Allegro 3D Receives Almost $1M in Grant Award to Develop Bioprinter
Bioprinting company Allegro 3D has been awarded a National Science Foundation (NSF) Small Business Innovation Research (SBIR) Phase II grant for $997,692. The grant money will support the development of a next-generation high-throughput 3D bioprinter for fabricating tissue models at the industrial scale to advance drug discovery and assay development.
Led by principal investigator and Allegro 3D’s CEO Wei Zhu, the project was previously awarded an SBIR Phase I funding of $299,281 in 2019. The grant was destined for research and development work on a high throughput optical projection (hTOP) bioprinter that could directly print human liver tissue models for applications in preclinical drug screening. During the Phase I grant duration, Allegro 3D focused on developing the hTOP bioprinter and learning how to use it to create 3D hydrogel constructs within a well cell culture plate.
Ultimately, the Phase I stage research aimed to demonstrate the use of hTOP to bioprint in vitro functional human liver tissues using human induced pluripotent stem cells (iPSC). These models are expected to facilitate preclinical drug screening and disease studies, leading to a significant reduction in drug development costs and time to clinical use, significantly improving the public health system. Attempting to optimize and characterize its technology, Allegro 3D went on to apply for a Phase II award and announced on April 27, 2021, that it had received the funding to advance further and eventually commercialize its innovative biotechnology.
As part of the SBIR and Small Business Technology Transfer (STTR) programs, U.S. startups are eligible to apply for a Phase II award once they have been awarded a Phase I grant. The federal government’s SBIR program, which the U.S. Small Business Administration (SBA) has labeled “America’s Seed Fund,” deploys more than $3 billion annually. NSF SBIR awards, in particular, disburse $200 million every year to startups and small businesses across the United States, transforming scientific discovery into products and services with commercial and societal impact. Division Director of the Industrial Innovation and Partnerships at NSF, Andrea Belz, said that the NSF funds the most creative, impactful ideas across all markets and areas of science and engineering. They also support deep technology startups that can guide basic science into meaningful solutions that address tremendous needs.
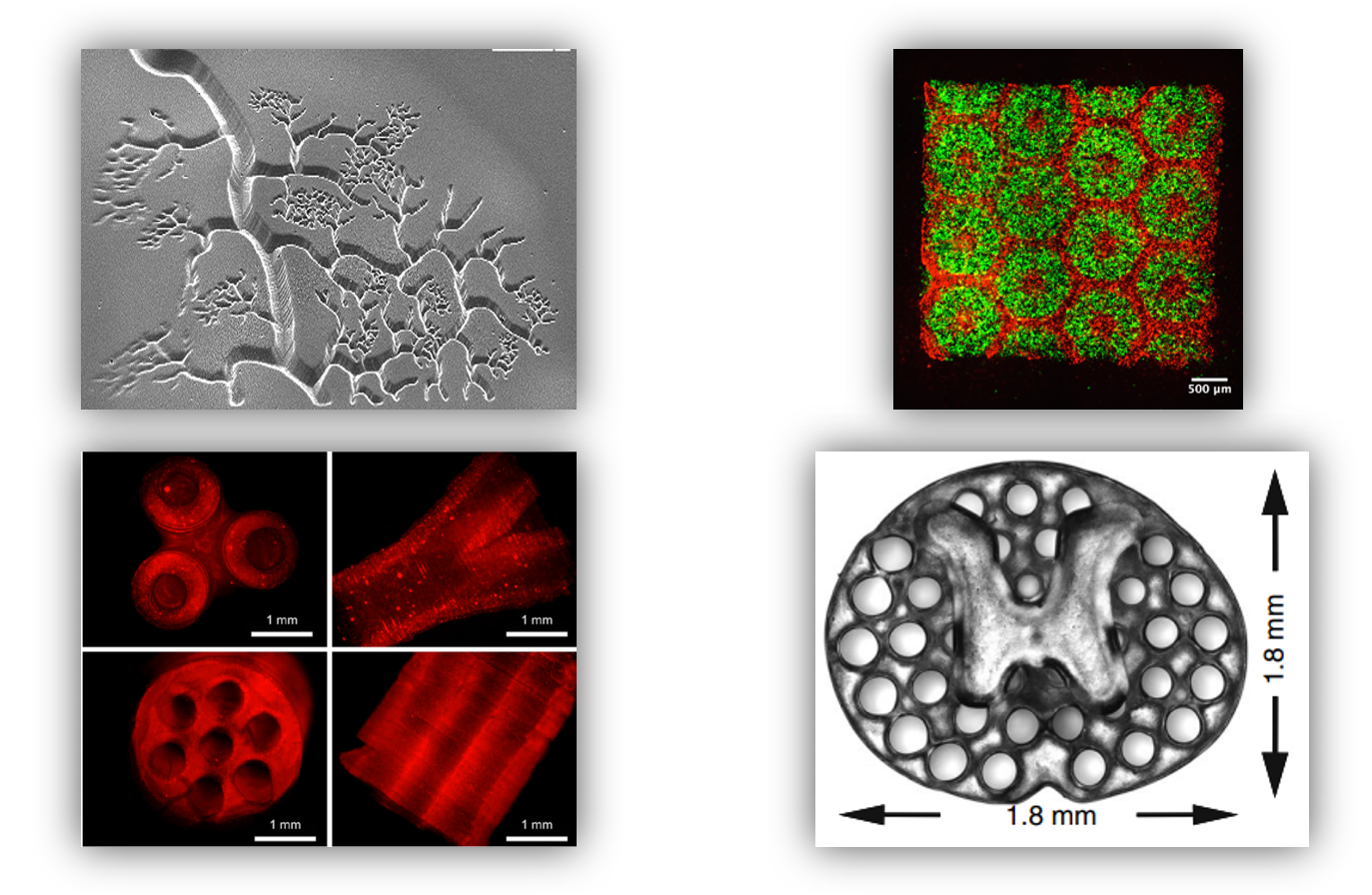
“Allegro 3D strives to provide transformative biofabrication solutions for the life science industry to advance human healthcare. We are grateful for NSF’s continuous support from Phase I to Phase II,” said Zhu. “Allegro 3D has recently launched its first bioprinter, Stemaker Model D, for direct in-well printing of 3D human tissues. With the newly awarded Phase II grant from NSF, we aim to develop a next-generation 3D bioprinter with higher throughput for fabricating tissue models at the industrial scale to advance drug discovery and assay development.”
The company is a spin-off out of the University of California San Diego (UCSD). Founded in 2018 by Zhu and Shaochen Chen, Vice Chairman of the Nanoengineering Department at UCSD, the emerging bioprinting startup is leveraging advanced bioprinting technologies developed at the university to provide customers with transformational biofabrication solutions. The goal is to revolutionize healthcare innovations related to regenerative medicine, drug discovery, and biomedical research.
With the growing adoption of 3D human tissue models in the pharmaceutical industry, there is a critical need for advanced manufacturing systems to enable rapid and streamlined tissue fabrication methods compatible with already established high-throughput screening (HTS) platforms preclinical drug testing. To address these market needs, Allegro 3D is developing advanced 3D bioprinting platforms and materials for in situ drug screening or assay testing directly within the wells. This approach can drastically improve biofabrication workflow efficiencies for the pharmaceutical industry and biomedical research communities.
Consistent with its mission, Allegro 3D has created the first digital light processing (DLP)-based bioprinter, the internationally patented Stemaker Model D. Designed for automated direct printing in multiwell plates, the company claims its platform offers superior resolution, speed, fidelity, and throughput for printing 3D scaffolds and tissues. Allegro 3D also provides a variety of naturally derived and synthetic bioinks with great biocompatibility to support the growth and function of various cells and tissues, as well as 3D products, such as the first reported bioprinted human liver tissue that mimics the microarchitecture and cellular composition of a hepatic lobule (the building blocks of the liver tissue).
Using bioprinting technology commercialized by Allegro 3D, the company founders along with fellow UCSD scientists are also researching a novel 3D bioprinted spinal cord implant that restores function in the hind limbs of rats with spinal cord injuries and has also begun printing human implants to prove the technique could be scalable to humans. Allegro 3D’s research and developments are quite revolutionary and target very specific biological applications. For the upcoming Phase II research, the company has up to two years to develop a novel high-throughput bioprinter for commercialization. Still, with so many unique bioprinting technologies to support the work, this upcoming platform is well poised to create physiologically relevant in vitro tissue models for various applications, including drug testing, assay development, therapeutics, and biomedical research.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: 3D Systems Returns to Growth in Q1 2026
3D Systems (NYSE: DDD) reported one of its strongest quarters in recent years, showing signs that the company may finally be moving past the tough slowdown that has weighed on...
3D People Case Study Details Development of 3D Printed POV Camera Rig
A POV, or Point of View, camera rig, is a wearable support system that helps filmographers capture first-person footage, making the images more immersive. Some good examples of movies shot...
3D Printing News Briefs, May 2, 2026: Soft Robots, Agricultural Waste, & More
In this weekend’s 3D Printing News Briefs, we’ll start off with a multi-laser metal powder bed fusion 3D printer and post-processing news. We’ll end with research into soft robotics and...
Industrial Applications on Display at RAPID 2026: CERATIZIT & 3D Systems
Applications are where it’s at in the additive manufacturing (AM) industry. At the recent RAPID+TCT in Boston, I met with a few companies to learn about some of their very...