As small 3D printing businesses and individual users jump at a chance to support efforts to manufacture critically needed medical supplies, larger corporations also see opportunities to lend aid. Among the names involved are HP, Airbus, and Ford.
Emergency Bagger and Splitter
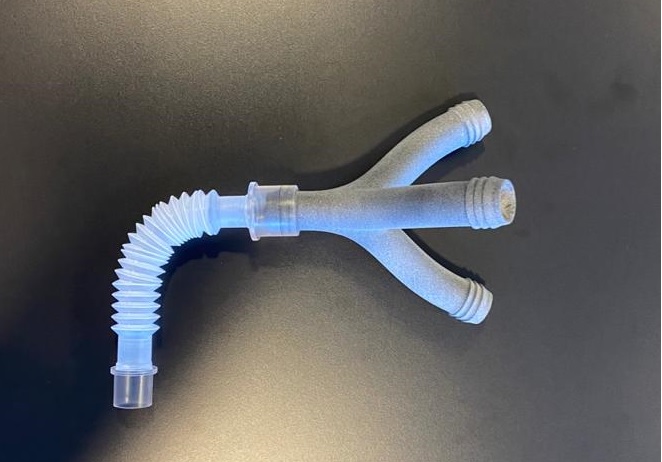
A consortium of Spanish companies has developed an emergency bag valve mask featuring 3D-printed components. Called the Leitat 1, the device is meant to be both simple and robust. Unlike a ventilator, the Leitat 1 is designed more for immediate emergency usage and not long-term use. The design was created by Leitat Technological Center, a private technical institute, and validated by the director of innovation at Hospital Parc Taulí. Airbus, Navantia and HP aim to aid in the production of the device, with manufacturing of 50 to 100 units sought to be established by next week.
Leitat has since gone on to develop a 3D printable splitter (similar to the part discussed in our previous post) that allows multiple patients to use a single respirator. Yesterday, the production started of a 3D printed splitter that will allow multiple patients affected by the Coronavirus syndrome COIVD-19 to access a single respirator. The company is relying on HP’s multi jet fusion technology with the idea that it might be possible to 3D print 17 parts in a single print job. A number of companies have already begun producing the parts.
Hospital de la Vall d´Hebron and the Consorci Sanitari de Terrassa in Spain are in the process of validating the splitter and ensuring an effective protocol is in place for their implementation. A first round of 200 devices is in the process of being delivered and 1,000 pieces are expected to be produced by next week.
Protective Gear 3D Printed by Ford
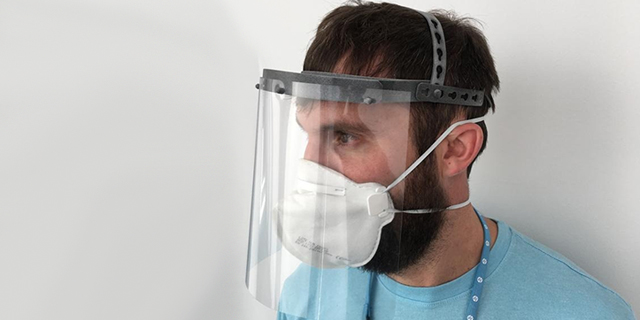
Ford Motor Company has partnered with 3M and GE Healthcare in an attempt to scale up its production capabilities for medical equipment and supplies using its in-house AM systems at its Advanced Manufacturing Center in Michigan. The company hopes to manufacture and assemble 100,000 face shields per week, which can complement N95 masks for further protection of medical staff. The first 1,000 shields are being tested this week at Detroit Mercy, Henry Ford Health Systems and Detroit Medical Center Sinai-Grace hospitals.
With 3M, Ford is adapting its existing off-the-shelf components, such as fans from its F-150 vehicle, to work with 3M HEPA filters in order to improve airflow and filter airborne droplets for 3M’s powered air-purifying respirators (PAPRs). The car company is also looking at how to manufacture these PAPR designs at its facilities in Michigan. GE Healthcare is working with Ford to produce simplified versions of GE’s ventilators, potentially manufacturing them at Ford’s own facilities and at a GE site.
Protective Gear 3D Printed by HP
HP has claimed that it is 3D printing critical parts for medical staff, including respirator parts, face masks and shields, mask adjusters, nasal swabs and hands-free door openers. The initial applications are in the validation stages and are now being finalized for mass production; however, HP has already produced over 1,000 parts to hospitals from its research centers in Spain, Oregon, California and Washington. The company is currently discussing with governments, health agencies and industry to determine the possibilities of working together to get these items to medical staff.
Devices being finalized for manufacturing include a hands-free door opener that has been designed by HP partner Materialise, which makes it possible for people to open doors without touching possibly contaminated handles; a mask adjuster that allows staff to improve the comfort of their masks; and brackets for attaching plastic face shields.
Other items that are in the testing face include the Leitat bag valve mask described above and FFP3 face masks. The files will be made available for download and production. Requests and suggestions can also be made directly to HP.
Specialty Test Swabs and Masks from Carbon and Alphabet (Google)
According to the company, Carbon has redirected all of its business operations in California to addressing the COVID-19 outbreak. Working with Verily, the Alphabet-owned company developing the COVID-19 online screening software Project Baseline, the startup has developed a face shield that is already being tested at Stanford Hospital and with Kaiser Permanente.
Carbon also claims to be discussing the use of its technology to 3D print test swabs with adidas, Mayo Clinic and ASU. The test swabs would feature a lattice structure to increase their testing capacity. This project is being performed alongside Stanford Medical Center, Harvard Medical School/Beth Israel Deaconess Medical Center, Desktop Metal’s Ric Fulop, Chan Zuckerberg BioHub and University of North Carolina at Chapel Hill.
In response to interruptions to business operations during the outbreak, Carbon is also allowing for a payment extension for its customers with fewer than 250 employees.
As helpful as any of these parties may wish to be, it is difficult to determine the line between marketing exploit and genuinely altruistic endeavor. Though it feels like there’s no need for elaboration on this point, it’s necessary to underscore the fact that we regularly witness large corporations take advantage of genuine public concerns and health crises to portray those businesses as good Samaritans. The sheer number of businesses “stepping up” might reflect that there is some marketing at work here, in spite of any good attentions. We may even be hitting a point at which nearly all 3D printing companies feel pressured or obligated to participate in COVID-19 efforts based on the general zeitgeist of the industry.
Regardless of the intent behind such efforts, it is worth noting that larger institutions have the ability and resources to potentially contribute and actually meet the regulatory requirements of the medical system. Carbon, HP, and Materialise, for instance, have already been working with medical companies to produce sterile parts. The general clout that big businesses have will also get their efforts placed in front of the proper government agencies and regulatory bodies to ensure the safety of their printed devices, whereas small firms may not have the same advantages.
As a doctor pointed out in our last post, it may be necessary for anyone who can contribute to the pandemic to do so, regardless of a company’s motivation. With that in mind, as long as 3D printed parts are safe and effectively deployed, their efforts may be desirable in this time of need.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Portsmouth Naval Shipyard Installs 3D Printed Part on In-Service Submarine in Key Milestone for US Defense Sector
I’ve been tracking the US Navy’s additive manufacturing (AM) buildup as it relates to submarines for a while now, and even amidst the AM efforts that all the branches have embarked...
University of Glasgow Researchers Develop 3D Printed Compostable Electronics
Recycling is one of those things that people support, but, for the most part, don’t actually do. If it sounds like I’m being judgmental, let me be the first to...
Continuum Powders Releases Two New Copper-Nickel 3D Printing Powders
The significance of critical minerals to global geopolitics is showing no sign of slowdown as we approach the new year; if anything, it looks like 2025 was just the very...
6K Additive Goes Public on ASX
After years of negative investor sentiment in public markets, the additive manufacturing (AM) industry has seen a modest resurgence in Initial Public Offering (IPO) interest in 2025. Back in August,...