While 3D printed patient-specific implants (PSIs) are helpful in craniofacial surgeries, it’s not always a practical option due to high costs and, as a research team from Switzerland’s University Hospital Basel puts it, “a lack of expertise.” They published a paper, “Accuracy Assessment of Molded, Patient-Specific Polymethylmethacrylate Craniofacial Implants Compared to Their 3D Printed Originals,” about their work to create a “simple and cost-efficient template-based fabrication workflow” that can help surgeons get past these issues and succeed.
“The aim of this study is to assess the accuracy of PSIs made from their original templates,” the researchers explained.
Cranial defects (CFD) and deformities can cause aesthetic, functional, and psychological problems for patients. A cranioplasty is performed to improve a patient’s neurological status, and to restore the function and structure of the missing cranial bone. PMMA is the most popular alloplastic material for cranial reconstructions, as it’s a cost-effective choice, resistant to functional stress, and is lightweight, yet not thermoconductive. But, intraoperative PMMA molding can be difficult in complicated cases that require a PSI.
“Other problems encountered with PMMA include the excessive heat generated by the exothermic reaction that occurs during the molding process, which might harm the surrounding tissues, or allergic reactions to monomers. In addition, the freehand PMMA molding technique is associated with an increased surgical time and often results in unacceptable cosmetic outcomes. Hence, to mitigate the problems associated with freehand implant fabrication, preoperatively or intraoperatively (extracorporeal) fabricated PMMA PSIs are used,” the team wrote.
3D printed templates for custom, pre-fabricated PMMA implants make the process easier, though there haven’t been many studies evaluating how accurate PSIs are, and if the silicone molds are reusable if the patient requires a revision operation. That’s why the team chose to compare molded PMMA PSIs to 3D printed, virtually designed templates.
They chose two cases – a CRD and a temporo-orbital defect (TOD) – and imported DICOM data from the CT scans into Materialise Mimics software. After generating a 3D volumetric image of the skull anatomies, and the overall shape of the PSIs, the files were smoothed out and exported in STL format. The templates were printed out of PLA filament on a MakerBot Replicator+, and post-processing was completed to fix little irregularities that occurred. Finally, an EinScan-SP 3D scanner was used to digitize the 3D printed templates, and the resulting point cloud data was converted and exported in STL format.
Twenty PMMA PSIs were made out of a high viscosity bone cement using the silicone molds – ten each for the CRD and TOD cases. They were digitized with cone-beam computed tomography (CBCT), and the CBCT DICOM data was segmented and extracted with Mimics; data from the digitized PMMA PSIs were then exported in STL format.
The team used the Materialise 3-matic analysis program to compare the accuracy of the 3D printed templates and PMMA PSIs.
“The accuracy of the PMMA PSIs was evaluated by superimposing the STL file data of the related template with the STL file data obtained from the CRD-PSIs (n = 10) or the TOD-PSIs (n = 10) test group. For accurate alignment, the datasets of the CRD- and TOD-PSIs were registered with the corresponding 3D printed templates. All registrations were achieved using the “align” feature. Therefore, five manually placed control points in the n-point registration and a global registration were performed,” they explained.

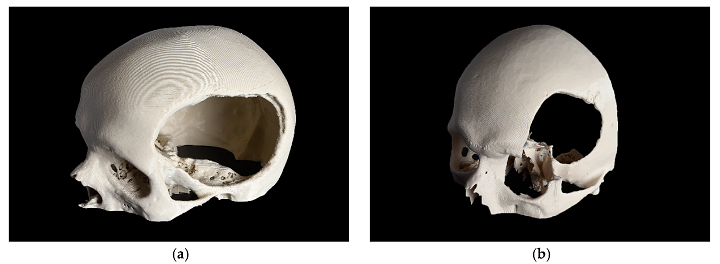
Comparison of 3D printed templates (a, b, beige) with the PSIs according to the n-point registration with five manually placed control points (c, d, purple), and superimposition (e, f). L: cranial template and PSI; R: temporo-orbital template and PSI.
They compared the differences with a maximally tolerated deviation of ± 2 mm, and the measurements were put in a color map.
“Using an identical coordinate system between the datasets, the quantitative values of the deviations were automatically calculated using a 3D analysis program with respect to the root mean square (RMS) values. The RMS values describe the absolute values of the deviations between two datasets. This comparison of the two datasets comprising n-dimensional vector sets provides a measurable value of the similarity after optimal superimposition. The higher the RMS value is, the greater the deviation error between the two datasets will be,” the researchers explained.
The RMS ranged from 1.128 to 0.469 mm, with a median RMS (Quartile 1 to Quartile 3) of 0.574 (0.528 to 0.701) mm for the CRD implants. For the TOD, the RMS was 1.079 to 0.630 mm with a median RMS (Q1 to Q3) of 0.843 (0.635 to 0.943) mm.
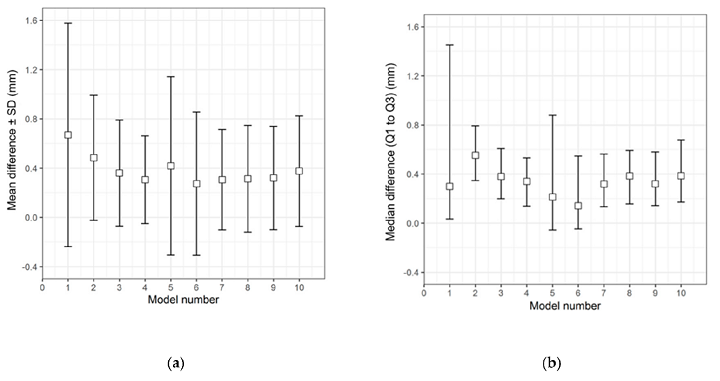
Descriptive data distribution illustrating the difference between the CRD-PSIs and the CRD 3D printed template. (a) Mean difference ± SD; (b) Median difference (Q1 to Q3).
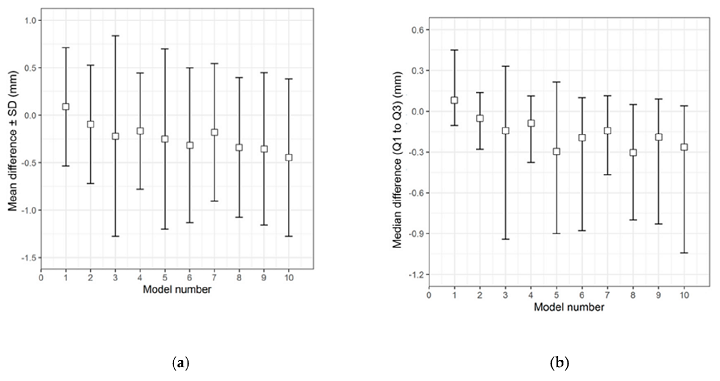
Descriptive data distribution illustrating the difference between the TOD-PSIs and the TOD 3D printed template. (a) Mean difference ± SD; (b) Median difference (Q1 to Q3).
You can see in the box plot graph below the quantitative data distribution results of the RMS values for the ten PSIs in the two test groups.
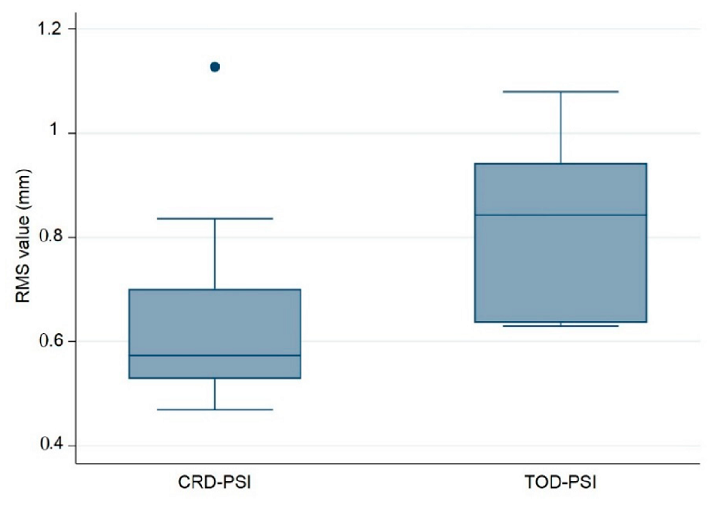
Box plot illustrating accuracy comparison with respect to RMS values between the PMMA CRD-PSI and TOD-PSI test groups (● describes the statistical outlier, CRD-PSI 01).
The deviation analysis for the CRD-PSIs and TOD-PSIs is shown in the heat map below. The blue areas represent negative deviations, and the red show positive. There was a slight positive deviation on the outer surface of the temporal region of the CRD-PSIs, and a slight negative on the inner surface “at the antero- and posterolateral margins.” For the TOD-PSIs, the outer surface of the infra-temporal region had a strong positive deviation, with a negative on the inner surface at the posterolateral margin.
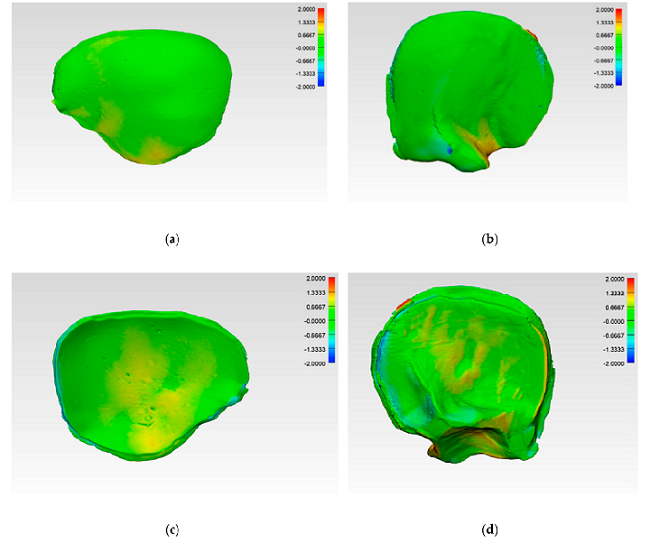
Color-coded deviation maps within each test group after applying best-fit method and generating a 3D comparison to evaluate accuracy. CRD-PSI: (a) squamous (outer) surface; (c) cerebral (inner) surface. TOD-PSI: (b) squamous surface; (d) cerebral surface.
“The present study demonstrates that the described manufacturing process of molded patient-specific PMMA implants based on 3D printed templates is highly precise, with a less than 1 mm deviation evaluated in two different defect patterns. This reflects the results commonly reported in the literature, where the overall inaccuracies of pure 3D printed anatomical models are also less than 1 mm,” the researchers wrote. “Thus, the PSIs are largely consistent with the 3D printed templates in terms of accuracy.”
As patient-specific treatments become more popular in diagnostic treatments and procedures, the demand continues to rise, especially when it comes to cranioplasty. You can easily see the difference between reconstructing a skull defect with a 3D printed PSI, and doing so with an implant manufactured manually. Even after “successive usage of silicone molds,” the study found that the dimensional accuracy of the PMMA PSIs was at a “clinically acceptable accuracy level.”
“The RMS values illustrate that, even after ten impressions (n = 10), the manufacturing method produces no clinically relevant deviations,” the researchers concluded. “Overall, the results suggest that the manufacturing process described in this study is an exact and reproducible technique. The median RMS values for each of the two test groups did not exceed 1 mm, which is an acceptable accuracy for clinical routine in craniofacial reconstruction.”
This workflow is an accurate, repeatable way to use PSIs in anatomical reconstructions, as it reduces time in the OR, makes common materials more available, and the silicone mold can be reused.
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
The Market and Industry Potential of Multi-Material 3D and 4D Printing in Additive Electronics
Additive manufacturing leverages computer-based software to create components for products by depositing either dielectric or conductive materials, layer by layer, into different geometric shapes. Since its birth in the 1980s,...
3DPOD 262: Bio-inspired Design for AM with Dhruv Bhate, Arizona State University
Dhruv Bhate is an associate professor at Arizona State University. There, he looks at structures, materials, and design. Previously, he worked at PADT as well as in the semiconductor and...
3DPOD 261: Tooling and Cooling for AM with Jason Murphy, NXC MFG
Jason Murphy´s NXC MFG (Next Chapter Manufacturing) is not a generalist service; instead, the company specializes in making tooling. Using LPBF and binder jet, the company produces some of the...
3DPOD 260: John Hart on VulcanForms, MIT, Desktop Metal and More
John Hart is a Professor at MIT; he´s also the director of the Laboratory for Manufacturing and Productivity as well as the director of the Center for Advanced Production Technologies....