For five years, Nanochon has been developing a 3D printed implantable device and novel nanomaterial to treat cartilage loss and damage in joints as an alternative to knee replacements. Now, BICO (formerly Cellink) and the Washington D.C.-based MedTech startup are teaming up to develop 3D printed regenerative joint implants that could start clinical trials as soon as 2023.
As part of the deal, Nanochon will purchase $1.5 million worth of products and services from BICO’s bio automation services subsidiary Scienion to create the joint implants, which promise faster and more successful recoveries for patients while reducing costs to health providers, payers, and patients. At the same time, BICO will invest $400,000 in Nanochon’s Seed funding round to help advance its regenerative medicine technology, scale its manufacturing processes, and accelerate clinical trials. The deal aligns with a crucial part of BICO’s Next Generation Core Industrial Ecosystems that enable tissue engineering, diagnostics, multi-omics, and cell line development.
“Nanochon’s approach to regenerative joint replacement has the potential to improve the lives of tens of millions of people each year,” explained Erik Gatenholm, CEO and co-founder of BICO. “We’re thrilled to leverage BICO’s bio automation manufacturing and technology expertise to help bring this product to millions of patients around the world.”
The agreement will provide BICO’s company Scienion, the opportunity to become Nanochon’s contract manufacturer for cartilage resurfacing implants. The process will leverage several key technologies from the BICO portfolio to enable what is described as a “first of its kind solution” and will also make Scienion a key manufacturing partner for Nanochon over the coming decade.
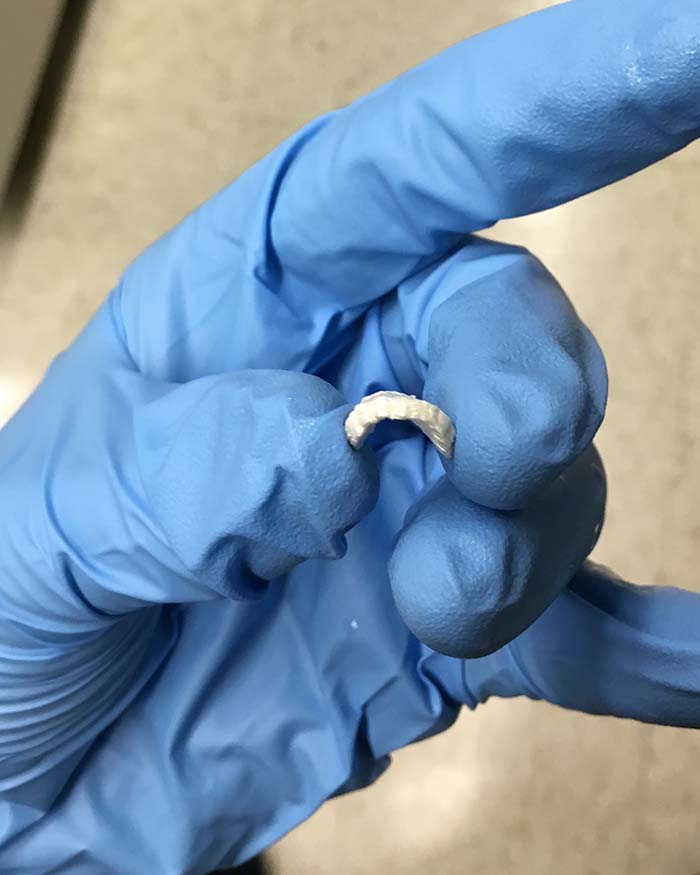
Founded in 2016 by mechanical engineer and life science entrepreneur Ben Holmes and tissue engineer Nathan Castro, Nanochon targets 3D printed products for soft tissue repair in orthopedics. Its first proprietary product, Chondrograft, is a 3D printed polymer implant awaiting FDA approval that fills and regenerates damaged areas of the cartilage surface in joints, typically associated with sports injuries and osteoarthritis. If left untreated, these injuries can cause the patient pain, lead to advanced joint disease, and the need for knee replacement surgery. So Nanochon’s ultimate goal is to commercialize a treatment with much better outcomes and be widely accessible to patients and surgeons.
Built upon research for scaling up existing experimental materials to meet clinical needs, Holmes and Castro saw potential in a composite material called nanostructured polyurethane (nPU) as a cartilage replacement due to its unique chemical and physical properties, which allow it to support rapid stem cell growth and tissue formation. Holmes was exploring strategies for scaling up existing experimental materials to meet clinical needs and discovered that 3D printed nPU has mechanical properties similar to cartilage and can support vascularized bone growth, which is essential for integrating any joint implant and its clinical success.
The duo developed the new proprietary material for a new type of cartilage implant that can replace lost cartilage on a short-term basis and can fuse with the body in the longer term. The implant is targeted at knee injury patients who are younger and more active, typically under 55 years old. The startup detected that most of these patients want to return to more strenuous activities. Current treatment options (like metallic joint replacements) are not good enough, mainly due to invasiveness, complications, or general lack of effectiveness.
Almost one million hip and knee replacement surgeries are performed in the United States every year alone, projected to grow 673% to 3.5 million procedures per year. In fact, the U.S. Centers for Medicare & Medicaid Services estimates that aging Americans will drive annual healthcare spending to $6 trillion by 2027, with much of that coming from aging-related disorders like osteoporosis. Worldwide, estimated orthopedic joint replacement surgeries are roughly 7.7 million, according to GlobalSurg, with a market size of $20 billion in 2019.
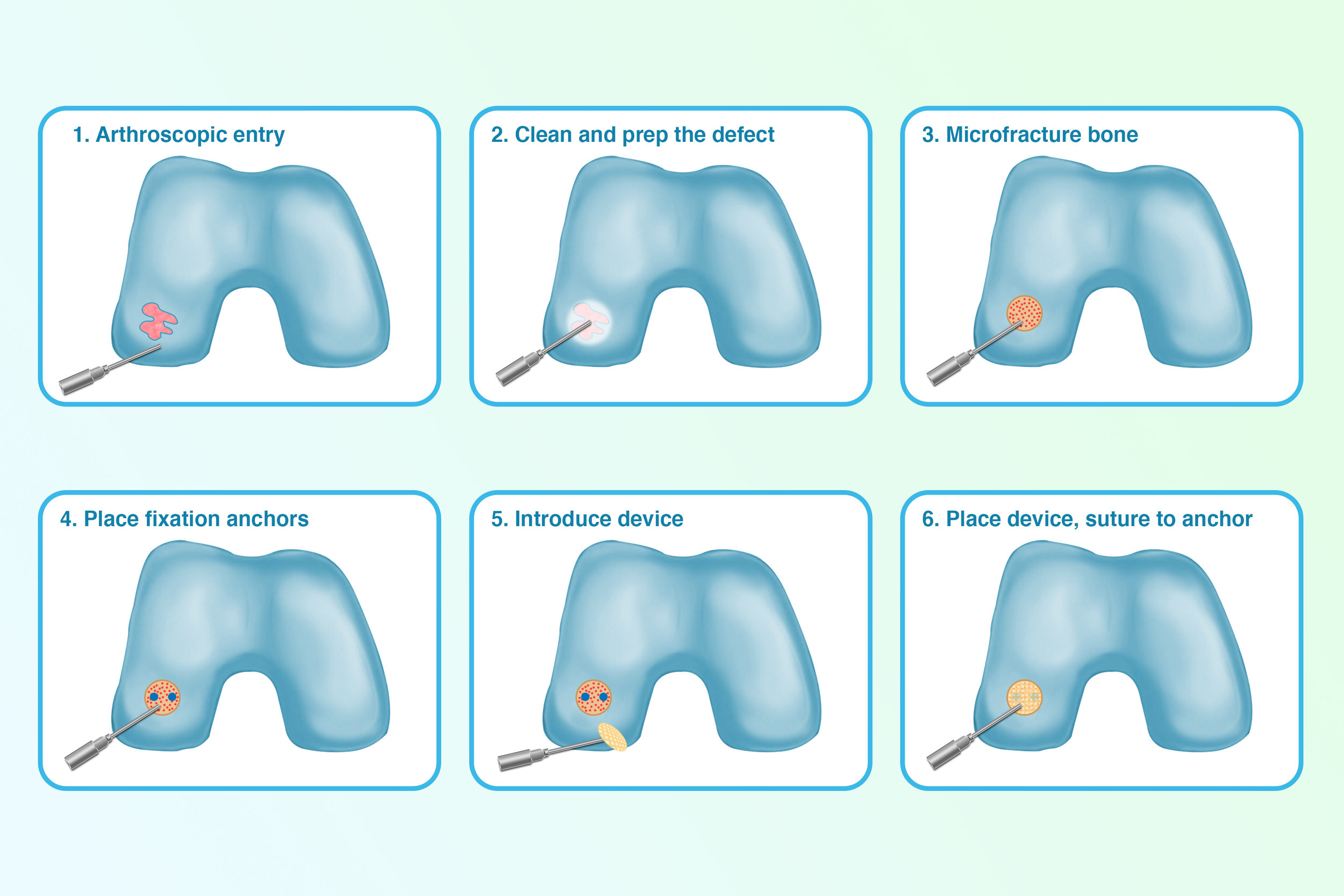
Hip and knee replacements are the most commonly performed joint replacements today. Still, they are expected to fail after 15-20 years due to a lack of organic cartilage growth and the highly disruptive nature of the surgery. Instead, Nanochon’s novel regenerative method would ideally solve this by replacing a highly invasive replacement surgery with a minimally invasive procedure, where 3D printed structures made of a novel polymer nanomaterial are implanted into damaged cartilage to promote healthy cartilage growth.
Working towards that goal, Nanochon has worked with clinical and research partners to conduct several validating and de-risking studies, including a small animal study with Matthew Oetgen, the Chief of Orthopedic Surgery and Sports Medicine at Children’s National Medical Center, and an extensive animal proof of concept trial with Colorado clinical trial management service company CARE Research.
Additionally, in 2017 Nanochon received a Phase I SBIR from the National Science Foundation to develop a production-ready 3D printed medical device for cartilage repair in the knee made from new tissue growth materials and designs compatible with selective laser sintering. The team also executed a manufacturing feasibility study with Belgian 3D printing company Materialise, a leader in 3D printed implants. However, through this new sales deal, Nanochon could become a springboard for future implant designs and cartilage restoration initiatives. Likewise, BICO will continue leveraging 3D printing for healthcare solutions, incorporating even more companies into its customer portfolio.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
How Decibel Landed the Brands Everyone Wants
The first thing Adam Hecht will tell you is that 3D printing already has the technology. The harder part has been finding applications people actually want. That’s the gap Decibel...
HADDY’s Large-Format Robotic 3D Printing to Power Red Cat’s Drone Boat Production
In May 2025, Joris Peels, as is his custom, wrote a prescient article about Unmanned Surface Vehicles (USVs) and Unmanned Underwater Vehicles (UUVs), i.e., drone boats. Listing a multifaceted range...
RusselSmith Brings Additive to Ghana’s Maritime Sector
Nigerian firm RusselSmith is in talks with Ghana’s Maritime Authority (GMA) to bring 3D printing to Ghana’s maritime sector. The Commonwealth Enterprise and Investment Council (CWEIC) helped make the discussion...
3D Printing News Briefs, March 26, 2026: AMUK, IP Dispute, Asbestos, & More
We’re kicking off today’s 3D Printing News Briefs with an America Makes Project Call, and then moving on to additive manufacturing in the UK. Then we’ve got some legal news...