Charles River Associates International on Bioprinting
Charles River Associates International is a company that advises governments, law firms, and companies on weighty strategic matters and issues related to specific expertise that the company has. CRA may be unknown to you but its work affects us all. The company’s management consulting practice helps steer many decisions in the business world while their litigation support practice arms attorneys with the knowledge to win some of the most significant and most complex court cases. In antitrust cases with huge implications for consumers, workers, and firms CRA economists are the ones providing judge and jury with the analysis and data to support their decisions. In large acquisitions and mergers, CRA analysts and experts build foundations to ensure that regulatory approval will follow. CRA was eager to shine its many candle power light on bioprinting. Their interest in the nascent area is another data point that indicates that this technology can have wide-reaching implications for all of us. We interviewed the Charles River Associates Innovative Technologies Team to find out what they thought of the emerging world of bioprinting. I was super pleased with the clarity of thought of their answers and how the firm seems to have a realistic view of the immediate future of bioprinting which we also share.
What is CRA?
CRA is a leading global economics and management consultancy, serving private and public clients across a broad range of industries, including life sciences. A key area of focus for our team is assessing innovative business solutions and technologies—such as bioprinting—and providing strategic guidance on how they may shape the future of healthcare. For more than 25 years, life science companies, law
firms, and regulatory agencies around the globe have turned to CRA’s 200+ life sciences consultants when they need clarity and solutions to the industry’s most complex issues.
Why are you interested in bioprinting?
Bioprinting is fascinating because of its groundbreaking 3D technology and its potential for commercial applications. The field has advanced rapidly over recent years through engineering step-changes in the use of 3D printing devices – with these developments, living cells can now be positioned layer-by-layer to produce functional tissue structures. Key attributes of this emerging technology are its scalability and modularity, which allows for the manufacturing of a large variety of tissues in an automated and repeatable way. These high-throughput biofabrication capabilities equip developers with exciting tools to develop 3D-tissues for broad applications, from in vitro drug testing models, to therapeutic tissue implants, to whole organ transplants.
What will the impact of bioprinting be over the next 12 months? Five years? Beyond 10 years?
The bioprinting market is young, but it is close to producing engineered tissue products with high commercial potential. Several companies, including Organovo, Regenovo, and Aspect Biosystems, have had success developing and marketing 3D bioprinted tissue products that are being used for drug testing and pharmaceutical studies. The use of 3D tissue models in drug development, paired with the potential to create living tissue replacements and a rapidly advancing technological platform, have led the bioprinting sector to be valued at as much as $4.7B by 2025. While the bioprinting field is close to creating tissue products with high therapeutic value, the ultimate vision of creating engineered organs is still far in the future. Commercial adoption of bioprinted products will likely occur for in vitro tissue models first, within 5-10 years; for simple tissue grafts like skin and bone second, in 10-15 years; followed by vascularized patches and functional organ regeneration implants third, in 20-25 years, and organ transplants last, 3-4 decades in the future.
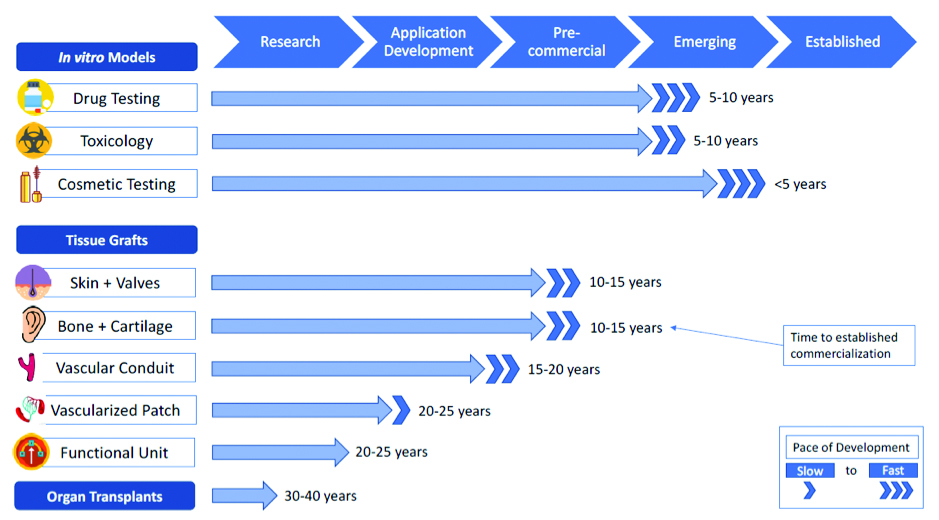
This figure demonstrates what the commercialization timeline for bioprinted products may look like in the coming years.CRA analysis based on findings from primary and secondary market research.
It looks as if this market is developing a bit like the medical device market where a lot of the pioneering work is being done by small firms. Does this mean that larger firms will start to swoop in?
Like the medical device industry, much of the pioneering development in the bioprinting field is being driven by smaller R&D players, but large pharmaceutical entities are already engaging the field. In particular, AstraZeneca (AZ) and Johnson & Johnson (J&J) are working to advance bioprinting technologies developed by smaller firms through early high-risk stages of commercial readiness. AZ established a ‘BioVentureHub’ that provides biotech companies with lab space and access to world-class technical and scientific professionals, with the aim of fostering collaboration. CellInk, a leading bioprinting company, has already moved four researchers into the lab space who are working to produce kidney, heart, liver, and lung tissue-models for use in pharmaceutical development. J&J announced the creation of a bioprinting research laboratory in early 2018 in collaboration with the Irish materials science institute AMBER at the J&J 3D Printing Center of Excellence in Dublin, an arrangement that will provide space for supported bioprinting research. While these collaborations are exciting, investors are still working to understand how to best provide bioprinting stakeholders the support needed to grow and evolve their products, through risk-riddled stages of commercial readiness and towards clinical reality.
The potential seems enormous but funding has been limited – why is that?
The bioprinting field is gaining traction at the commercial and technological level, but three key developmental hurdles are limiting funds flowing to bioprinting players who are attempting to evolve basic-demonstrative technologies through higher levels of proof and technical readiness. First, investors and developers need clearly identified and navigable regulatory pathways defined by the FDA to support
their confidence in the commercial potential of bioprinted products. Second, a target consumer population has not been clarified, given the wide variety of potential applications of bioprinted products and the many possible end-users of these technologies, which makes investors hesitant to commit to the field and does not allow developers to effectively direct their R&D efforts. Third, developers require an established R&D infrastructure to drive engineering innovations towards delivering commercially viable products, which is only possible if the bioprinting field can reach a critical mass of industry players equipped with a profitable revenue stream to support the creation of this fundamental infrastructure. All three of these hurdles must be overcome before the ambitious goal of developing and commercializing scalable, safe, and therapeutic bioprinting offerings can be broadly realized.
In 3D printing, in general, we’ve seen that the FDA has taken a very proactive and open role in working with the technology. Can we expect the same for bioprinting?
We are already seeing the FDA actively address and prepare for the imminent impact of bioprinted products by establishing close collaborations with bioprinting manufacturers and requesting specific proof-of-efficacy data. In a 2017 press release referencing bioprinting the FDA stated that they are, “preparing for a significant wave of new technologies that are nearly certain to transform medical practice,” and are, “working to provide a more comprehensive regulatory pathway that keeps pace with those advances and helps facilitate efficient access to safe and effective innovations that are based on these technologies.” While this proactive engagement is impressive, the FDA has yet to establish clear regulatory guidelines for bioprinted products. As noted above, bioprinting investors and manufacturers will require navigable regulatory pathways to support their confidence in the real-world applicability of bioprinted products.
Given the regulatory burden bioprinting firms will require a lot of cash. What kind of firms will fund them?
Right now, federal agencies are stepping up to the plate to provide the funds needed to support the development of basic demonstrative bioprinting technologies. Agencies such as the U.S. Department of Defense (DoD), European Medicines Agency (EMA) and NASA are committing extensive resources to advance bioprinting. These federal agencies have recognized the promise of bioprinting technologies in applications from expedited clinical trials to regenerative therapies for wounded veterans, and are directly investing in the biological and manufacturing infrastructure that is required to support the advancement of these technologies. The National Science Foundation (NSF) awarded multiple grants in September 2018, valued at $20 million each, to institutions dedicated to studying and developing
biomaterials specifically for use in bioprinting applications. In addition, the DoD recently awarded an $80 million grant to the Advanced Regenerative Manufacturing Institute (ARMI) in Manchester, New Hampshire, whose mission is to manufacture large-scale engineered tissues and organs and develop a trained and ready workforce necessary for that manufacturing.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
RAPID 2026 in Pictures: The Coolest & Craziest Things on the Show Floor
Last week, North America’s premier 3D printing trade show, RAPID+TCT 2026, came to Boston. I spent two days trekking the show floor, trying to see as much as I could,...
ExOne Cuts Costs for U.S. Customers as Printhead Production Moves to Detroit
ExOne Global Holdings, created through the 2025 integration of ExOne and voxeljet, is making changes across its U.S. operations. These include starting printhead manufacturing in the Detroit area and lowering...
Euler Viewer for Metal LPBF 3D Printing Released
Icelandic software startup Euler has released Euler Viewer, a real time build viewer for metal LPBF. The product does not need to be installed, and doesn’t require hardware to be...
3D Printing News Briefs, April 11, 2026: Energy Targets, DoW Contracts, Nike Air Max, & More
We’re starting with 3D printing for energy applications in this weekend’s 3D Printing News Briefs, and then moving on to military and defense 3D printing. Finally, Nike Sportswear is focusing...









































