FluidForm’s Adam Feinberg will be speaking at 3DPrint.com’s upcoming AMS online industry summit (Feb 9-10, 2021). Register here.
On January 16th, 2021, President Joe Biden introduced his science team for the incoming administration and made it clear that science and technology will see growing investment over the next 4 years. Even more exciting was Biden’s statement to imagine a future with “3-D printers restoring tissue after traumatic injuries and hospitals printing organs for organ transplant.”
It seems that this may finally be the decade that 3-D bioprinting moves from the realm of science fiction to a widely available clinical reality. Indeed, the past 5 years has seen major research advances in 3-D bioprinting that include perfusable vascular-like networks for rapidly building tissues, functional pieces of organs such as breathing lung alveoli and beating heart chambers, implantable scaffolds for repair of the spinal cord and the musculoskeletal system, and even full-size human heart scaffolds. However, it is important that the industry remains clear-eyed about the challenges that stand between where we are today, and the future that we are all striving to achieve.
It has been over 30 years since the field of tissue engineering first promised the ability to build replacement tissues, and nearly a decade since Organovo emerged as the first public company promising that 3-D bioprinting is the technology to make it happen. But the reality has been harsh. Today there are essentially no tissue engineered medical products on the market. The question many of us in the field have is whether now is the time that industry and the federal government will make the large-scale investments necessary to make good on the vision of 3-D bioprinted tissues and organs.
At FluidForm we believe the answer is yes, and that science will lead the way. More specifically, that we need to develop not just better printers and materials, but a deep understanding of how living cells interact with the scaffolds we build to develop instructive environments that speak the language of biology. Built on a technology known as Freeform Reversible Embedding of Suspended Hydrogels (FRESH™) printing licensed from Carnegie Mellon University, FRESH™ leverages a unique gel-like environment within which we can 3-D bioprint the widest combination of cells, bioinks, and biomaterials possible in nearly any direction to match properties of living tissue.
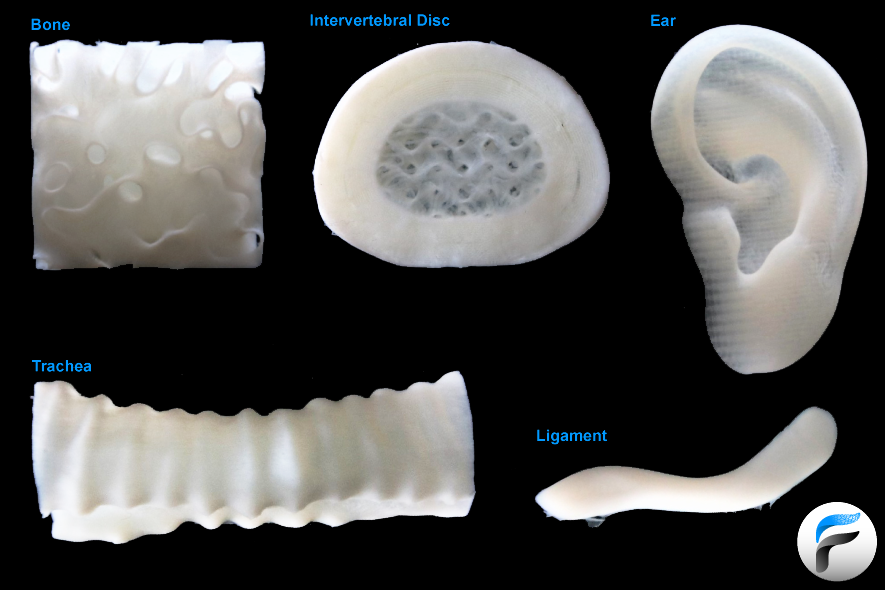
As a starting point, one can look at the human body and the way tissues and organs are created for inspiration. By dry weight, each of us is more collagen than anything else. In fact, collagen is the primary extracellular matrix (ECM) protein of nearly every tissue and organ in the body, making it one of the most exciting biomaterials to use in 3-D bioprinting. In 2019 our team published an improved version of FRESH™ printing in Science demonstrating that not only can native collagen be printed with high resolution, but that we can use this to create functional parts of the human heart. Importantly, the impact of using collagen for biofabrication is its ubiquity, enabling FluidForm to build tissue scaffolds for bone, cartilage, connective tissue, and many other applications.
Looking forward, we see FRESH™ 3-D bioprinting as a foundational technology, not because of what we have achieved to date, but because of what we see being built with FRESH™ in the future. As noted, it is going to take rigorous science to drive us towards realizing the vision of 3-D bioprinted organs that President Biden has brought to national attention. This is a primary reason that FluidForm’s first commercial product is LifeSupport™, a version of the FRESH™ support gel that we have made available through major partners to enable academic and industrial researchers to do innovative science and develop new high-impact applications. To that end, our internal development projects are focused on accelerating translation forward to the clinic and include medical devices, regenerative scaffolds for soft tissue repairs, biopharma models, and a 1st generation of 3-D bioprinted organs.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing News Briefs, April 13, 2024: Robotics, Orthotics, & Hypersonics
In 3D Printing News Briefs today, we’re focusing first on robotics, as Carnegie Mellon University’s new Robotics Innovation Center will house several community outreach programs, and Ugogo3D is now working...
Rail Giant Alstom Saves $15M with 3D Printing Automation Software 3D Spark
3D Spark has entered into a three-year deal with the rail giant Alstom. Alstom, a transport behemoth with annual revenues of $16 billion, specializes in the manufacture of trains, trams,...
Meltio Expands Global Reach with New Partnerships in the Americas and Europe
Spanish 3D printing manufacturer Meltio has expanded its sales network across the globe. With the addition of three new partners in the United States, Brazil, Argentina, and Italy, Meltio aims...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...