3D Printing for COVID-19, Part Seven: New 3D-Printed Parts and Partners
Corporate, government and individual efforts to use additive manufacturing (AM) to address the medical supply shortages resulting from the COVID-19 outbreak are continuing. We continue to stress that the industry can not yet verify the safety of many of the items being 3D-printed. Unless these parties are working with medical professionals and both sides understand the potential inherent risks involved in 3D printing medical devices to be used in contact with a highly contagious disease, we will suggest that they may potentially do more harm than good.
One example of such a likely medically sound project comes from 3D printing startup Formlabs, which is 3D printing 150,000 COVID-19 test swabs daily using its 250+ in-house 3D printers at its Ohio printing facility. Over one weekend, the company worked with Northwell Health, New York’s largest hospital system, and USF Health to create a nasal swab prototype and test it in a lab.
Within two days, USF Health and Northwell were using Formlabs’ 3D printers and printing autoclavable resins (i.e., sanitizable in an autoclave) to create prototypes that not only received approval for use by hospitals, but also FDA Class I Exempt status and are in compliance with new CDC guidelines. Northwell Health and USF Health. So far, the swabs are being used at Northwell hospitals and Tampa General Hospital to meet the demand for COVID-19 testing. The design will also be shared with other institutions using the Formlabs COVID-19 Response page.
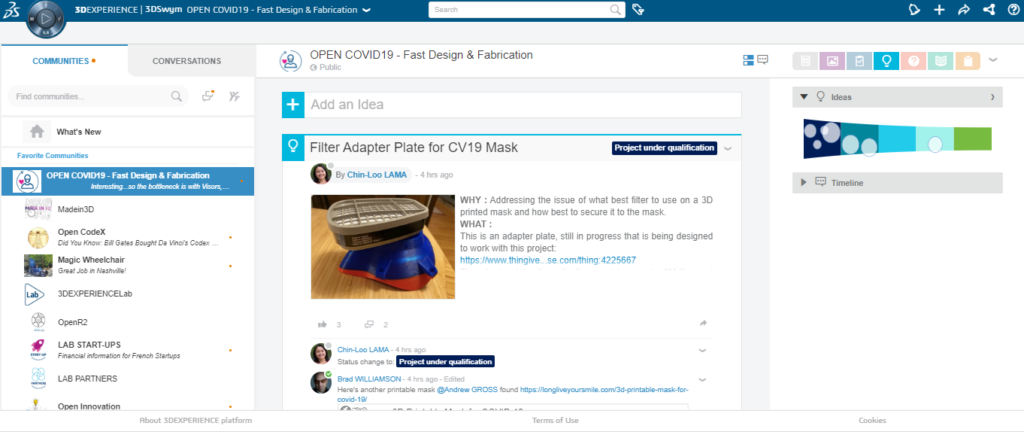
Other recent initiatives include Dassault Systèmes’ Open COVID-19 Community, which is meant to act as a forum for sharing design and production solutions for producing necessary medical parts and devices. Users have access to blog posting, a wiki and Q&A, 3D model storing, viewing and sharing, millions of standard parts and manufacturing resources, as well as mapping of FabLabs, MarketPlace resources and hospitals.
As an online community, much of the desired outcomes have yet to be executed as envisioned. For instance, experts within the community are to be the ones qualifying community solutions and SIMULIA experts are imagined providing simulations of designs alongside scientists to determine their validity. Through SOLIDWORKS xDesign, Dassault sponsors are hosting design challenges, including projects for OpenSourceVentilator and Montreal Code Life Challenges. The CAD giant is also allowing academic research groups dedicated to COVID-19 vaccines to use its BIOVIA Discovery Studio for six months without charge.
Dassault is not alone in its aim to act as a hub for coronavirus-related manufacturing resources. As previously covered 3D Hubs has its own pandemic-related fund tied to manufacturing services. 3D printing material seller MatterHackers is also positioning itself as a central resource to connect those who need parts manufactured to those who can 3D print them.
Additional 3D printing related efforts underway feature work from Roboze in Italy, which claims to be 3D printing respirator valves and parts for C-PAP masks, alongside Isinnova and Dallara Automobili. Isinnova, which developed the initial 3D printable ventilator valves in Italy, ultimately relied on another local firm, Lonati SpA, to mass produce the valves. Lonati happens to be a 3D Systems customer, as well, and is now using one of the manufacturer’s SLS machines to batch manufacture over 100 such valves.
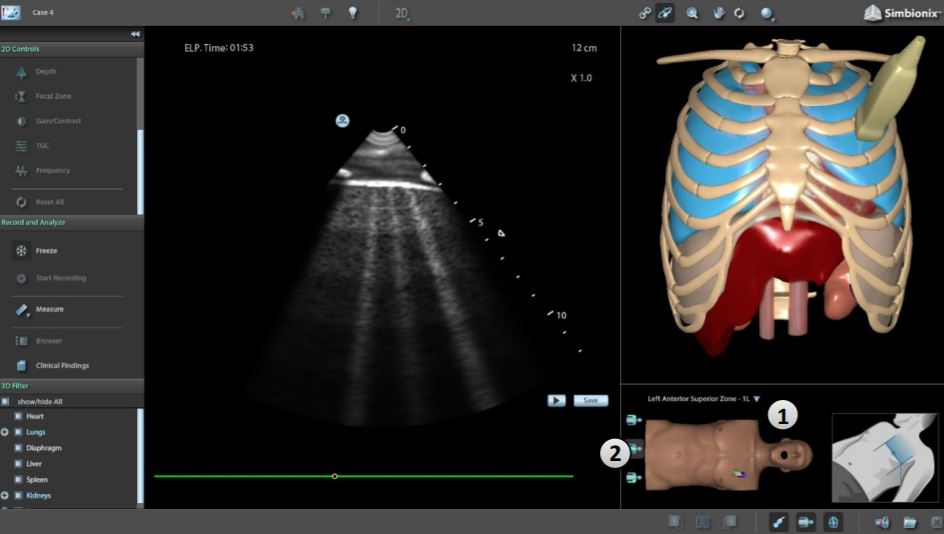
3D Systems is now prototyping the same valves in South Carolina and Tennessee with the potential to mass manufacture 12,600 clear plastic units and 1,400 nylon parts per week. The company has also developed a new COVID-19 module for its Simbionix U/S Mentor simulator to educate medical professionals in lung ultrasound skills necessary for triage and coronavirus monitoring. Along with this new module, the company is offering free online learning tools associated with Simbionix.
As with many of the AM stories related to COVID-19 that are being publicized in recent weeks, it is important to take many of these initiatives with some healthy skepticism. The line between humanitarian aid and marketing campaigns are so blurred that the pandemic is being used as an opportunity to provide free software trials, sell simulation hardware and software, and show off 3D printing hardware that much of it feels quite exploitative and craven.
Given the fact that Formlabs has material that can be effectively sanitized in an autoclave, their efforts may truly be helpful—if medical partners truly believe that 3D-printed test swabs work as well or better than traditionally made counterparts.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
China Becomes Latest Space Power to Demonstrate Metal 3D Printing in Orbit
China has demonstrated metal 3D printing in space as part of its plan to develop manufacturing technologies for future space missions, including Moon construction. The experiment took place aboard the Qingzhou...
AMPulse Asia: APAC 3D Printing Market Roundup
Key Takeaways Coverage window: April 27 to May 10, 2026. Roughly 30 additive manufacturing (AM)-relevant announcements were tracked across eight Asia-Pacific countries. Largest disclosures: Farsoon Technologies (688433.SH) filing a RMB...
3D Printing Financials: Stratasys Bets on Defense and Drones as Printer Sales Slow
Stratasys (Nasdaq: SSYS) started 2026 with lower revenue and a larger loss as customers continued to slow down spending on new 3D printers. Still, the company pointed to stable recurring...
3D Printing Financials: Xometry Surges After Record Quarter and Siemens Deal
Shares of Xometry (Nasdaq: XMTR) surged on Thursday, May 7, after the company reported record first-quarter 2026 results and announced a major partnership with Siemens. The stock climbed as much...