3D Systems’ Gautam Gupta and Katie Weimer will be speaking at 3DPrint.com’s upcoming AMS online industry summit (Feb 9-10, 2021). Register here.
Authors: Gautam Gupta, Ph.D., Vice President and General Manager, Medical Devices, 3D Systems and Zachary Wilczynski, M.S., Senior Product Development Engineer, 3D Systems
Nearly 10 years ago, the first FDA 510(k) clearance in total knee arthroplasty brought the combination of personalized medicine, additive manufacturing, and orthopedic surgery into the spotlight. An increase in the use of patient-specific surgical planning solutions across many surgical specialties has resulted in a greater need for medical device companies to provide patient-specific workflows and tools alongside their off-the-shelf implant systems.
Total ankle arthroplasty is one of the fastest-growing arthroplasty segments, with a compound annual growth rate of 8.4% and $327M in revenue anticipated by 2026.1 There are currently 10 total ankle systems with 510(k) clearances in the US, and two companies with patient-specific technology to accompany the implant (Wright Medical/Stryker and Exactech).2 The Exactech Vantage® Total Ankle System, one of the current total ankle designs received its first FDA clearance in 2016.
3D Systems and Exactech have collaborated for more than a decade, since the development of the first FDA-cleared, 3D-printed orthopedic implant. The combination of 3D Systems’ VSP® surgical planning solution and Exactech’s Vantage Total Ankle System created the opportunity for innovative and collaborative development of patient-specific cutting guides. 3D Systems’ provided a proven digital workflow, additive manufacturing expertise, and regulatory understanding, while Exactech provided implant system aptitude and an eager surgeon design team. Together, this combination of knowledge and application development created Vantage Ankle PSI.
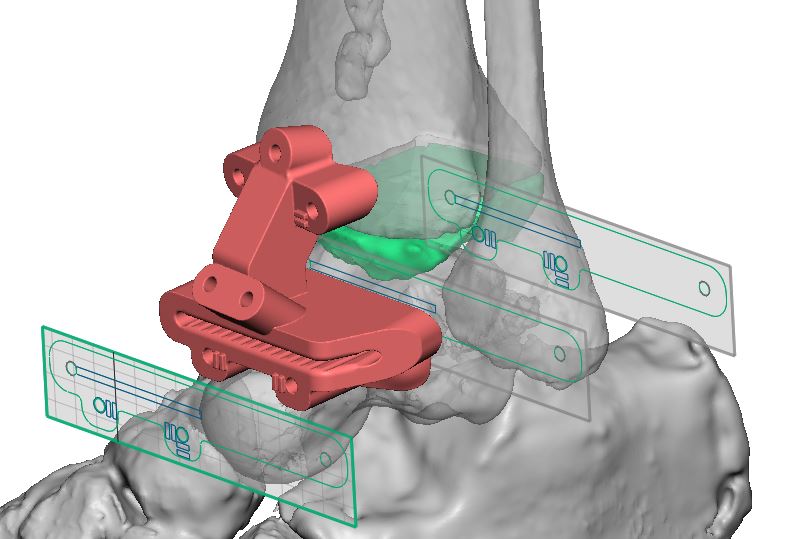
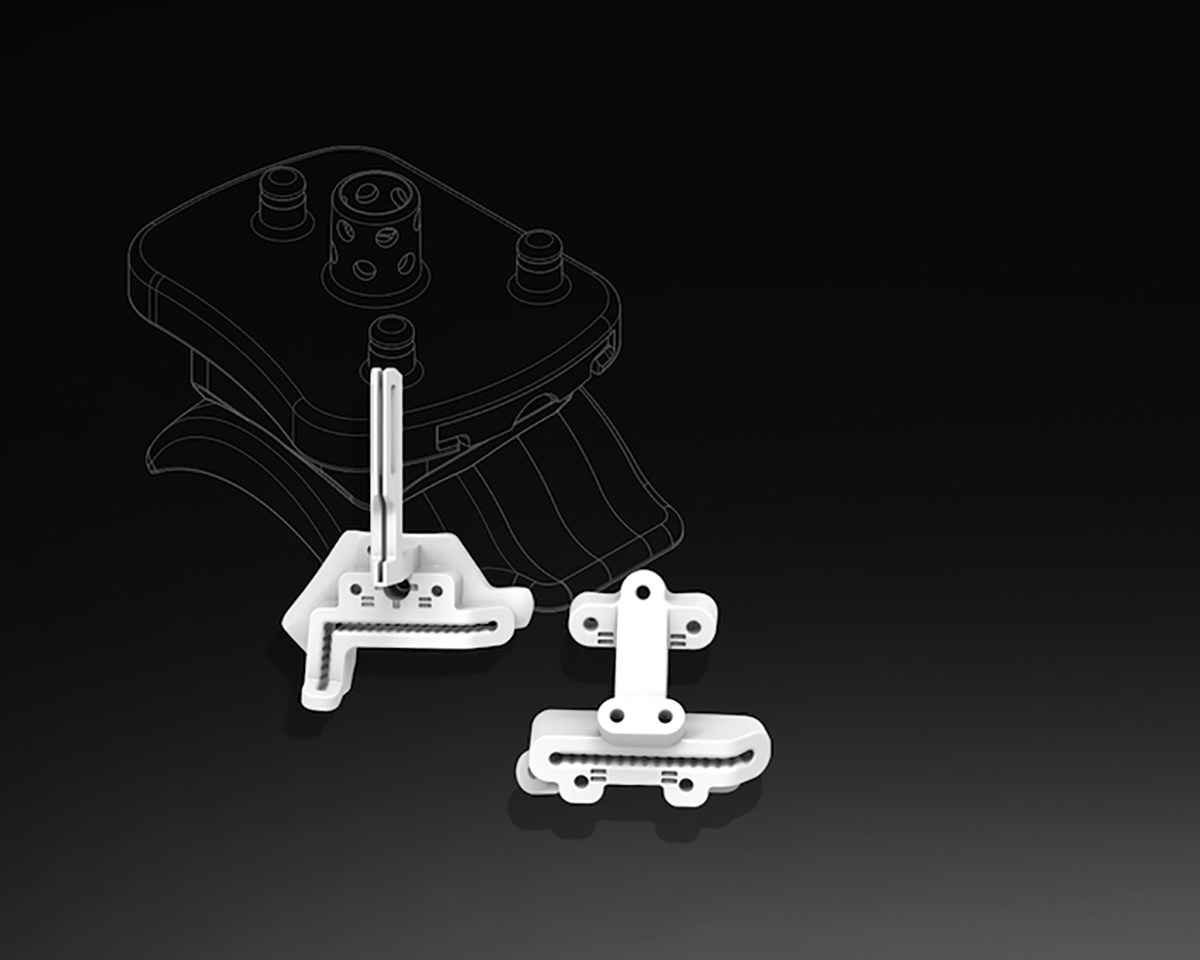
Vantage Ankle PSI is the second patient-specific total ankle system on the market in the US and consists of cutting guides and anatomic models produced via selective laser sintering (SLS) technology. The digital workflow begins with CT imaging data, segmented using D2P®, followed by device design using Geomagic FreeForm®. Once the design is complete the patient-matched guides are 3D-printed on the ProX® SLS platform using the biocompatible DuraForm® ProX PA material. This combination of printer and material provides a repeatable and durable solution for anatomic model and guide manufacture. The DuraForm ProX PA cutting guides demonstrated consistent accuracy and ability to withstand surgical forces during benchtop and cadaveric studies. 3D Systems’ regulatory experience in patient-specific applications of additive manufacturing enabled the submission and clearance of the Vantage Ankle PSI.
Utilizing pre-operative planning and patient-specific tools, Vantage Ankle PSI aims to reduce time in the operating room and improve patient outcomes.3, 4 3D Systems’ team of engineers complete imaging segmentation, pre-surgical planning, and design with interactive feedback from surgeons across the country as part of the VSP surgical planning workflow. The surgeon then receives anatomic models that allow them to confirm the fit of the patient-specific guides, which increases surgeon confidence during the procedure.5 Vantage Ankle PSI cutting guides are uniquely designed to allow the surgeon to cut through the saw slots on each nylon guide, reducing the number of operative steps per case. The Vantage Ankle PSI has been utilized by several surgeons to complete nine cases thus far in a limited pilot launch after FDA clearance.
Orthopedic surgery continues to provide opportunities for innovation through the use of additive manufacturing and collaborative development agreements. Additive manufacturing technology is continually evolving with more efficient printers, innovative materials, and an increased presence at point-of-care. Patient-specific products have become a staple in many orthopedic procedures with total ankle arthroplasty taking the spotlight as of late. Implant manufacturers and surgeons desire innovative and patient-centric products, additive manufacturing often answers this need.
- https://www.medgadget.com/2019/09/total-ankle-replacement-market-2019-overview-industry-opportunities-and-outlook-to-2026.html
- https://www.podiatrytoday.com/keys-maximizing-outcomes-fourth-generation-total-ankle-replacements
- Patel A, Levine J, Brecht L, Saadeh P, Hirsch DL: Digital technologies in mandibular pathology and reconstruction. Atlas Oral Maxillofacial Surg Clin N Am 20:95-106, 2012
- Roser SM, Ramachandra S, Blair H, Grist W, Carlson GW, Christensen AM, Weimer KA, Steed MB: The accuracy of virtual surgical planning in free fibula mandibular reconstruction: comparison of planned and final results. J Oral Maxillofac Surg 68:2824-2832, 2010
- McCormick S, Drew S: Virtual model surgery for efficient planning and surgical performance. J Oral Maxillofac Surg 69:638-644, 2011
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Inside The Barnes Global Advisors’ Vision for a Stronger AM Ecosystem
As additive manufacturing (AM) continues to revolutionize the industrial landscape, Pittsburgh-based consultancy The Barnes Global Advisors (TBGA) is helping shape what that future looks like. As the largest independent AM...
Ruggedized: How USMC Innovation Officer Matt Pine Navigates 3D Printing in the Military
Disclaimer: Matt Pine’s views are not the views of the Department of Defense nor the U.S. Marine Corps Throughout this decade thus far, the military’s adoption of additive manufacturing (AM)...
U.S. Congress Calls Out 3D Printing in Proposal for Commercial Reserve Manufacturing Network
Last week, the U.S. House of Representatives’ Appropriations Committee moved the FY 2026 defense bill forward to the House floor. Included in the legislation is a $131 million proposal for...