Medical scientists have made enormous progress in bioprinting. The mere reality that they have advanced far enough to sustain tissue in a lab—as well as implanting it into the human body for successful treatment—is astonishing. The world of 3D printing and bioprinting continues to escalate in progress as researchers break through limitations and barriers. Now, a research team has developed a novel drop-on-demand method for bioprinting cells with a low energy nanosecond laser, and their findings published in ‘Drop-on-demand cell bioprinting via Laser Induced Side Transfer (LIST).’
Growing cells and keeping them alive is difficult, to say the least, requiring the suitable formulation of materials and printing techniques; in fact, most technology today lacks the comprehensive capability for fabrication with bio-inks. Viscosity is a major problem as most bioprinting is performed via nozzles. This type of technology requires lower viscosity if fragile cells are to survive being extruded.
In this study, the researchers introduce the LIST technique, generating microbubbles at the end of a glass microcapillary. This action results in the ejection of a micro-jet filled with cells, in a direction perpendicular to the irradiation axis.

Overview of LIST (a) Schematic side view representation of LIST bioprinting (left) and indicative high-speed imaging of bio-ink ejection (right). The distal end of the capillary and the substrate have been identified with numbers. (b) Detailed schematic of the LIST bioprinting setup.

Sequences of snapshots showing micro-jet evolution and drop formation for different laser energies. The laser pulse was focused at the middle point of the capillary and 500 μm above its distal end.
While refining the process, the researchers noted the best conditions for bioprinting a human umbilical vein endothelial cell (HUVEC), with success at a 30Hz repetition rate and very little loss of viability. The automated LIST setup is comprised of:
- Laser beam delivery system
- Drop-on demand unit
- Micro-jet visualization system
- MATLAB automation and control via a graphical user interface (GUI)
To optimize printing, the researchers experimented with laser energy, varying it from 90 μJ (i.e., ejection threshold) to 130 μJ. In this specific process, the bio-ink jets were considered “less energetic,” and unable to penetrate the substrate. In some cases, the droplets bounced back in terms of decreased energy, with higher energy “splashing” behavior exhibited and different levels of relaxation occurring at varying contact angles. This type of behavior was discovered in LIFT micro-jets too.
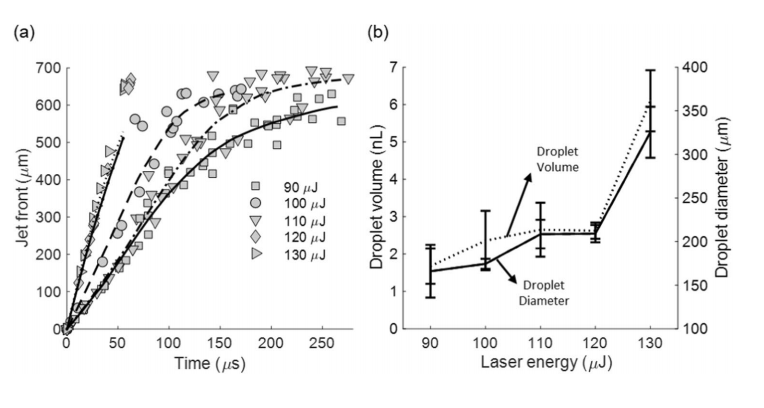
(a) The dependence of the bio-ink jet front position on the laser energy. Gray symbols represent data points and black lines represent the best-ftted curve. N=10 (per energy). (b) The dependence of the droplet volume (dotted line) and droplet diameter (solid line) on the laser energy. N=10 (per energy).
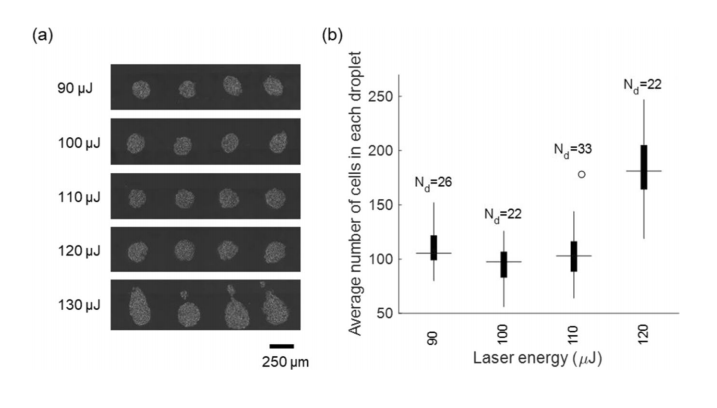
(a) Optical microscopy images of LIST-printed HUVECs for various laser energies. (b) The number of cells per droplet for various laser energies.
A continual 500 μm distance between the laser focus position and the capillary distal end was used in this study, but the researchers suggested LIST performance could be better optimized with the “fine-tuning” of the parameter. While cells were still highly viable after printing, with a droplet size of 165 μm, the researchers recommended further experimentation with thinner microcapillaries or varying of viscosity. LIST is an adaptable technology, but range and issues with viscosity bear more study.
“LIST is technically uncomplicated and can foster 3D printing applications,” concluded the researchers. “It can potentially cover a technological gap in bioprinting technologies, between ink-jet printing and LIFT, as it could not only print bio-inks of high-viscosity but also support 3D printing of constructs with clinically relevant size.”
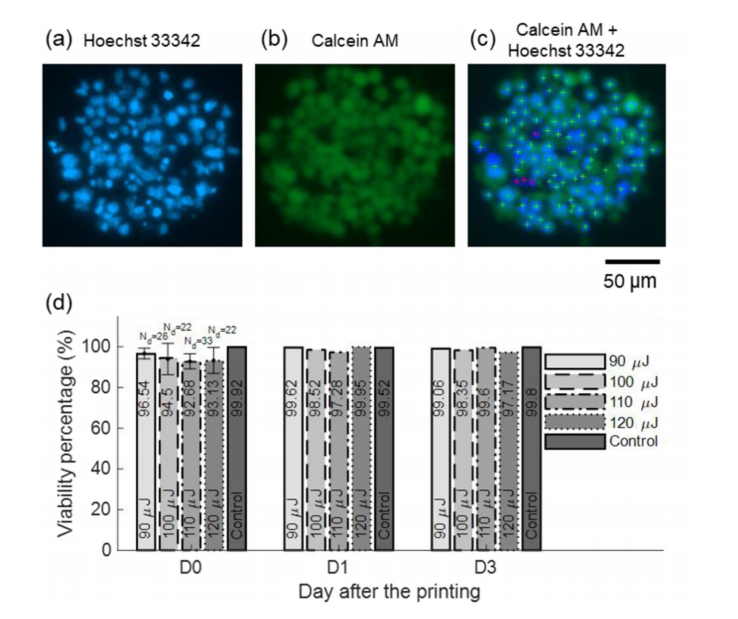
(a,b) Fluorescence microscopy images of LIST printed cells at 90 μJ. (c) Combined imaging channels, including algorithm-generated cell labeling marks. Green crosses indicate live cells and red crosses indicate dead cells. (d) The dependence of the HUVEC cell viability on the laser energy for 0, 1, and 3-days post-printing.
Nd indicates the number of droplets.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
The Magic of AMUG as Reported by a First-Time Attendee
There’s a special kind of magic about AMUG. I’ve heard about it for years, but never experienced it myself until last week. It’s different than what you see at some...
Cobra’s 3D Printed Golf Clubs Reveal What the Technology Can Do for Sports
When 3DPrint.com attended the PGA Show in Orlando this January, one booth stood out for a reason that had nothing to do with marketing hype or big-name tour pros —...
COBRA Golf Releases More 3D Printed Putters
COBRA Golf first used HP‘s BinderJet to make putters in 2020, releasing a line in 2021. The King series putters had 10 models and were made of 316L stainless steel...
The Business of Customized Sports Equipment: How 3D Printing Is Changing Athletic Gear
For years, 3D printing in sports has been linked mostly with prototypes, concept shoes, and one-off experiments for elite athletes. Helmets, cleats, and footwear midsoles often took over the headlines,...





































