Cells aren’t enough: A materials-centric approach to tissue engineering

Dimension Inx’s Caralynn Nowinski Collens will be speaking at 3DPrint.com’s upcoming AMS online industry summit (Feb 9-10, 2021). Register here.
Even with the clinical success seen with cell therapies, they are not sufficient as a regenerative strategy. In particular, we have observed inefficient cell survival and growth in injured tissue environments such as cardiac infarction and osteochondral lesions. Accordingly, medical device and biopharma companies are beginning to recognize the renewed potential for tissue engineering.
We are at a unique moment in time in which advances in cellular biology, manufacturing, and biomaterials are converging. This convergence has created an opportunity to dramatically improve the way we design and manufacture products for tissue repair and regeneration. Utilizing these new tools, tissue engineers can design for both biofunctionality and manufacturability – and their interdependencies.
Form designed for function
It is generally accepted that what an implant is made of (composition) and how porous it is (microstructure) are critical for tissue remodeling and regeneration. A growing body of evidence demonstrates the importance of the cellular microenvironment and the ability to alter cell behavior by controlling that microenvironment. The challenge is engineering a product that is capable of providing the right cues at the same time to generate the desired biological response – and doing it in a way that is affordable and scalable.
For instance, in bone repair, hydroxyapatite granules have long been used to patch defects. Why? Because that’s what our bones are made of. Our cells can react intimately with the granules and remodel them over time as they would do with natural bone. However, just placing a block of solid hydroxyapatite in a bony defect is not going to regenerate bone.
That’s where microstructure comes into play. For example, we know that porosity is important. That’s why we see porous titanium now for orthopedic applications. Cells and eventually tissue infiltrate the implant through the pores and anchor it more tightly in place. Again, there’s a greater intimacy between the product and our natural tissue. However, with titanium or plastic, that intimacy is all that happens. The tissue doesn’t actually remodel because the composition is not right. Further, the porosity in this case is limited to the architecture, not to the material itself.

Utilizing extrusion 3D-printing and a novel formulation of hydroxyapatite, engineers at Dimension Inx have designed a construct with a composition and microarchitecture that promotes bone formation.
Today, new systems are emerging that allow product development engineers and scientists to design regenerative constructs with the precise interplay among a number influential factors – biochemical, structural, mechanical, and more – to achieve the desired function.
Architecting the microenvironment
By designing regenerative microenvironments, the construct itself provides the necessary instructions to surrounding or incorporated cells that promote the desired biological response such as cell infiltration, matrix deposition, and new blood vessel formation.
To achieve such regenerative templates, engineers must be empowered with the necessary design and manufacturing tools. This is what’s changing in the industry. For 30 years, tissue engineers have had a limited set of tools, but today we have new ways of controlling the three-dimensional microenvironment. 3D-printing allows for the spatial organization of material and cells to create more complex and reproducible artificial environments that may lead to better biological outcomes.
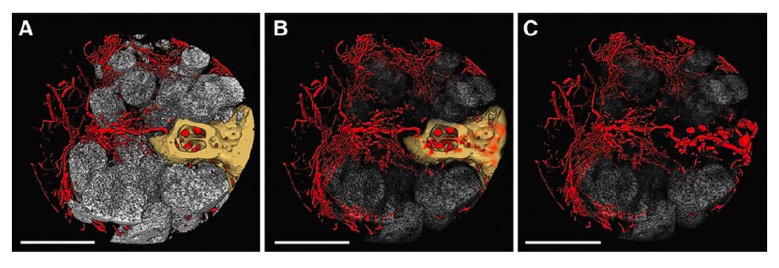
Engineers at Dimension Inx, along with their collaborators, have demonstrated the importance of the microenvironment to instruct cell behavior, including vascularization. These microCT images illustrate new bone growth and blood vessel formation in a rat spine following implantation of a proprietary 3D-printed scaffold. Bone (yellow) and scaffold struts (white) are shown with progressively greater transparency from A to C to allow for better visualization of the blood vessels (red). Scale bars = 1mm. (From: Hallman M., et al. Influence of Geometry and Architecture on the In Vivo Success of 3D-Printed Scaffolds for Spinal Fusion. Tissue Eng Part A. 2021 Jan;27(1-2):26-36. http://doi.org/10.1089/ten.tea.2020.0004)
At the core, this requires access to a broad range of biomaterials that can be architected and tuned to create the ideal microenvironment for regeneration. Composition and structure cannot be limited to the inherent properties of a single source biomaterial. Plus, the construct must contain or be compatible with bioactive factors, e.g., growth factors, extracellular proteins, and cells, that can provide the right cellular signaling.
Such constructs need to be designed to have structural integrity yet be agile enough to integrate with and adapt to the body’s dynamic environment. They must hold shape but also exhibit the right porosity, pore size and shape, and degradation profile. Moreover, multi-level porosity is key, such that there are pores within the material fibers themselves at the micro- and nano-scale. Creating the ideal microenvironment will likely require multiple materials working in concert with each other, particularly for more complex tissues.
When all the necessary features align, only then can these constructs promote tissue restoration. Of course, this approach does not minimize the importance of cells. In fact, one could argue that this approach is designed specifically for cells. Whether seeding cells into a construct, printing cells, or relying on interaction with endogenous cells, ultimate healing results when the right interplay is at work such that cells self-organize and remodel the temporary regenerative template into viable long-lasting tissues.
A step closer to commercialization?
This materials-centric approach offers promise for the future commercialization of regenerative medical products. Beyond the technical advantages, it may also facilitate regulatory and manufacturing strategies that can overcome challenges restraining the field (see https://3dprint.com/275909/three-key-hurdles-restraining-the-growth-of-the-tissue-engineering-field/).
If regenerative “templates” can be reproducibly manufactured from materials already deemed safe by regulatory agencies, and not hampered by the limitations of cell expansion or the need to develop and test novel engineering hardware, perhaps an avenue exists to accelerate the commercial development of TEMPs.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
AM Drilldown: the Beginning of 3D Printing’s Next Phase in the Energy Sector
For much of the last decade, many have pushed an unjustifiably optimistic view of global energy consumption, along the lines of, fossil fuels are “on their way out.” Sadly, this...
Mikhail Gladkikh on Digital Inventory: “Think of It as Netflix for Manufacturing”
As manufacturers continue looking for ways to reduce supply chain risk, additive manufacturing (AM) is increasingly being discussed as more than just a production tool. Across aerospace, energy, defense, and...
ROBOZE Buys Dimanex Assets to Build “Physical AI” Platform
Dutch firm Dimanex got its start as an MRO platform for the railways. The company had a contract with the Dutch Army in 2018, and later that year signed one...
3D Printing Financials: Xometry Surges After Record Quarter and Siemens Deal
Shares of Xometry (Nasdaq: XMTR) surged on Thursday, May 7, after the company reported record first-quarter 2026 results and announced a major partnership with Siemens. The stock climbed as much...















































