A.D.A.M. Seeks Investors for Bone 3D Printing
We recently reported on what appears to be financial woes at Ukrainian 3D printing startup, Kwambio. According to a Ukrainian site, AIN.UA, the company acknowledged some difficulties in terms of product delays but is still in operation. Collateral damage from whatever is taking place there has affected another startup that was originally spun out of Kwambio, but which claims to be completely independent: A.D.A.M. Company.
Along with the news about Kwambio’s operations, what was apparently a fraudulent press release about A.D.A.M. was sent to news distribution service ZEX PR WIRE. The “announcement” from the firm claimed that A.D.A.M. was shutting down, with a quote from its CEO, Denys Gurak. The release event went so far as to quote Gurak as saying that the investigation into Kwambio at AIN.UA was accurate.
3DPrint.com received an email from A.D.A.M. informing us that the press release we quoted was fraudulent, providing numerous pieces of evidence, from emails to business activity, indicating that A.D.A.M. continues to work toward its goal of 3D printing bones and more. Dmitriy Skomorokhov, CEO of Kwambio, is still the vice president of Business Development at A.D.A.M., but is not on the Board of Directors which is made up solely of Denys Gurak. Though Kwambio was an initial investor in A.D.A.M., it does not have a right to exercise control over the company and there are no other current or former managers from Kwambio at A.D.A.M.
Given its apparent relative independence from Kwambio and the fact that it shows promise in the field of medical 3D printing, we spoke to Gurak to learn more about exactly what it has accomplished so far and where it plans to go.
Established in 2018 in Connecticut, the startup is developing an on-demand personalized implant manufacturing infrastructure, meaning that it isn’t dedicated solely to 3D printing implants, but a complete set of services connected to those implants. This means, beginning with 3D modeling to ending with sterilized, personalized medical devices ready for implantation.
“The initial vision was to create a car shop-like infrastructure where any part of the human body could be replaced after it breaks,” Gurak said.
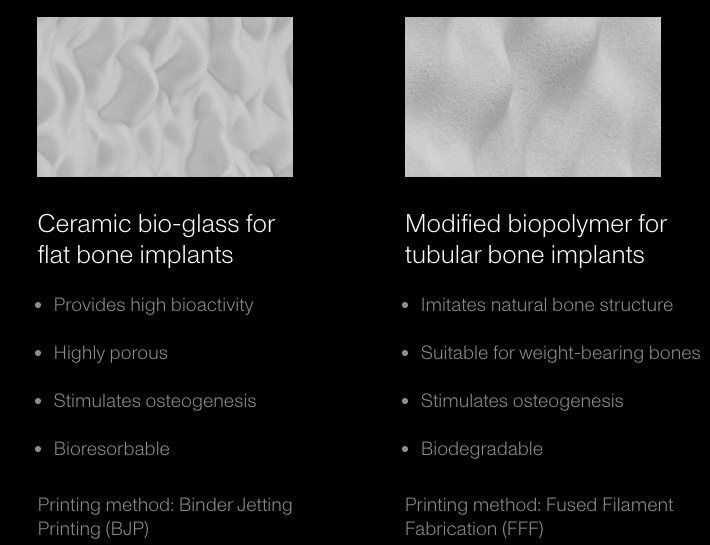
This began with a ceramic binder jetting process, initially developed under Kwambio, as well as fused filament fabrication, which the startup thought could be directed toward 3D printing bioceramic (borosilicate glass-hydroxyapatite and borosilicate glass-bioglass) and modified biopolymer (polycaprolactone hydroxyapatite, polycaprolactone-bioglass) bone implants.
Naturally, A.D.A.M.’s technology offers advantages over traditional implants similar to other 3D printing technologies in that patient-specific devices can be made on-demand and with osteogenic characteristics. However, it may also offer improvements over some 3D printing processes as well.
The benefit of these materials over 3D printed metal, the primary method for bone transplants in additive manufacturing, include lower costs and higher production speeds. There are polymer 3D printing firms dedicated to medical applications, such as Oxford Performance Materials, but their equipment, materials and focus are highly specialized. It’s also worth noting that the startup’s implants are meant to be bioresorbable, so that as bone grows back, the 3D printed device degrades over time.
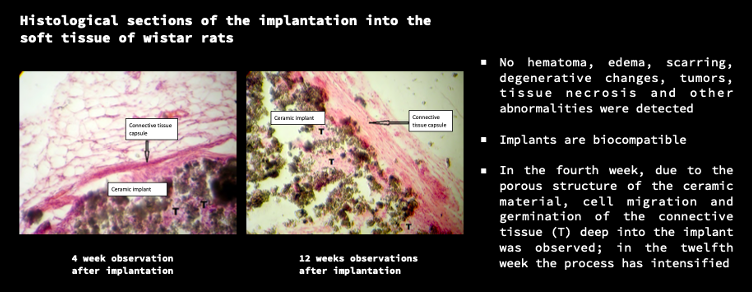
So far, A.D.A.M. has achieved some significant milestones. The firm has completed clinical trials on the implantation of bone implants in rats, which led to positive feedback from the U.S. Food and Drug Administration (FDA). According to the company, the FDA confirmed 510 (k) clearance eligibility for their 3D printed ceramic and polymer bone implants.
“[This] means that we don’t have to do human trials on our implantable medical devices. We can base our approval methodology on predicate devices,” Gurak said. “And that means that we’ll be able to deliver our product, installed at the point of care, to 3D print bones.”
With this certification workflow in place, it would mean that the company could reduce approval time from five years to one-and-a-half years. A.D.A.M. believes that it would be able to receive approval by Q2 of 2022.
Ideally, the firm would see bone transplants 3D printed on-site via hospital labs, but it will begin by delivering the items directly to hospitals. Even with a delivery model, Gurak believes that it would be able to reduce production time from several weeks to just three days, cutting costs by roughly three times.
The company has plenty of guidance from established experts. In 2020, it joined the University of Connecticut Technology Incubation Program and is a part of the BioCT accelerator in Connecticut. It has also just announced that Dr. Mark Horowitz, Professor at the Department of Orthopaedics and Rehabilitation at the Yale University School of Medicine and the Director at Yale Orthopaedic Histology and Histomorphometry Laboratory, has joined A.D.A.M.’s Advisory Board as a Chief Trials Advisor, which will see him coordinate the animal studies for biopolymer and bioceramic bone implants.
The startup’s Chief Development Advisor is Dr. Adnan Mjalli, founder of vTv Therapeutics, which assists in the clinical approval and commercialization of new medicines. He is also the CEO of High Point Clinical Trials Center, Chairman of PharmaCore, and CEO and Chairman of the Mjalli Investment Group. A.D.A.M.’s Chief Scientific Advisor is Dr. Anthony Tether, a former Director of the Defense Advanced Research Projects Agency (DARPA), where he initiated the DARPA Grand Challenge series. He also re-formed the Sequoia Group, which provides management and strategy development services to government and industry.
Once its bone products are successful, A.D.A.M. would seek to expand globally to the Asia-Pacific region, Europe, the Middle East and North Africa. In the meantime, the startup is aiming to conduct further animal studies at the University of Connecticut. The research may expand from rats to dogs or pigs, based on FDA advice. This will include adding advisors and consultants who can further guide the process. Gurak said that it will take about a year to conduct these trials and another half-year for FDA approval.
Also essential to a successful business implementation is executing the best business model. A.D.A.M. will be validating its business model with hospitals and insurance companies in the U.S. At the same time, it will seek to secure its IP portfolio and expand to polymer-based 3D printing of tissues, possibly including heart valves, bronchial tubes and blood vessels.
With this in mind, the startup has initiated Series A fundraising has been accepted onto a major equity crowdfunding platform with accredited investors, though it is unable to disclose which one due to SEC regulatory reasons. Once it is able to secure funding, the company shows promise for the future of 3D printing medical implants. Whether or not it is able to achieve all that it strives for will become clear over time.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Colibrium Additive Gets $31 Million NAVAIR Contract
Colibrium Additive has been awarded a $31 million contract by the Naval Air Systems Command (NAVAIR). The contract is part of the Additive Manufacturing Capability initiative, focused on qualification and...
Nikon AM Synergy Secures DLA Contract for On-Demand Military Parts
Nikon AM Synergy has gotten a DLA contract under the JAMA IV IDIQ Pilot Parts Program. The contract with the Defense Logistics Agency (DLA) is a step forward for Nikon...
ExOne Cuts Costs for U.S. Customers as Printhead Production Moves to Detroit
ExOne Global Holdings, created through the 2025 integration of ExOne and voxeljet, is making changes across its U.S. operations. These include starting printhead manufacturing in the Detroit area and lowering...
3D Printing News Briefs, April 8, 2026: LiDAR Scanning, Vapor Smoothing, FDM Optimization, & More
We’ll kick off today’s 3D Printing News Briefs with some 3D scanning news from Artec 3D, and then move on to new America Makes Project Calls. Then, Raise3D and AMT...