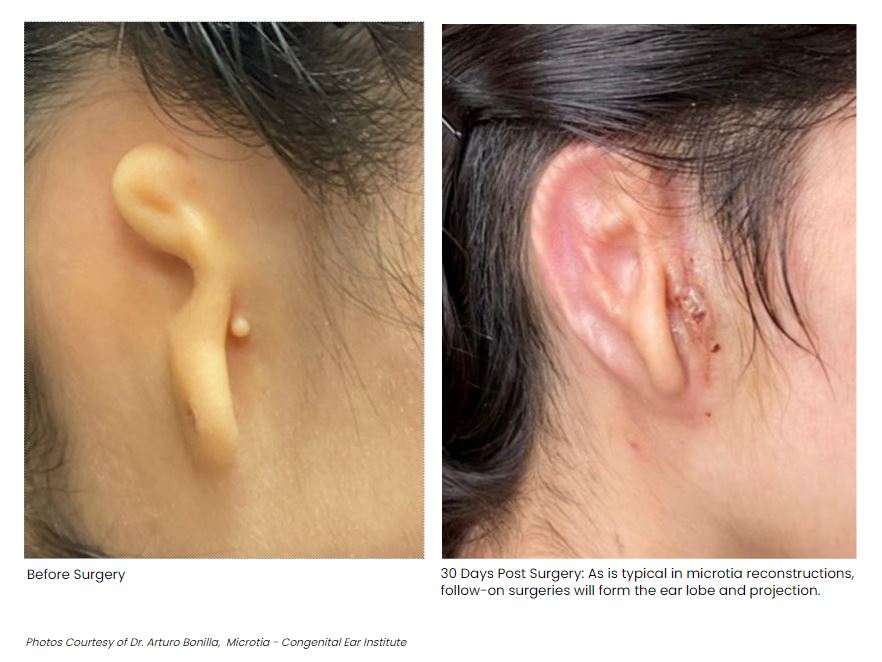
We’ve been looking at various 3D printed ear stories over the years, like this reconstruction and this UK clinical trial. I would expect a lot of similar products to emerge, should funding be available. It’s tough to think of any other bioprinted implant that is lower risk. The company has been working on the implant for seven years and hopes to parlay success in this area to a similar product for nasal defects, lumpectomies, and, later still, orthopedic applications, such as rotator cuff injuries, torn meniscuses, and herniated disks. This is a huge step forward for an impressive firm in an admirable field.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Making 3D Printing Personal: How Faraz Faruqi Is Rethinking Digital Design at MIT CSAIL
What if your 3D printer could think more like an intelligent assistant, able to reason through a design idea, ask questions, and deliver something that works exactly the way the...
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Heating Up: 3D Systems’ Scott Green Discusses 3D Printing’s Potential in the Data Center Industry
The relentless rise of NVIDIA, the steadily increasing pledges of major private and public investments in national infrastructure projects around the world, and the general cultural obsession with AI have...
Formlabs Teams Up with DMG MORI in Japan
In late June, Nick Graham, Chief Revenue Officer at Formlabs, announced on LinkedIn that the company had partnered with DMG MORI, one of the world’s leading machine tool companies, to...

































