Researchers See Potential for 3D Printed Inhalers
US researchers created a study to compare conventionally made inhaler actuators with those that were 3D printed. The results were promising, as outlined in the recently published ‘Supporting Inhalation Drug-Device Combination Product Quality Using 3D Printing Technology.’ Continued, progressive research is critical to ongoing improvements in patient treatment—and especially with items like inhalers as they are used to treat breathing problems which could ultimately be life threatening.
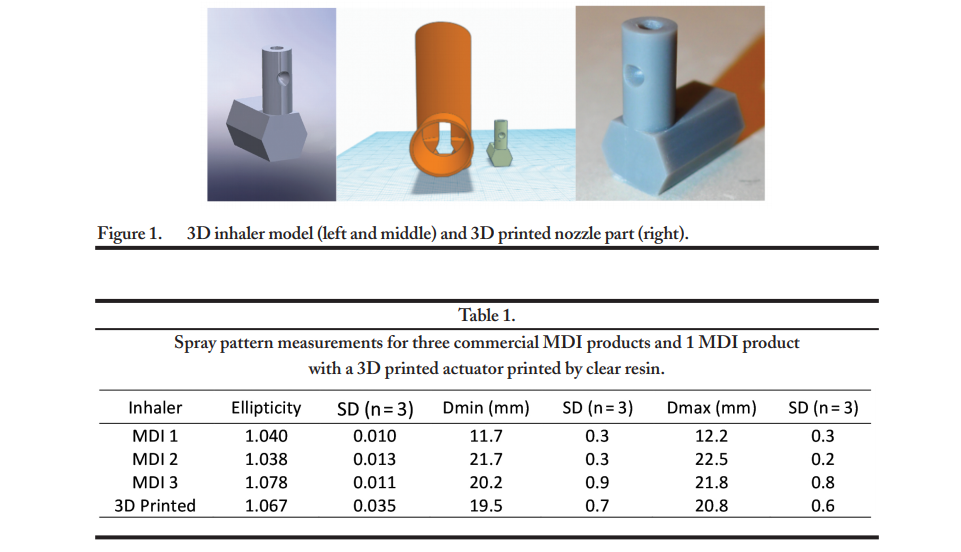
The researchers harkened back to the original form of 3D printing with stereolithography (SLA) 3D printing, using a Form 2 3D printer, with both grey and clear photoreactive liquid resin. The 3D printed actuators were fabricated in both vertical and horizontal positions, with a layer thickness of 25 µm.
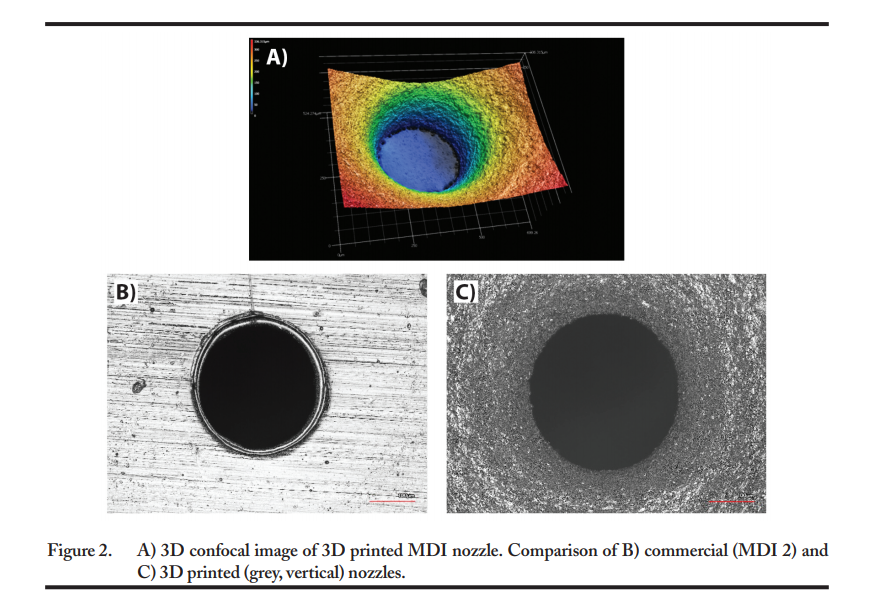
“Combined with a marketed MDI drug canister (120 metered actuations, 40 mcg dose of beclomethasone dipropionate in 50 microliters of solution formulation per actuation), the 3D printed actuators were characterized for their performance attributes including spray pattern, emitted dose, and aerodynamic particle size distribution,” explained the researchers. “The results were further evaluated by confocal and conventional microscopy to understand the impact of 3D printing material and process parameters on the quality and performance of MDI products.”
The 3D printed actuators exhibited spray patterns of mean ellipticity of 1.067, while the three commercial samples showed a range from 1.038 to 1.078.
“The elliptical ratio of the spray pattern was therefore used as rapid screening for the initial evaluation of actuator nozzle printing quality,” stated the researchers.
In this study, the research team also performed in vitro performance testing, specifically regarding particle size distribution and emitted dose.
Both conventionally made actuators for the inhalers and 3D printed samples ‘showed a close similarity,’ offering intense potential for the future of creating them through digital fabrication.
“When properly validated, data generated using 3D printed inhalation devices has the potential to provide supporting information for the scientific review of NDAs and ANDAs for oral inhalation drug products (OIDPs) submitted to the Agency,” conclude the researchers.
“The study of 3D printed device components using a series of innovative analytical tools, including 3D imaging, micro-computed tomography, high resolution, and high speed in situ spray visualization techniques, may help the pharmaceutical industry efficiently develop new and generic orally inhaled drug products. The information obtained from these studies may inform recommendations for MDI device constituent parts related to quality and performance, and current MDI device constituent part user interface recommendations for generic MDI products.”
While 3D printing in relation to inhalers is an important innovation, this technology has been used to create a wide array of medical devices just in the past few years—from the use of antibacterial materials to patient-specific implants, to new ways for creating diagnostics and training models too. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘Supporting Inhalation Drug-Device Combination Product Quality Using 3D Printing Technology’]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
China Becomes Latest Space Power to Demonstrate Metal 3D Printing in Orbit
China has demonstrated metal 3D printing in space as part of its plan to develop manufacturing technologies for future space missions, including Moon construction. The experiment took place aboard the Qingzhou...
AMPulse Asia: APAC 3D Printing Market Roundup
Key Takeaways Coverage window: April 27 to May 10, 2026. Roughly 30 additive manufacturing (AM)-relevant announcements were tracked across eight Asia-Pacific countries. Largest disclosures: Farsoon Technologies (688433.SH) filing a RMB...
3D Printing Financials: Stratasys Bets on Defense and Drones as Printer Sales Slow
Stratasys (Nasdaq: SSYS) started 2026 with lower revenue and a larger loss as customers continued to slow down spending on new 3D printers. Still, the company pointed to stable recurring...
3D Printing Financials: Xometry Surges After Record Quarter and Siemens Deal
Shares of Xometry (Nasdaq: XMTR) surged on Thursday, May 7, after the company reported record first-quarter 2026 results and announced a major partnership with Siemens. The stock climbed as much...