India: Researchers Continue to Review AM Processes in Bioprinting
In the recently published ‘A Review on Additive Manufacturing for Bio-Implants,’ authors Tajeshkumar R. Jadhav, Dr. Nitin K. Kamble, and Pradnesh R. Padave explore one of the most fascinating topics in 3D printing today as researchers make huge strides in developing medical devices with the use of innovative materials.
While some scientists are focused on the complex task of tissue engineering human organs, many others have made huge progress in the area of patient-specific treatment. This includes the development of devices like titanium bone implants, while others continue to develop new 3D printed prosthetics, dental and orthodontic implants, and more. In this review, researchers from Patil College of Engineering in Pune, India discuss scientific advances in the biomedical realm with digital fabrication.
As millions of patients are operated on daily, medical scientists, doctors, and surgeons are always exploring new ways to treat patients better. Tissue engineering and 3D printing are quickly moving to the forefront as one of the most innovative alternatives for tissue, bone, and organ regeneration, usually through the fabrication of scaffolding and other biocompatible structures used to promote growth. Additive manufacturing via extrusion is being used often with a wide range of materials to include polymers, inks, hydrogels, pastes, and more.
“While applications of bioprinting of oral tissues are still in early stages, this strategy has displayed interesting results in various preclinical studies and seems encouraging, progressing beyond templates and models,” state the researchers. “However, for successful clinical translation it is important to develop a road map, which includes studies to receive the required FDA approval and CE marking at an early stage in the process.
Steps are being taken to create more safety and standardization guidelines, while also finding a balance with new developments and methods for making patient-specific treatment plans and customizations previously unheard of in medicine.
While the technology of 3D printing and additive manufacturing has already led to countless, groundbreaking inventions—some of which may substantially improve or even save lives—there are still many challenges to overcome; for instance, equipment is often out of reach financially, materials may be difficult to come by, and there are other complexities and inconveniences like processing and finishing issues.
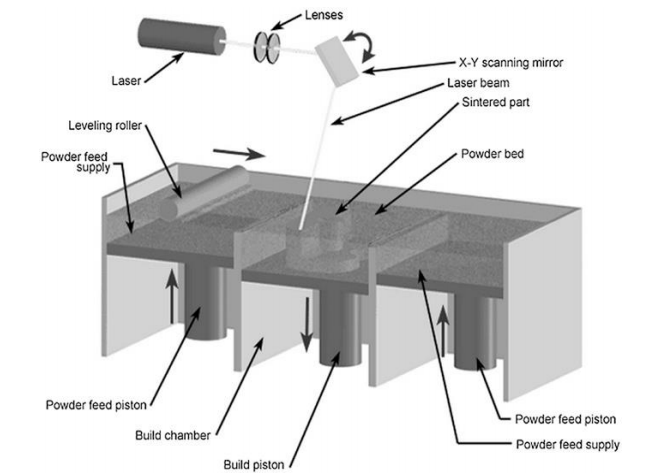
Scientists use a variety of different methods today for the fabrication of bio-implants, to include:
- Inkjet printing
- 3D printing
- Stereolithography
- Selective laser melting
- Bioprinting
“Currently, there are three main ways that cells can be printed on the implants directly, (i) Inkjet, (ii) Extrusion and (iii) Laser Assisted Based (LAB). Indirect printing technologies do not print biomaterials. Such methods are used mainly for the construction of scaffolds which are then used for the seeding of cells, drug delivery systems, potential biochips or biosensors,” state the researchers.
Users have many options to choose from today but must be aware of the pros and cons of each method of digital fabrication, as well as that of different software, hardware, and materials.
Researchers are already working on both the macro- and micro-scale, however, learning more about how to manipulate larger materials as well as nanosized particles during in vitro studies.
“Direct fabrication of implants and prosthetics is however limited to the direct metal AM technologies that can produce parts using FDA (The US Food and Drug Administration) certified materials plus the small number of technologies that are capable of non-load bearing polymer scaffolds,” concluded the researchers.
“As more inter-disciplinary researchers are recruited into the field together with the advancement in biomaterials, it is likely that AM machines and techniques will be further improved over the years.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
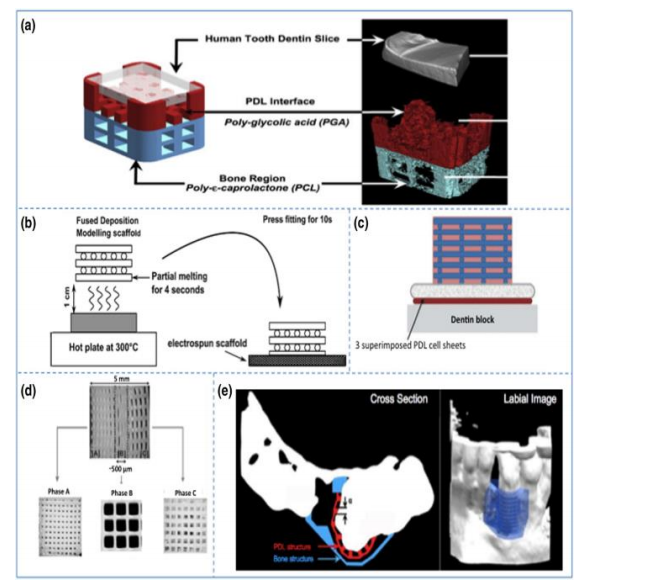
– Additively manufactured scaffolds for periodontal regeneration. (a)
Biphasic scaffold facilitating fiber orientation (b) Biphasic scaffold in
combination with cell sheet technology (c) Enhanced biphasic scaffold (d)
Triphasic scaffold (e) First additively bio manufactured scaffold for
periodontal regeneration applied in human
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Amnovis Expands to the US
Belgian firm Amnovis is a scalable partner for orthopedic innovations. From design to production and regulatory, you can rely on them to take your innovation to market. Coupled with deep...
Scaling 3D Printing Takes More Than You Think; HP’s Webinar Looks at Making It Work Long Term
3D printing is no longer new. In fact, most manufacturers already know what it is, where it fits, and what it can do. But knowing the technology and actually scaling...
Astrobotic Tests Rocket Engine Made with Elementum 3D Materials
Astrobotic has completed a series of hot-fire tests for its Chakram rotating detonation rocket engine, with additive manufacturing (AM) playing an important role in how the engine was built. The...
The Additive Chicken Coop, Part I: Million Dollar Petri Dishes
After decades of tinkering with our individual technologies, billions were poured into speculative claims and optimism. Now the attention is gone, and sometimes it can seem like we’re surrounded by...