VELO3D Releases Assure for 3D Metal Printing: Stratasys Direct Manufacturing as First Customer
With the release of the VELO3D Assure™ Quality Assurance and Control System for its Sapphire® 3D metal printers, VELO3D also brings on board a heavy hitter in their first customer as Stratasys Direct Manufacturing, a subsidiary of Stratasys, Ltd., will be the first to integrate Assure into their manufacturing processes.
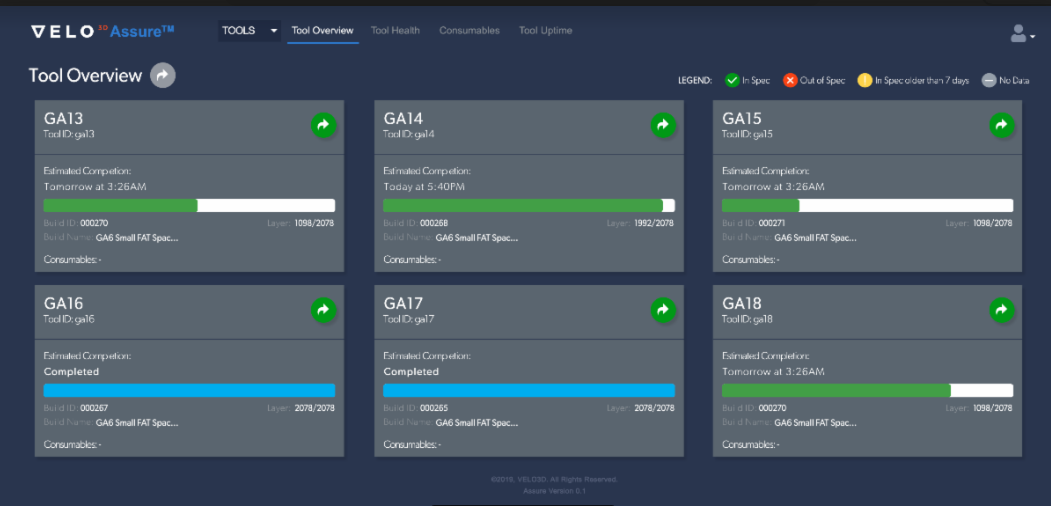
VELO3D’s Assure™ quality control dashboard enables engineers to track the quality and progress of Sapphire® machines in real-time.
Assure offers unprecedented monitoring and substantiation of part quality, offering the following features:
- Detects process anomalies
- Flags issues
- Highlights necessary corrective actions
- Offers traceability
“Assure is a revolutionary quality-control system, an inherent part of the VELO3D end-to-end manufacturing solution for serial production,” says Benny Buller, founder and CEO of VELO3D. “Assure is part of our vision to provide an integrated solution to produce parts by additive manufacturing with successful outcomes.”
Upon receipt of their own Sapphire 3D printer earlier this year, Stratasys Direct not only began using Assure, but they produced an entire study from their evaluation, which included:
- Monitoring integrity of builds
- Validating bulk material density
- Observing ongoing process metrics
- Verifying calibration of the system
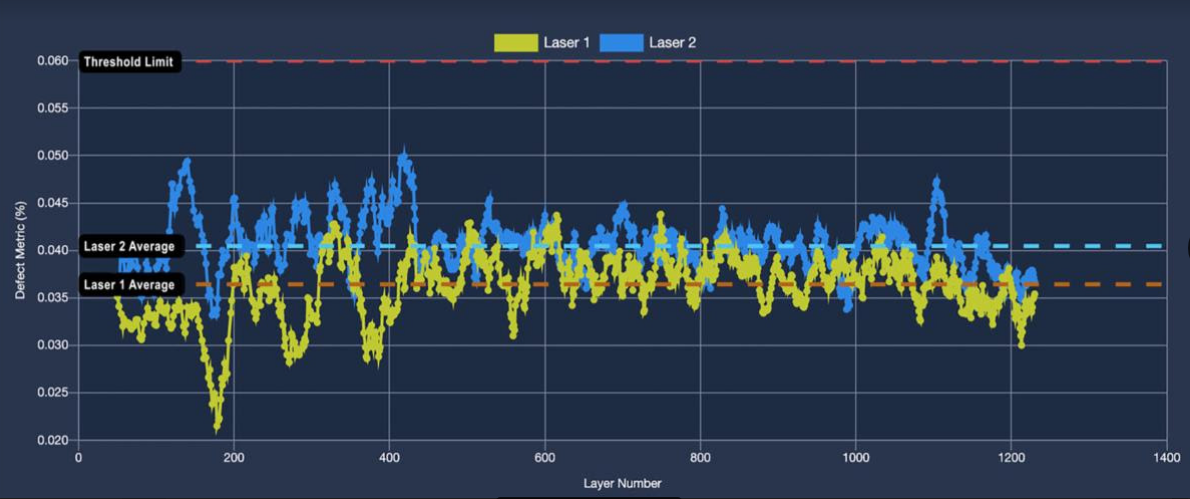
Assure predicts defectivity as a function of layer number. An increase in the defectivity metric is correlated with increasing defectivity in the bulk core of the part.
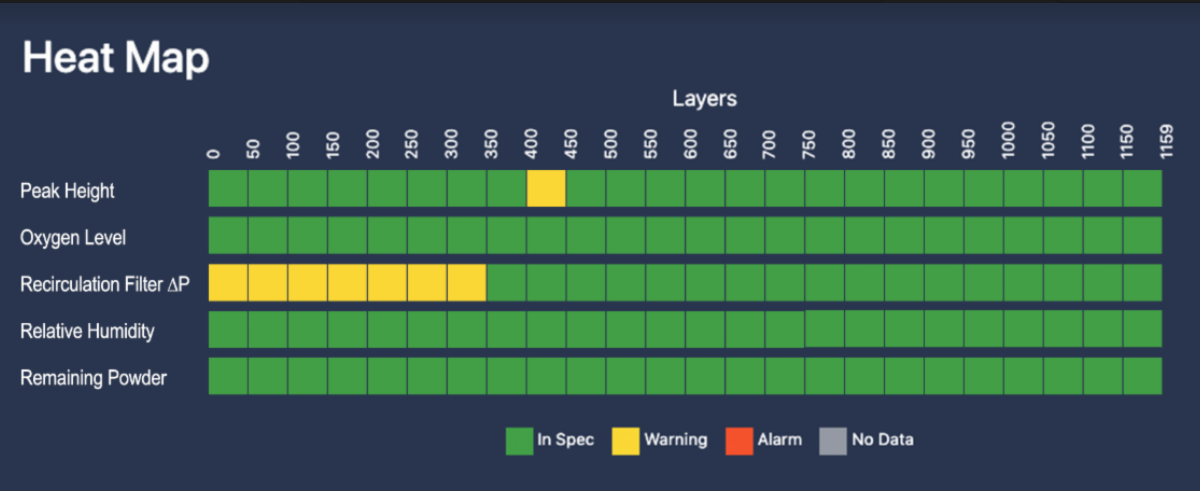
Before and during a build, Assure validates that critical parameters stay within control limits ensuring high quality parts. Clicking on individual squares reveals details on the underlying event.
These results were published in ‘Stratasys Direct Manufacturing Performs Field Validation of VELO3D Assure™,’ after the Stratasys Direct team used Assure for 12 weeks, verifying findings produced by VELO3D. They are now using the system in ongoing production efforts.
“AM can print parts and meet requirements for single units but scaling from a single part into serial production has been challenging. OEMs lack confidence in AM process control, and AM users struggle to demonstrate it. Without visibility into each part’s deposition lifetime AM becomes a risk,” states author Andrew Carter, Sr. Manufacturing Engineer at Stratasys Direct Manufacturing.
Assure boosts manufacturing techniques for the user as they can understand tool health better, calculate part quality, and perform field validation. Engineers 3D printed test structures during their study, producing wedges measuring 20mm x 41mm in width and length respectively. The wedges could be stacked into a tower shape, making a structure to match the build z-height. For each test run, they created two towers.
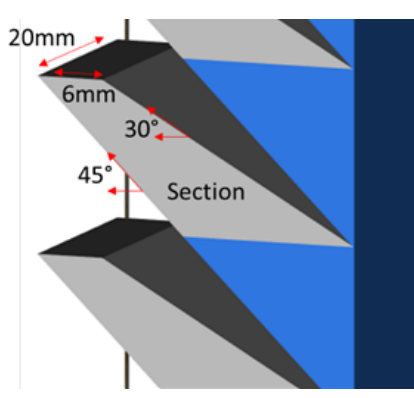
Test structure added to production builds to enable destructive testing. Image from ‘Stratasys Direct Manufacturing Performs Field Validation of VELO3D Assure™.’
Ultimately, 75 test structures were created and then analyzed via X-rays. Bulk porosity measured at 0.02 percent, and the researchers pointed out that there was no ‘single part exhibiting porosity higher than 0.1 percent. There were no deviations in print quality for the test builds.

Bulk defectivity measured on test parts by x-ray imagery. Image from ‘Stratasys Direct Manufacturing Performs Field Validation of VELO3D Assure™
“Stratasys Direct has built a culture of continuous improvement that means we are continually setting new standards for our industry on quality,” said Kent Firestone, CEO of Stratasys Direct Manufacturing. “We integrated Assure into our quality control workflow because it produces highly actionable insights. The user interface features intuitive graphs and charts that enable us to see and interpret the vast amount of data collected during builds. This information helps our engineers verify the quality of the build each step of the way and enables them to make quick decisions in the event of an issue. Assure helps us reduce production variation, improve yields, and circumvent anomalies to ensure consistent additive manufacturing.”
If you are interested in finding out more about Assure, check out the webinar on November 14th at 10 am PST, offered by Stratasys Direct. Click here to register. Also, if you are attending formnext in Frankfurt, Germany, don’t miss the joint press conference at the VELO3D booth (Hall 11, E79) on November 19 at 10 a.m.
VELO3D continues to be a dynamic presence in the 3D printing realm, from fabrication of a supersonic flight demonstrator to their efforts to expand on design and build limitations. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
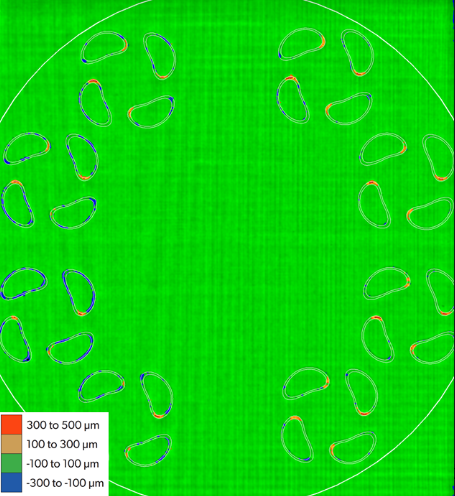
Assure provides true z-height quantitative powder bed and part metrology. Note the sections of parts with red lobes indicating metal protruding >300um above the powder bed but still below control limits.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
DMG Mori Joins $10M Defense 3D Printing Program
To look at the Biden administration and the Trump administration that succeeded it and find areas of policy overlap is obviously a bit of a challenge. But such areas certainly...
Where the Money Is Going: The New Infrastructure Landscape
This is Part 1 of a two-part PRO series examining where infrastructure investment is flowing and how those trends are reshaping manufacturing, energy, logistics, and additive manufacturing. Part 2, by...
First Containerized System from AML3D Now Operational at US Navy AM CoE
Shipbuilding giant Austal USA recently announced the launch of the Digital SEA (Secure Exchange for Additive) platform, which should play a major role in expanding accessibility to the sorts of...
INDOPACOM Advanced Manufacturing Team Saves Thousands Per Week at Joint Exercise in the Philippines
In the summer of 2025, the US Indo-Pacific Command (INDOPACOM) opened a new advanced manufacturing facility at Schofield Barracks, the Hawaiian home of the US Army’s 25th Infantry Division. INDOPACOM...














































