3D Printing Plasticizer-Free Hydrophilic Matrices for Drug Delivery Systems
International researchers have been performing what could be very important work in relation to 3D printed medications, with their findings outlined in the recently published ‘Plasticizer-Free 3D Printed Hydrophilic Matrices: Quantitative 3D Surface Texture, Mechanical, Swelling, Erosion, Drug Release, and Pharmacokinetic Studies.’
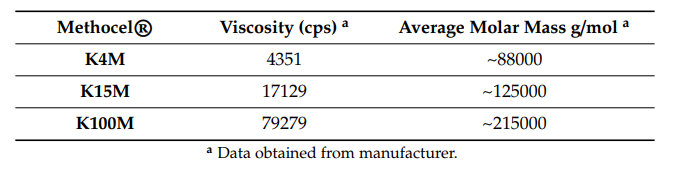
The focus of their study is hydroxypropyl methyl cellulose (HPMC), a hydrophilic polymer which could have enormous potential for fabrication of matrix tablets. Here, the research team analyzed plasticizer-free 3D printed hydrophilic matrices with drug-loaded filaments, examining their performance level in both in vitro and in vivo applications.
And while pharmaceutical companies worldwide may already be able to manufacture medication in the appropriate dose and shape—sometimes affordably so—with 3D printing they can offer patients what undeniably is the future of medicine with patient-specific treatment. This has already been documented with the FDA approval of Spritam, the first 3D printed epilepsy drug, and much discussion over how such technology will be harnessed in the future with the potential for DIY drugs.
“Although the current methods are common and cost-effective, they offer little opportunity for personalization and on-demand manufacturing as the change in size, shape and dose of a tablet require alterations at each manufacturing step and retooling of tableting machines,” said the researchers. “Consequently, a technology capable of producing dosage forms with a variety of strengths, such as three dimensional (3D) printing, is required to accomplish the personalized therapeutic needs of individuals.”
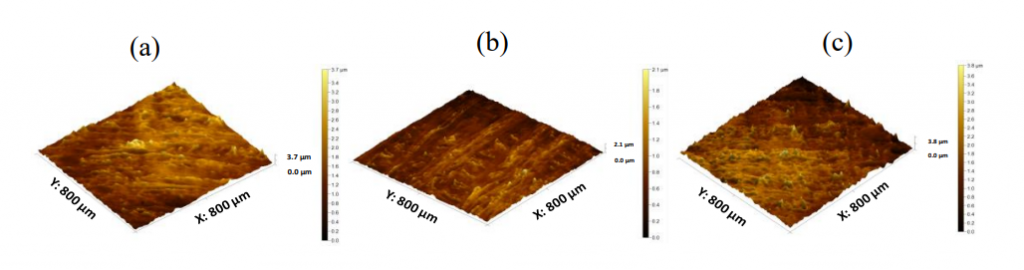
Previous researchers have tried to create 3D printed medications using a variety of different filaments, and with different composites and hardware. Polymers are an extremely common choice for such medications, with combinations such as plasticizers used to make extrusion easier. Such additions could, however, affect drug absorption negatively. For 3D printing via FDM, the materials must be strong and ‘mechanically robust’ enough to endure the fabrication process. Seam-line issues may arise, as well as problems with too much porosity. To compensate for previously known obstacles, the research team avoided the use of plasticizers. They also tested and evaluated the surface texture, employing both mercury intrusion porosimetry and white light optical profilometry.
“Glipizide which is an anti-diabetic drug of the sulfonylurea class indicated to treat type-2 diabetes was used as a model drug. Glipizide is a weak acid (pKa = 5.9), practically insoluble in water and acid, and is highly permeable (biopharmaceutics classification system, BCS II),” stated the researchers. “It appears to be an effective insulin secretagogue which reaches a peak plasma concentration within 1–3 h after a single oral dose with an elimination half-life of about 2–4 h. Such rapidly absorbed drugs having fast elimination rates with short half-life make it a suitable candidate to be considered for sustained delivery.”
The research team was successful in 3D printing the HPMC materials without using plasticizer.
“Overall, XRD spectra of glipizide and drug loaded HPMC filaments were consistent with the DSC profiles, thus, both methods showed that glipizide has a crystalline structure, whereas HPMC and drug loaded filaments have amorphous nature. This may potentially enhance dissolution but could negatively impact stability,” stated the researchers.
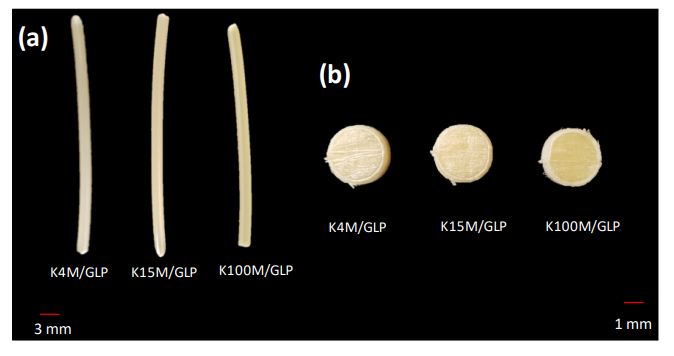
Ultimately, the loaded filaments did produce 3D printed hydrophilic matrices that were all similar in thickness, diameter, and weight; however, the 3D printed surfaces were found to be ‘considerably rougher.’
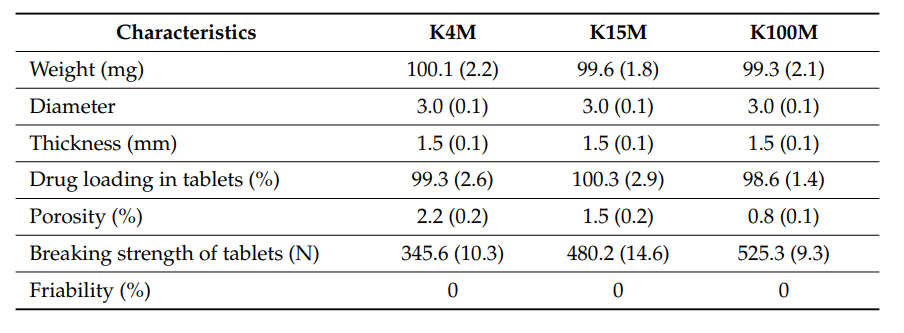
Geometrical and morphological characteristics of 3D printed hydrophilic matrices (n = 5, standard deviations are in parenthesis).
“… it can be concluded that the viscosity of HPMC has a noticeable impact of the swelling, erosion, HPMC dissolution, drug release and pharmacokinetic properties. The highest viscosity grade (K100M) tends to have a higher degree of swelling, decreased HPMC dissolution, low matrix erosion, decreased drug release and extended drug absorption profile,” stated the researchers. “Overall, this study confirmed the successful fabrication of 3D printed matrix tablets which have functionalities analogous to matrix tablets fabricated using conventional technologies. Moreover, the current study has also demonstrated the usefulness of the FDM technique, providing a simple solution to develop personalized pharmaceutical formulations in a time and cost-effective manner addressing challenges confronted by conventional manufacturing processes.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘Plasticiser-Free 3D Printed Hydrophilic Matrices: Quantitative 3D Surface Texture, Mechanical, Swelling, Erosion, Drug Release, and Pharmacokinetic Studies’]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
RAPID 2026: 6K Additive’s Domestic Metal Powders & Consolidation Plan
6K Additive (ASX: 6KA), a U.S. supplier and manufacturer of metal powders for additive manufacturing (AM), has been very busy lately. I caught up with CEO Frank Roberts and Chief...
3D Printing News Briefs, April 22, 2026: DINOs, Post-Processing, AM for Aerostructures, & More
We’ll start with event news in today’s 3D Printing News Briefs, as AMUG presented its DINO Award to six members at this year’s conference, and Axtra3D celebrated its five-year anniversary...
Medical, Electronics, & Semiconductors: Detailed 3D Prints at RAPID 2026 with Boston Micro Fabrication & Lithoz
They say that good things come in small packages, and that’s certainly the case when it comes to Boston Micro Fabrication (BMF). A leader in micro-precision additive manufacturing, the company...
Bambu Lab’s 3D Printed Drone Add-On Helps Measure Whales from the Sky
Bambu Lab has helped Tandem Ventures develop the WHASER, a 3D printed attachment for a drone that can measure whales. This seems like a nifty tool for researchers. It is...