Back in 2015, Erik Gatenholm realized there was no place to purchase bioink for 3D bioprinting. So, blown away by this gap in the market, he quickly worked with co-founder Hector Martinez to create a universal bioink that anyone working with bioprinting could use. It was quite a high stakes bet, and at the beginning they set up a webshop to see if they got any bites. It only took 24 hours for the first sale. With more orders quickly coming in, they realized the enormous potential of the product they developed, and CELLINK was born, becoming the first company to commercialize a universal bioink for bioprinting of human tissues and organs.
In the United States alone, every 10 minutes another person is added to the growing waiting list for organ transplants, most of them (60%) in need of a kidney, and with over 130,000 organs transplanted every year worldwide, is no wonder how demand certainly outweighs supply almost everywhere. In some countries the wait can take years, making 3D printing of organs one of the most sought after technologies out there. Bioprinting in the future could allow patients and doctors to reduce waiting times, increase compatibility and decrease immunological failure. For this to happen medical researchers will need to design organs using modeling software, and then print them with biomaterials such as polymers and hydrogels, in addition to the patient’s own cells. Although currently focused on growing cartilage and skin cells suitable for testing drugs and cosmetics, the Swedish company founded in Gothenburg in 2016, hopes to progress the technology far enough to create replacement organs for transplant in humans in the next 15 years.
“In the coming decade we would like to continue to push the boundaries of 3D bioprinting until it becomes an established technology in the medical field. We have the vision of becoming the first and number one provider of bioprinters, bioinks, software and technical know-how for the next generation of medical device manufacturers,” co-founder and CTO Hector Martinez told 3DPrint.com.
Their unique bioink is a biomaterial innovation that allows human cells to grow and thrive as they would in circumstances close to their natural environment. The startup has already managed to print human skin and is also working on producing liver tissues, as well as the beta cells that produce the insulin we need to survive. In 2018 it began printing tumors to combat cancer as part of a research project that doesn’t endanger human lives, and just a few weeks ago, it teamed up with Volumetric to develop Lumen X, a digital light processing bioprinter, designed to enhance inventions in creating more substantial vascular structures. Skin care products, topological drugs and medical treatments are all in need of enhanced testing procedures that can increase the transability from in vitro testing to in vivo usage of products. With tissue engineering and 3D bioprinting more representative in vitro models can be constructed, limiting the use of testing in animals.
Actually, academic labs and companies worldwide are trying to bioengineer all kinds of sophisticated creations for regenerative medicine, drug testing, screening, and tissue engineering. So it’s no wonder CELLINK has their research team focused on creating the next generation of bioinks. Their top selling product is making bioprinting much easier than it used to be some 10 years ago, with 30 different types of bioink available, with prices that go from 99 to 900 dollars. So, what makes one bioink more expensive than the other? It’s all about the components. Collagen and laminin are more expensive to produce than gelatin, raising the price of the end-product. According to CELLINK, scientists mix their live cells into the company’s bioink, a kind of gel designed to allow cells to survive and multiply. The ink is then loaded into a 3D printer by the customer, which forms the desired shape layer by layer as the gel solidifies. By the time the lights inside CELLINK’s box turn green, researchers have an object that acts like human tissue, and can then apply their drug and see how the living cells inside respond.
“Today we are taking the necessary steps to build and expand our technology offering and exploring new methods for bioprinting tissues. Such technologies include multiple contact-less dispensing methods and light-based bioprinting techniques that enable the bioprinting of high resolution tissue constructs. Refining such technologies will take a close collaboration with our customers as we define the best practices for bioprinting different tissues and specific functions. We can already anticipate that the integration of different bioprinting technologies with post-bioprinting, real-time monitoring systems will be of utmost importance as the bioprinted tissue matures and attains a specific function through an active and precise manipulation of its environment.” Chief IT Officer Jockum Svanberg explained to 3DPrint.com.
 Creating the raw material for bioprinting processes is no easy task. Cellink has been focusing on process-compatible soft biomaterials loaded with living cells to create its bioinks since September 2015. The process of bioprinting requires a delivery medium for cells which can be deposited into designed shapes acquired from computer-aided design (CAD) models, which can be generated using 3D medical images obtained through MRIs or CT scans. Some important features of an ideal bioink material are bioprintability, high mechanical integrity and stability, insolubility in cell culture medium, biodegradability at a rate appropriate to the regenerating tissue, non-toxicity and non-immunogenicity, and the ability to promote cell adhesion.
Creating the raw material for bioprinting processes is no easy task. Cellink has been focusing on process-compatible soft biomaterials loaded with living cells to create its bioinks since September 2015. The process of bioprinting requires a delivery medium for cells which can be deposited into designed shapes acquired from computer-aided design (CAD) models, which can be generated using 3D medical images obtained through MRIs or CT scans. Some important features of an ideal bioink material are bioprintability, high mechanical integrity and stability, insolubility in cell culture medium, biodegradability at a rate appropriate to the regenerating tissue, non-toxicity and non-immunogenicity, and the ability to promote cell adhesion.
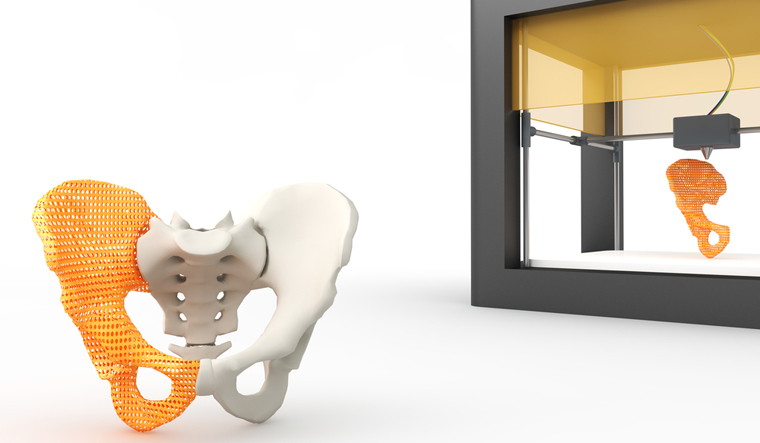
Some bioink types, like hydrogels, are not always suitable as construction materials which is why CELLINK is working on a study to provide an upgraded version of the current CELLINK BONE bioink by incorporating collagen and hydroxyapatite. The bioink currently offered does not get close to the real stiffness of the natural bone tissue, but finely resembles its chemical composition. The advantage of such a soft material is to be able to incorporate cells and, during the bioprinting process, to locate them at a precise position throughout the scaffold. This is still for research use only and might take a few years until it is compatible for human use.
Since its start the technology firm has grown to become one of the big competitors in the industry. CELLINK had only been in existence for ten months before they decided to pursue their IPO in November of 2016, listing on Nasdaq First North after a 1070% oversubscribed IPO, which means that demand for their shares was ten times what they expected. Since then, shares have risen over 400%, giving the company a present-day market cap of around $257 million. CELLINK’s affordable printers have already been bought by customers in 25 countries around the world, mostly universities, like Stanford, Harvard, Yale, Princeton and MIT, and some private customers, including Shiseido, Roche, Merck, Johnson and Johnson, and Toyota
But it’s not just about bioprinting it’s way into the future of medicine, CELLINK is also working with other disruptive technologies, such as machine learning. CELLINK told 3DPrint.com that “they want to empower our users with better tools to simplify the bioprinting learning process and broaden its adoption”. One example of this is by developing algorithms that analyse printed structures and based on the results can recommend printing parameters to the users. Using this tool in the development, has helped them speed up the bioink development process. They have just launched a new product: CELLCYTE X, a live cell imaging microscope with live monitoring and analysis of cells in the cloud. Traditionally cell studies have involved manual labor and relied on analysis of the images from an expert, but using deep learning models they are automating this process to provide better and more reliable analysis to their users. The system relies on the latest in serverless system architecture to provide the most scalable, reliable and most intuitive system on the market.
What do you think, will CELLINK continue its upward trajectory? Will it become superseded by other larger firms or get passed by newer start ups? Find out more through our series of articles exploring bioprinting, Biodiscoveries.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Orano Federal Services & UNC Charlotte Show How AM Could Cut Costs in Nuclear Energy Resurgence
Outside of the defense sector, few industries have been impacted by Russia’s ongoing occupation of Ukraine more than nuclear energy. The same appears to already be happening in response to...
HADDY’s Large-Format Robotic 3D Printing to Power Red Cat’s Drone Boat Production
In May 2025, Joris Peels, as is his custom, wrote a prescient article about Unmanned Surface Vehicles (USVs) and Unmanned Underwater Vehicles (UUVs), i.e., drone boats. Listing a multifaceted range...
EOS to Spotlight AI, Robotics, and Industrial Tooling at Hannover Messe
The US-Israel war on Iran is already catalyzing the sorts of major shifts to global supply chains that will effectively amount to permanent economic changes. In this context, the nations...
When Castings Take 18 Months: How 3D Printing Helped Fix the Soo Locks
This article is Part II of a two-part series on Lincoln Electric’s large-format metal additive manufacturing operations. In Part I, we looked at how Lincoln Electric built one of the...