University of Hong Kong: Sterilization & Infection Rates in 3D Printed Models & Guides Used Intra-operatively
Hong Kong researchers explore the use of 3D printed medical models but advance in a more unique direction with concerns over infection. Detailing their findings in the recently published ‘A review of the manufacturing process and infection rate of 3D-printed models and guides sterilized by hydrogen peroxide plasma and utilized intra-operatively,’ the authors discuss the use of innovative bespoke devices for surgical planning.
As 3D printing has made impacts within the medical field, medical professionals, patients, and their families benefit due to better avenues for diagnosis, treatment, and education for everyone involved. But there are even more specific uses for 3D printed medical models today in promoting patient-specific treatment with improved pre-operative planning procedures and even intra-operative processes.
“In the specialty of orthopedics, 3D models can allow for visualization of bony anatomy and implant contouring, whilst guides can be created to direct osteotomies as well as screw entry sites,” explained the researchers.
Much attention is paid to the strides being made due to 3D printing, but outside issues such as sterility are critical to the health of patients also. Typical methods include ethylene oxide (EtO) gas and hydrogen peroxide plasma, which is a result of excitation beyond the gaseous phase—with free-radical formation allowing for sterility.
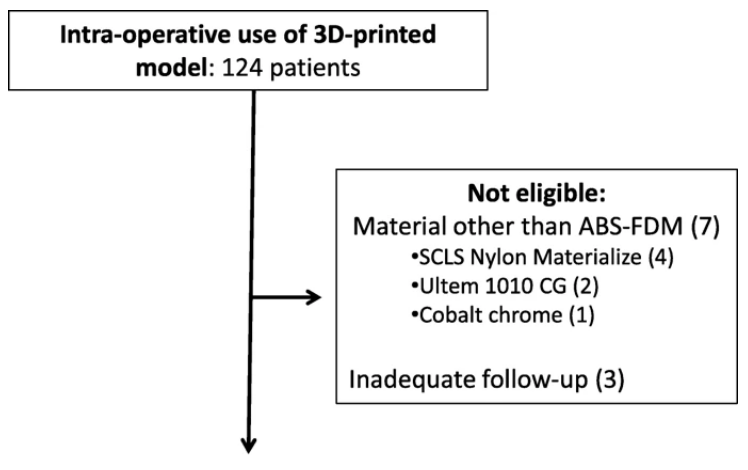
Patient eligibility and exclusion. More than 300 models were rendered by computer software from 2015 – 2019. The numbers of models proceeding to manufacturing, sterilization and intraoperative use amounted to 124. A further ten patients were excluded from analysis due to use of materials other than ABS (7 patients) as well as failure to reach 3-months of follow-up subsequent to surgery (3 patients), leaving a total of 114 patients eligible for analysis.
To date, the University of Hong Kong has produced over 300 3D models and guides. Beginning in 2015, their orthopedic academic unit began 3D printing models and guides on-site; and while they were at first fabricating models exclusively for orthopedics, over time they also began 3D printing for ‘other surgical contexts’ too. The authors confirm that out of the 300 models produced, 114 have been used for intra-operative purposes. Their review goes on to cite details regarding cases using models and guides, identifying those in which infections occurred, and highlighting risk factors.
3D printed models and guides were designed using Meshmixer and printed on a Fortus 450mc 3D printer with ABS-M30i.
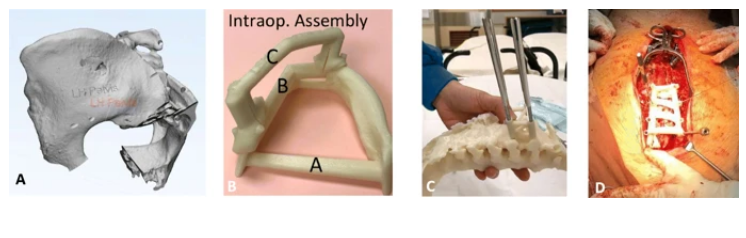
Aspects of model/guide manufacturing unique to intra-operative usage. a 3D-rendering of pelvis model with initials engraved upon the left ilium to allow for correct patient identification. b Photos taken by instrument nurse demonstrating proper grouping and assembly of a surgical guide for pedicle screw placement so corresponding components may be packaged and sterilized together. The assembled guide was contoured to fit upon bony surface landmarks of the posterior spinal vertebra, as demonstrated during testing upon a 3D-model of the same patient c and during the definitive surgery (d)
3D prints were sterilized with hydrogen peroxide plasma, and low temperatures prevented deformation in material.
“A surgical time-out procedure ensured that the printout was used for the correct patient, anatomical region and procedure, in accordance to initials upon the surface,” explained the authors. “Post-operatively, printouts were similarly subject to low temperature disinfection then returned to the surgeon in charge.”
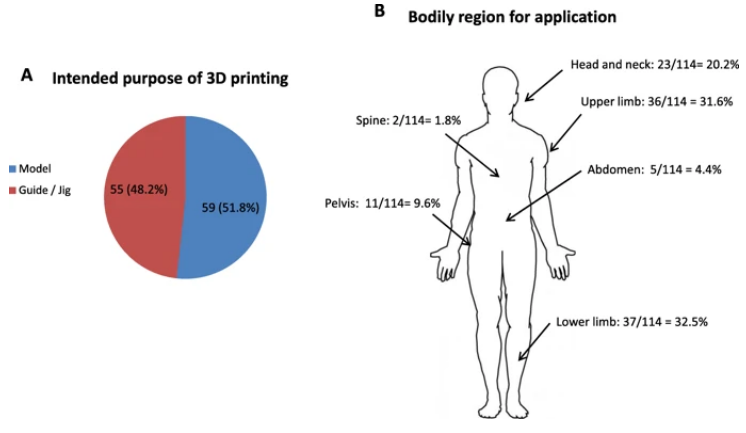
Application of 3D printouts. a Intended purpose of 3D printout showing 59/114 (51.8%) of printouts being utilized as anatomical models and 55/114 (48.2%) as guides/jigs intra-operatively. b The 124 cases utilized intraoperatively spanned different regions of the body as well as surgical specialties. The numbers relevant to each region and their percentages in relation to the whole patient cohort is shown
The researchers examined 3D prints from 124 patients with models used intra-operatively during surgical planning and management.
“Seven cases were excluded as printouts were not constructed from ABS, of which four cases utilized nylon, two case utilized polyetherimide (Ultem1010 CG), and one case utilized cobalt chrome,” explained the authors.
“Three cases were excluded because of inadequate follow-up. A total of 114 models remained for subsequent analysis. Fifty nine out of 114 (51.8%) were anatomical models utilized on-table for planning and / or implant contouring. The remaining 55/114 (48.2%) were utilized as guides or jigs specific to patient anatomy to facilitate corrective osteotomies, screw insertion or pin placement.”
Ultimately, 10.9 percent of the guides or jigs developed infections, while 3.3 percent of the models developed infections at the surgical sites.
“All six cases of guides/jigs with infection were utilized to facilitate osteotomies. Both models with infection were utilized for implant contouring, one during fixation of a pilon fracture and the other for an orbital floor blowout fracture,” stated the researchers.
Pointing out that while the infection rate of 7 percent was comparable to previous literature published regarding traditional techniques, the authors realize the importance of users to ‘be aware of potential caveats,’ despite the overall safety of the application. There are also intrinsic challenges in the fabrication of patient-specific devices and ensuring the safety of tissue biocompatibility.

Cross-section of pelvis 3D model demonstrating irregular luminal spaces. a Arrows indicate surface openings upon the posterior ilium on a 3D model of the pelvis. The dotted line and arrowhead demonstrates the level of transverse sectioning subsequently performed. Cross-sectional appearance following software rendering b and physical sectioning c of the same pelvis model demonstrating irregular trabecular spaces contained within

Intraoperative use of osteotomy guide. a Software rendered image of guide intended for corrective osteotomy and shown upon the tibial shaft (b) with the osteotomy site marked in teal. c Intra-operative photo with the guide secured and oscillating saw engaged in preparation for osteotomy and (d) upon completion. e Intra-operative x-ray demonstrating reduction and fixation of the tibial shaft following corrective osteotomy. f Similar guide retrieved post-op demonstrating damage to ABS over the osteotomy slit with the potential to release debris
“It is worth noting that prior studies detailing infection-related outcomes of 3D printouts have not explicitly utilized them intra-operatively, and this is one of the first studies to have done as such. Our overall impression was that our process of sterilization and on-table usage is safe, and that surgical complexity and tissue manipulation as reflected by increased operating time were the main culprits for infection,” concluded the authors.
“In detailing the design, printing, and sterilization of 3D printouts as well as infection-related outcomes amongst this sizable cohort, we demonstrate that our production process is safe for continuation and may be adopted elsewhere.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[‘A review of the manufacturing process and infection rate of 3D-printed models and guides sterilized by hydrogen peroxide plasma and utilized intra-operatively’]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Scaling 3D Printing Takes More Than You Think; HP’s Webinar Looks at Making It Work Long Term
3D printing is no longer new. In fact, most manufacturers already know what it is, where it fits, and what it can do. But knowing the technology and actually scaling...
How AtomForm’s 12-Nozzle System Cuts Multi-Color FDM Transition Waste by Up to 90%
Pull up the print stats on multi-color FDM jobs. The number that stings isn’t time; it’s material efficiency. On six-color models, single-nozzle systems consume significant filament during transition flushing, expelling...
RAPID 2026: Democratizing Metal Laser Powder Bed Fusion 3D Printing with Mastrex
Aside from a quick mention in an episode of our Printing Money podcast, I didn’t know much about Mastrex. But when I heard that the company had developed a $39,000...
3D Printing News Briefs, April 25, 2026: Competition Winners, AI Platform, X2D Printer, & More
In this weekend’s 3D Printing News Briefs, AMUG announced the winners of its Technical Competition, and Authentise launched AI platform Whisper at RAPID. Bambu Lab wasn’t at RAPID, but launched...













































