3DHEALS 2020 Virtual Medical Summit: 3D-Printed Materials in Healthcare
There were a multitude of sessions and sub-sessions to follow at 3DHEALS 2020 (running from June 5-6), with over 70 speakers and four workshops, covering many topics on complex design, and patient-specific treatment. Here at 3DPrint.com, we have covered many stories on materials, as researchers and manufacturers delve into their uses in other major applications too like automotive, aerospace, construction, and so much more.
At the “Material Science in Healthcare 3D Printing” session, medical applications were discussed in detail by Balaji Prahbu (Director of Strategic Marketing for Medical Device Solutions/Evonik Industries), Steve Kranz (Lab Manager and Senior Scientist at Origin), Sean Dsilva (Medical Marketing Segment Head for 3D Printing Henkel) Adhesive Technologies Division), and Mike Vasquez (Founder and CEO, 3Degrees). Topics covered included the importance of bioresorbable materials, biocompatible materials, those used to create devices and tools during the COVID-19 pandemic, as well as workflow systems designed by materials engineers.
Balaji Prahbu opened the presentation with statistics on osteoporosis, a condition that causes millions of fractures in patients around the world. Currently, Evonik uses a variety of polymers to create materials and implants that assist in treating and healing and reconstructing human bone. While some materials dissolve in the body over time, other types of materials created by Evonik can also be used permanently.
The Germany-based company is also developing other new powders and filaments for patient-specific implants that can be produced in just a few hours and completely suited to the individual needs of patients—evidence of some of the greatest benefits in using 3D printing technology.
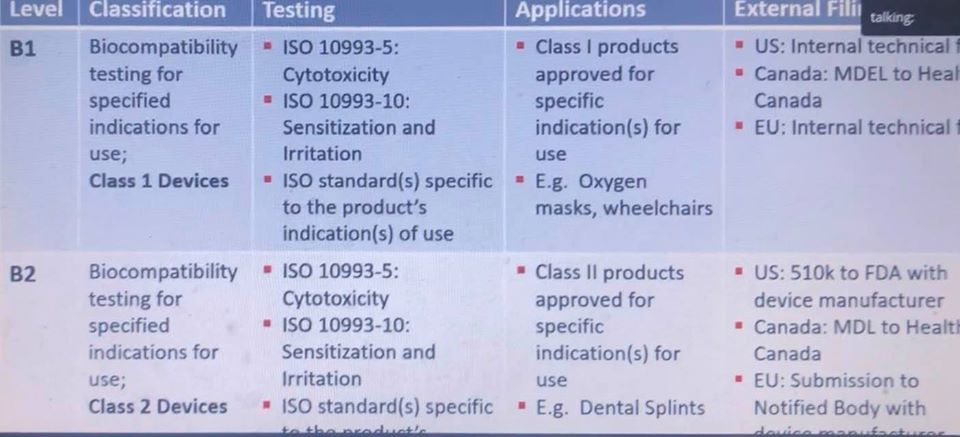
Sean Dsilva offered information regarding Henkel’s biocompatible materials, explaining more too about the keys to developing high-quality, high-performance UV resins—all tied together with effective workflow processes. The $20 billion company specializes in adhesives technology, including that for the medical field. Currently, Henkel offers four different levels of biocompatible materials: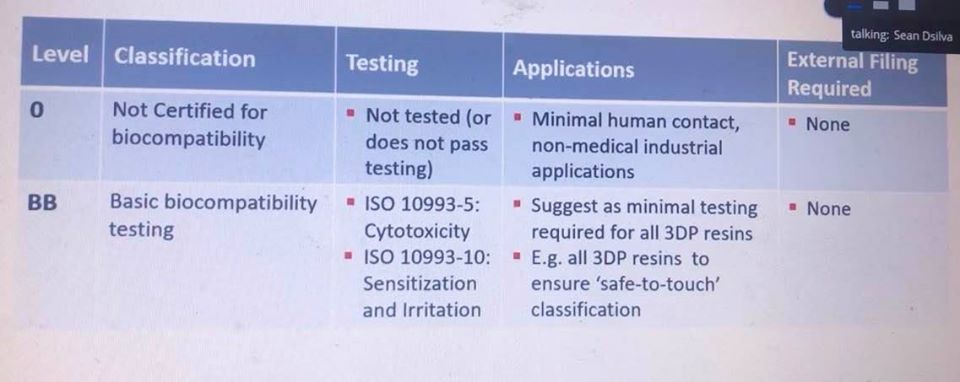
Henkel’s materials can be used for an extremely diverse number of applications, from auditory devices like hearing aids, to prosthetics such as orthotics, bionics, and more. 3D-printed models can also be created, offering a host of benefits like better diagnostics, treatment, and education for patients and their families. Not only that, 3D-printed medical models allow for better training of medical students and allow surgeons to prepare for delicate procedures too. Henkel’s materials are also used to fabricate other industrial components like jigs, fractures, and devices.
Henkel’s materials are also used to fabricate other industrial components like jigs, fractures, and devices.
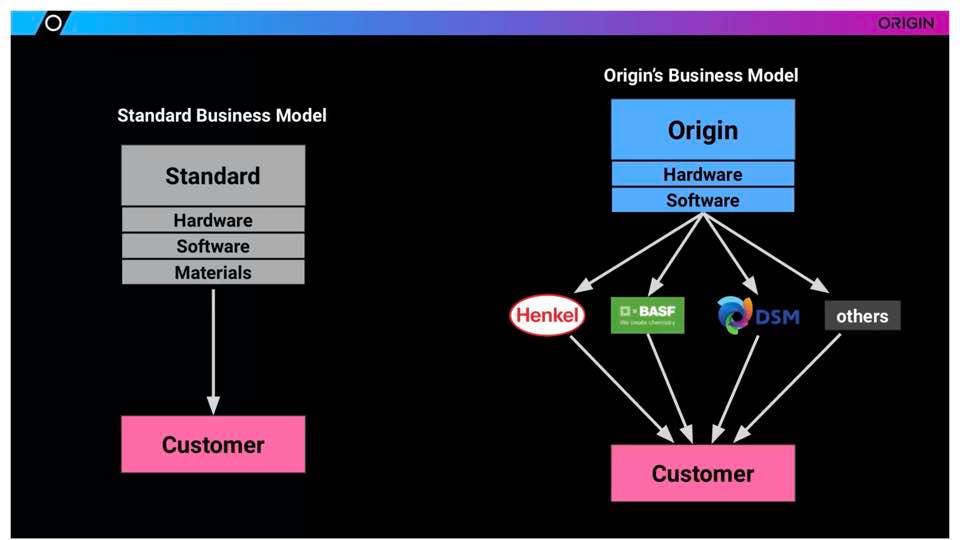
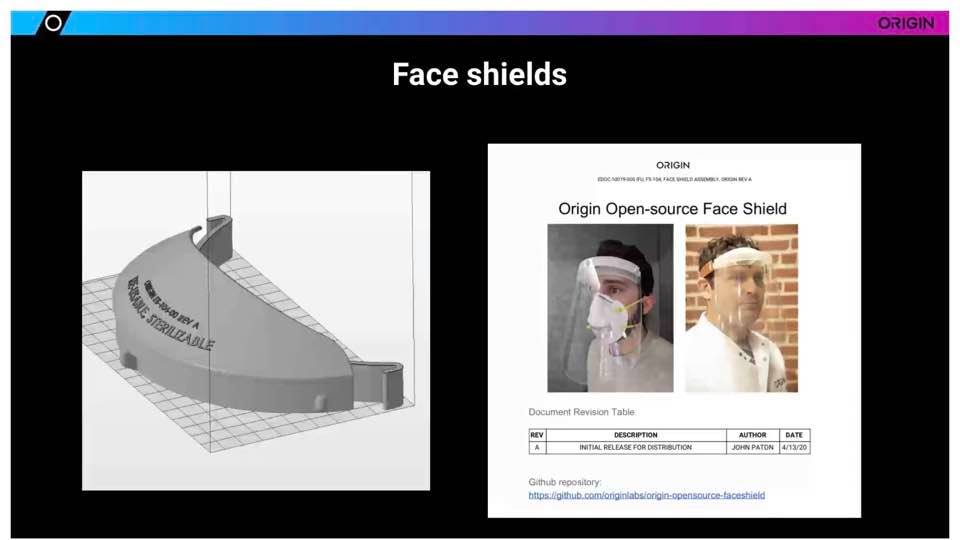
Steve Kranz definitely offered some of the most interesting information regarding materials, and Origin’s recent transformation from software developer and 3D printer manufacturer to a ‘swab factory’ in response to the coronavirus pandemic. The San Francisco-headquartered company began manufacturing a variety of different 3D-printed swabs for testing purposes, as well as originally designing shields and other personal protection equipment, to include adapting snorkels to be transformed into N-95-style face masks.
Kranz began by explaining, however, that while Origin has—previous to the COVID-19 pandemic—been centered around software and hardware endeavors, they do not develop materials; instead, they rely on experts like Henkel, BASF, DSM and others.
“When COVID-19 hit, things changed for us. It was a lot different, so we had to adapt to survive,” explained Kranz. “We transformed ourselves from a platform that allowed other people to do 3D printing to becoming a factory, to printing parts ourselves.”
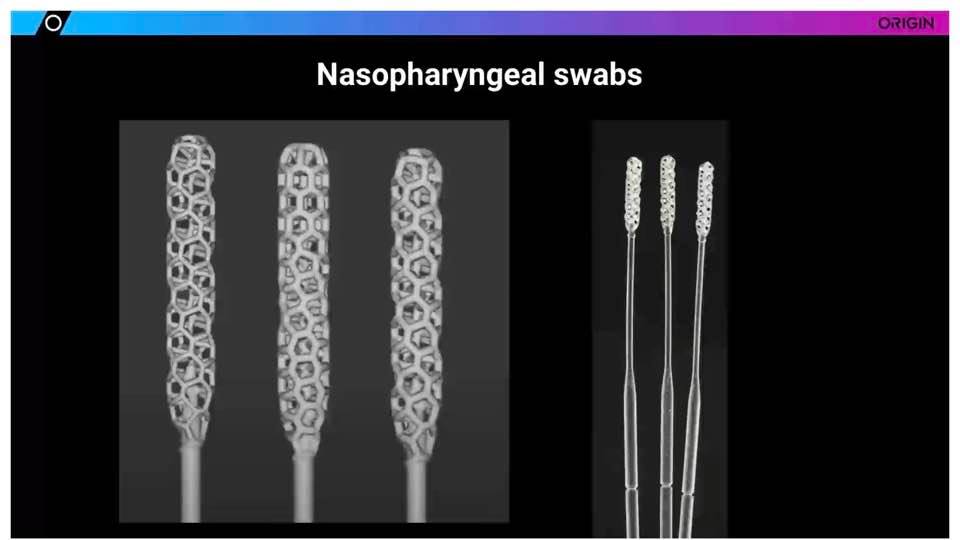
 While the Origin team did initially begin creating other types of COVID-19 devices, Kranz stated that they quickly realized they could offer the most important contribution by 3D printing nasopharyngeal swabs. They began collaborating with nTopology, drafting a flexible, effective design.
While the Origin team did initially begin creating other types of COVID-19 devices, Kranz stated that they quickly realized they could offer the most important contribution by 3D printing nasopharyngeal swabs. They began collaborating with nTopology, drafting a flexible, effective design.
While the two companies were able to work together in creating the actual swab, there were numerous obstacles. Some supplies were difficult to attain, such as isopropyl alcohol, gloves, and paper products. They were also challenged in scaling up with more inventory and other resources, dealing with waste, and hiring additional staff to work a lot of long, and “sometimes crazy” hours.
“That’s been Origin’s journey for the past couple of months. It has been very intense, challenging, strange at some times, but also really rewarding and I feel like we have learned a lot. We’ve kind of put ourselves right in the fire in terms of testing out our own production, our own capabilities, and we have learned a lot that is going to help improve our own printers in the future,” said Kranz.
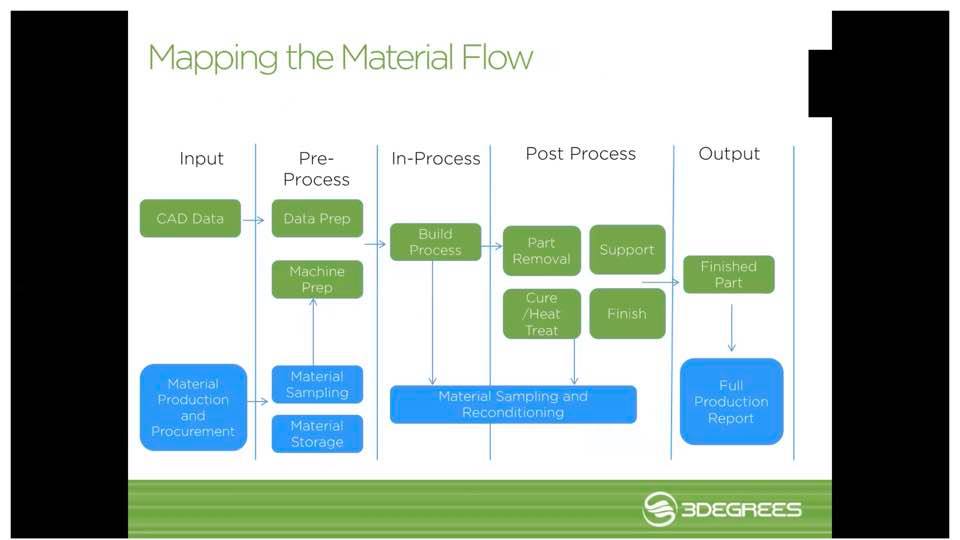
Mike Vasquez opened by explaining that, as a materials engineer, he realizes that additive manufacturing is “fundamentally driven by materials, but it is complicated.” This is due to a lack of accessibility in many cases, an “opaque and often confusing” supply landscape, and limited standards. Material properties may be an issue as well, as they often do not match up with what users are expecting or needing for specific projects.
Because there can be so many challenges—and so much data—involved with creating a 3D-printed part, the 3Degrees team developed the TRACE process for 3D printing workflow management. In creating TRACE, they spoke with over 50 additive manufacturing users, auditors, manufacturers, and standards organizations. The workflow management tool, complete with comprehensive analytics is meant to be customized for different projects.
During the fabrication of 3D-printed medical devices, TRACE can be used to keep track of variables like data inputs, specifications for materials and machine processes, post-processing, and inspections.
Although originally set for ‘the heart of San Francisco’ as a venue, this year’s 3DHEALS Global Healthcare 3D printing conference became a virtual—and inspiring—event. Focusing on the continued impacts to the field of medicine, rather than cancel the annual event due to the COVID-19 restrictions, founder and CEO, Dr. Jenny Chen, committed to an online format, and along with seeing every speaker conform to the changes, she was even able to able 25 percent more in programming.
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: 3DHeals 2020 – from the ‘Material Science in Healthcare 3D Printing’ session)Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
China Becomes Latest Space Power to Demonstrate Metal 3D Printing in Orbit
China has demonstrated metal 3D printing in space as part of its plan to develop manufacturing technologies for future space missions, including Moon construction. The experiment took place aboard the Qingzhou...
AMPulse Asia: APAC 3D Printing Market Roundup
Key Takeaways Coverage window: April 27 to May 10, 2026. Roughly 30 additive manufacturing (AM)-relevant announcements were tracked across eight Asia-Pacific countries. Largest disclosures: Farsoon Technologies (688433.SH) filing a RMB...
3D Printing Financials: Stratasys Bets on Defense and Drones as Printer Sales Slow
Stratasys (Nasdaq: SSYS) started 2026 with lower revenue and a larger loss as customers continued to slow down spending on new 3D printers. Still, the company pointed to stable recurring...
3D Printing Financials: Xometry Surges After Record Quarter and Siemens Deal
Shares of Xometry (Nasdaq: XMTR) surged on Thursday, May 7, after the company reported record first-quarter 2026 results and announced a major partnership with Siemens. The stock climbed as much...