3D Printing Microstructures for New Drug Delivery Systems with SPHRINT
In the recently published, ‘SPHRINT – Printing Drug Delivery Microspheres from Polymeric Melts,’ authors Tal Shpigel, Almog Uziel, and Dan Y. Lewitus explore better ways to offer sustained release pharmaceuticals via 3D printed structures.
Currently, numerous materials are used to create microparticles for sustained drug release, with numerous choices in biodegradable materials, such as include poly(lactic acid) (PLA), polyglycolic acid (PGA), their copolymer poly(lactic-co-glycolic) acid (PLGA) and polycaprolactone.
“Numerous microparticle-based depots products are FDA approved, amongst them are: Zmax® (Azithromycin), Decapeptyl®/Trelstar® (Triptorelin), Vivitrol® (Naltrexone), Arestin® (Minocycline), Risperdal® Consta® (Risperidone), Sandostatin® LAR Depot (Octreotide), Nutropin Depot® (Somatropin), Lupron Depot® (Leuprolide), DepoCyt® (Cytarabine), DepoDur® (Morphine), Bydureon® (Exenatide) Somatuline LA (Lanreotide) [1] and recently approved ZILRETTA™ (triamcinolone acetonide),” state the authors.

Frames obtained from high-speed imaging capturing the evolution of the shape of a molten 30% IBU-PCL blend interacting with either a superoleophobic surface (top) and preserving its spherical shape after 1.8 s or an aluminum surface (bottom), in which the droplet gradually flattens (See SI Video 4 and SI Video 5, respectively). Scale bar: 500 µm. These frames emphasis the significance of using non-wetting surfaces responsible for the formation of spherical droplets.
With the advent of 3D printing and inkjet technology, researchers have experimented with numerous techniques. Many have encountered obstacles though, and challenges regarding methods relying on both inkjet printing and those that are solvent based. Because of that, the researchers created an affordable, yet solvent-free technique for fabricating polymer melts in this study—showing the capability of their SPHRINT technique.
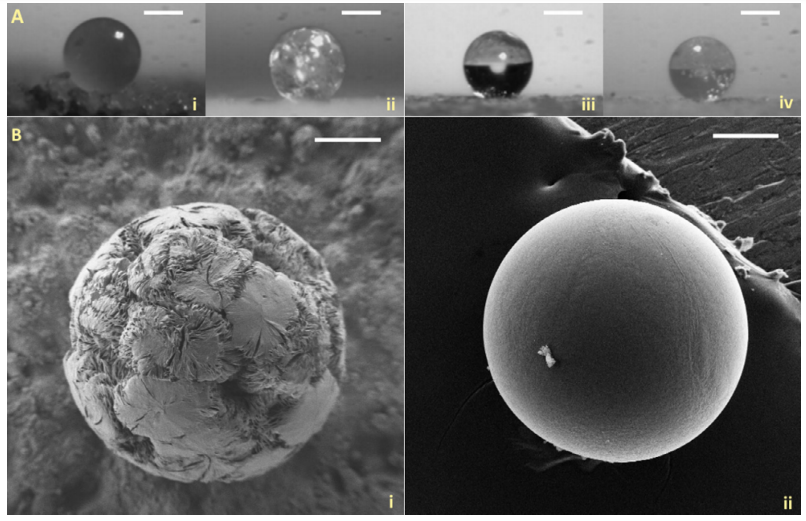
(A) Optical images of (i) neat PCL (154 ± 3°), (ii) 30% IBU-PCL (171 ± 4°), (iii) neat PLGA (167 ± 6°), and (iv) 30% IBU-PLGA (169 ± 4°) microspheres, cooled at room temperature (RT). The values in brackets denote the “sphericity,” expressed as the contact angle values (mean ± S.E.M., n = 10). Scale bar: 200 µm. (B) SEM images of (i) 30% IBU-PCL and (ii) 30% IBU-PLGA microspheres, cooled at RT. Scale bars: 100 µm
Producing drug delivery microspheres from a polymer loaded with a sample drug like ibuprofen, the researchers experimented and conducted an analysis regarding the potential for fabrication of amorphous polymeric microspheres. The researchers evaluated microsphere size, morphology, and texture. Ultimately, they were able to produce ‘near-perfect microspheres.’
“We discovered intricate physical phenomena governing the mechanism of sphere-formation; beside process and performance efficiencies, which in turn render microsphere products more accessible,” stated the researchers. “SPHRINT printing eliminates the use of organic solvents and surfactants; it offers microspheres with reproducible size, shape, and morphology within and between the batches; and the produced microspheres can be easily collected owing to their spherical shape.”
Jetting rate and shear rate were calculated, along with an investigation of melt interaction with the superoleophobic substrate and sphere formation.
In terms of drug encapsulation efficiency, they found that values in connection with SPHRINT were on the highest scale. The authors were encouraged to find ‘stable, consistent, reproducible results.’
“… we believe that SPHRINT may turn microsphere production ubiquitous, allowing for desktop manufacturing of microspheres easily scalable to industrial quantities (with a production rate of 25 Hz, in 1 h, 4.7 g of PCL microspheres may be printed from a single printing head). Finally, SPHRINT may provide a new dimension in reservoir-injectable drug delivery technologies, enabling the employment of multifarious polymers for microsphere production and tuned release profiles,” concluded the researchers.
3D printing has been used in connection with a variety of different drug delivery systems, from experimenting with microreservoirs to hydrophilic matrices, and even spermbots. What do you think of this news? Let us know your thoughts; join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘SPHRINT – Printing Drug Delivery Microspheres from Polymeric Melts’]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
AM & the Military’s Self-Infliction of Rapid Change
I’ve noted before that the additive manufacturing (AM) market for defense has started to evolve so quickly that it’s impossible to even keep track of all the updates in real...
ROBOZE Buys Dimanex Assets to Build “Physical AI” Platform
Dutch firm Dimanex got its start as an MRO platform for the railways. The company had a contract with the Dutch Army in 2018, and later that year signed one...
3D Printing Financials: Nano Dimension Reports Q1 Growth Amid Restructuring and Asset Sales
Nano Dimension (Nasdaq: NNDM) started 2026 with a much larger first quarter business than it had a year ago, mainly because Markforged is now part of the company and included...
3D Printing Financials: Stratasys Bets on Defense and Drones as Printer Sales Slow
Stratasys (Nasdaq: SSYS) started 2026 with lower revenue and a larger loss as customers continued to slow down spending on new 3D printers. Still, the company pointed to stable recurring...













































