On the centuries-old quest to cure cancer, researchers have made major advances in learning more complex details about how to prevent, diagnose, treat, and survive one of the world’s biggest killers. From successful immunotherapies to the growing role of precision medicine, these major advances are helping clinicians save lives. But with approximately 14 million new cases and close to 10 million deaths every year, it is difficult to say that the battle against cancer is close to being won. Moreover, experts consider that, once cancer spreads, or metastasizes, from one place in the body to another, the chances of survival decrease to 10%, making this the most deadly feature of the disease.
Up until now, advances in the management of systemic metastasis have been scarce, but scientists from the Lawrence Livermore National Laboratory (LLNL) developed a unique approach that they believe will help clinicians and researchers anticipate the spread of cancer within individual patients. By pairing 3D bioprinting technology and advanced computational flow simulations, a team of experts believes they have laid the foundation for developing a predictive capability to understand tumor cell attachment to blood vessels, the first step in secondary tumor formation during cancer metastasis.
The discovery was described in a paper published in the journal Science Advances as a new approach to training computational models on biological processes and provides insights into how and why cancer cells metastasize in certain areas of the vasculature. Using a custom-built extrusion-based bioprinter, LLNL researchers 3D printed living human brain vasculature and paired it with advanced computational simulations to address the physics involved in cancer spreading via metastasis.
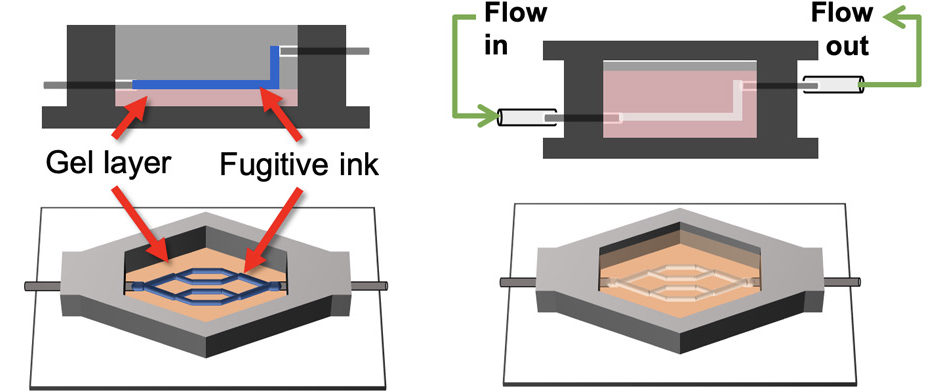
Bioprinting endothelialized vascular beds with complex geometry using the sacrificial ink method. (Image courtesy of Claire Robertson/LLNL)
According to LLNL biomedical engineer Monica Moya, principal investigator of the study and lead researcher on LLNL’s bio-printed vasculature device, tumor cells tend to escape from a primary tumor and travel through the vasculature, where they eventually attach to a vessel wall, pass through the endothelium into the tissue and grow like a seed in the soil, usually in areas such as forks in the blood vessels. However, by combining bioengineering and computation to analyze the physics behind circulating tumor cell (CTC) behavior and the cells’ attachment to the vascular endothelium – the layer of cells that line the interior surface of blood vessels – the team provided new insight into the mechanisms underlying CTC flow dynamics and attachment behaviors.
“Computational modeling is definitely a useful tool, but you still need to benchmark it against something real,” Moya said. “With this approach, we can make the biology as simple and clean as it needs to be to validate the models, and we can increase the complexity, both in the biology and the computational model. Physics matters in biology, and this paper really sets the framework of how you can use these in vitro models, paired with simulations, to tease apart the contribution of the biology and the physics and really bring a strength to the field that has been lacking.”
Moya also explained that scientists typically use animal models to understand how physics contributes to cancer metastasis, yet these models are not entirely useful or relevant to human biology. Instead, her team created an in vitro system by 3D printing vasculature out of human cerebral endothelial cells and subjected them to flow conditions in the fluidic platform.
After the cells completely covered the device’s channels, they aligned within the vessels and after about a week, researchers injected a breast cancer cell line into the device to see how and where the tumor cells began to metastasize inside the newly formed brain vasculature. Experts consider that some regions of the brain are more susceptible to breast cancer metastasis, but the mechanisms of local susceptibility remain poorly understood and impossible to prevent, which is why this research can be crucial to understand the behavior of breast cancer cells once they metastasize.

Attachment sites of CTCs show differences between acellular (B) and endothelialized (A) channels. Full acellular vascular bed exhibits a greater CTC burden compared to endothelialized. (Image courtesy of Claire Robertson/LLNL)
Researchers said that after the tumor cells circulated at physiological flow rates, Claire Robertson, a Lawrence Fellow who works on developing models of early breast cancer, mapped more than 6,000 cells that adhered to the vessel walls and compared them to local biophysics. These experimental results were then compared to 3D computational simulations replicating geometries gathered from the 3D maps to reproduce the exact geometry of the bioprinted vessels, enabling highly accurate fluid dynamics analysis of the attachment conditions, according to the study.
“Adapting this advanced bioprinting process to engineer functional, perfusable human brain vasculature was extremely challenging, however we now have a strong handle on the technique and can potentially fabricate a wide variety of living human tissue constructs,” said lead author and LLNL research staff engineer William Hynes. “Using this approach, we were able to test, observe and measure a biological phenomenon that was previously impossible, and we will continue to iterate on these findings to shed light on how and when circulating tumor cells choose their targets in vivo. By pairing our engineered platform with computational modeling, we can directly interrogate the behavior of metastatic cells and the rules that govern them far more rapidly than through experimentation alone.”
LLNL scientists used an algorithm called “HARVEY“, developed by biomedical engineer Amanda Randles from Duke University, to capture flow rates throughout the human body that would be difficult to do by in vitro devices alone, allowing researchers to “turn off” specific parameters that wouldn’t be possible in experiments, such as the elasticity of a cell, number of cell receptors or flow profile, Randles said.
This new approach could allow researchers to decouple the biological and physical contributions that drive the metastatic colonization of organs. The researchers expect the simulations will be used to predict locations where tumors will spread, enabling targeted screening of high-risk patients and therapeutic intervention aimed at the most vulnerable areas. This, according to Moya, could offer clinicians an alternate way to treat patients, allowing them to use MRIs to simulate where circulating tumor cells are likely to get stuck, which areas to focus their attention to greatly enhance the efficacy of treatments.
As part of their ongoing research, the team is looking for more funding to support additional bioprinting efforts and increase the complexity of bioprinted vasculature to further examine the role of biophysics in the progression of the deadly disease. This new approach could eventually replicate what really happens in the body when cancer metastasizes, giving the healthcare industry a new tool to customize treatment for millions of patients.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
HP Webinar Breaks Down Where Industrial Filament 3D Printing Works Best
As additive manufacturing continues to move into production, one question keeps coming up: not just whether a technology works, but where it actually makes sense to use it. HP’s upcoming...
ATO and Dynamism Partner to Expand Metal Powder Production in the U.S., Announced at AMS 2026
ATO Technology is expanding its presence in the United States through a new partnership with Dynamism, a well-known distributor of advanced manufacturing technologies. The collaboration was announced during the Additive...
Creality Quietly Gauging Interest in a Desktop Filament Recycler
Creality is testing the waters on a desktop filament recycling system suitable for home use. The Creality Filament Maker M1 and Shredder R1 are in the engineering stage and can...
Will Desktop Firms Push Shoe 3D Printing Forward?
Recently, Bambu Lab announced that it was working with FORMISM by SCRY on releasing shoes. These six designs will be shared and printable through its Makerworld platform. Using the platform,...






























