Dassault Systèmes Announces Extended FDA Partnership, Dassault Aviation Collaboration, Earnings
 Dassault Systèmes makes numerous announcements this week, as they continue to lead in partnerships and projects around the world, usually connected to their proprietary 3DEXPERIENCE platform.
Dassault Systèmes makes numerous announcements this week, as they continue to lead in partnerships and projects around the world, usually connected to their proprietary 3DEXPERIENCE platform.
Currently the FDA has extended their collaboration regarding development of their cardiovascular device review tool for five years. This is meant to offer a completely new process for the medical industry, accelerating diagnosis and treatment of heart disease.
The second part of the collaboration is connected with the 21st Century Cures Act, with the creation of a fascinating process—again geared toward improving medical treatment—with the use of virtual patients and simulation for clinical trials.
“Our collaboration with the FDA underscores the relevance and sustainability of digital twin experiences created with the 3DEXPERIENCE platform to test devices and drugs in scientific and medical innovation,” said Claire Biot, Vice President, Life Sciences Industry, Dassault Systèmes. “Enriching technology already well established in regulated industries such as aerospace and automotive, virtual patients support the complex development of therapies for the heart, brain and more by eliminating traditional cost and time bottlenecks. With this new review process, Dassault Systèmes and the FDA can be partners in the transformative impact of the virtual world on industrial innovation, new treatments and the patient experience.”
The project is multi-faceted, also including a Living Heart simulated 3D heart model meant to function as a ‘source of digital evidence for new cardiovascular device approvals,’ including an in silico (by way of computer simulation) trial meant to decrease the need for testing on animals—or for patients to be involved in trials. Dassault Systèmes expects for this project and innovative digital process to be more efficient and less expensive.
“Modeling and simulation can help to inform clinical trial designs, support evidence of effectiveness, identify the most relevant patients to study, and assess product safety. In some cases, in silico clinical trials have already been shown to produce similar results as human clinical trials,” said Tina Morrison, Ph.D., Deputy Director in the Division of Applied Mechanics, Office of Science and Engineering Labs, Center for Devices and Radiological Health, FDA. “The FDA continues to encourage research to facilitate the introduction of safe and effective therapeutic solutions.”
Dassault Aviation also continues to push their Next Generation Enterprise platform forward, with six new ‘industry solution experiences’ meant to offer better performance for businesses with streamlined performance and better savings on the bottom line. They will be focused on the following features, based on the 3DEXPERIENCE platform:
- Winning Concept
- Program Excellence
- Co-design to Target
- Ready for Rate
- Build to Operate
- Keep Them Operating
With 3DEXPERIENCE, businesses can integrate 3D design, analysis, simulation, and more in one, comprehensive digital environment. Teams can collaborate more efficiently—along with communicating with their entire global supply chain, partners, contractors, and more.
“As we move forward with our digital transformation, we are using the 3DEXPERIENCE platform to develop a digital, multi-program approach with easy access to data, especially for program management, procurement and quality processes,” said Jean Sass, Chief Digital Officer, Dassault Aviation. “All of our industrial partners will work in collaboration with their customers on one digital platform. Ultimately, this will enable us to provide our customers with new, innovative flying experiences.”
Dassault Aviation’s Falcon range of aircraft has been in demand for over 50 years, with more than 2,100 of them in service in 90 countries. In the future, these aircraft will continue to be refined for the following:
- Low fuel consumption levels
- Low CO2 emissions
- Comfort and distance features
“The 3DEXPERIENCE platform represents a holistic approach to innovation,” said David Ziegler, Vice President Aerospace & Defense Industry, Dassault Systèmes. “It equips Dassault Aviation with integrated technologies and capabilities to connect all the dots from concept to operations, bring real-world data into the virtual world for analysis and action, build powerful, dynamic value networks of suppliers, and coordinate knowledge, know-how and processes to build their future aircraft.”
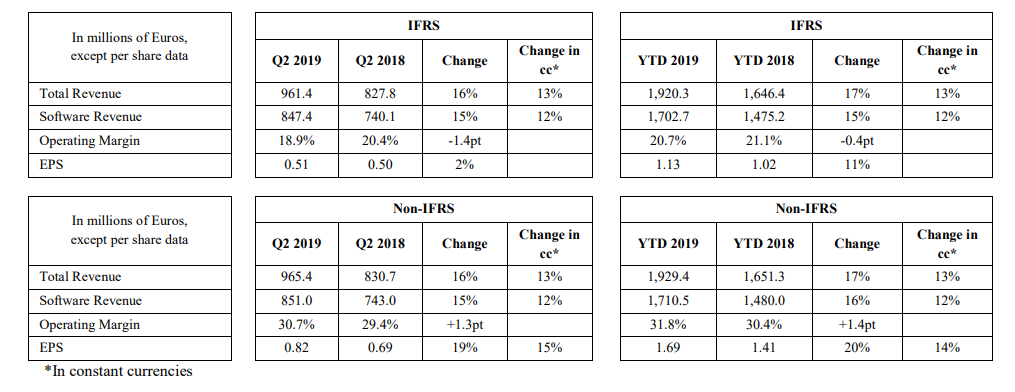
Dassault Systèmes has also released their first-half revenue statement, reporting growth in in the double digits—along with reaffirming their financial goals for 2019. These figures offer results for both the second quarter and the first half, ended June 30, 2019:
- The second quarter shows non-IFRS revenue increasing by 13 percent, and software revenues increasing by 12 percent. According to Dassault Systèmes, these figures are on the ‘high end’ of their financial objectives range.
- From an ‘organic basis,’ non-IFRS revenue was up 10 percent and non-IFRS software revenue was up 9 percent in Q2.
- 3DEXPERIENCE non-IFRS software revenue was up 40 percent in H1.
- Dassault Systèmes and Medidata signed and definitive acquisition agreement on June 12, 2019.
- Updated 2019 financial objectives target non-IFRS EPS of €3.45-3.50, well aligned with a five-year plan to double non-IFRS earnings per share in 2019.
“We believe the 3DEXPERIENCE platform is a critical enabler for innovation and transformation across all major industries where the drive to provide new types of customer experiences and new business models is emerging and accelerating. We see these possibilities across the three spheres addressed by our purpose: Product, Nature and Life and the proof points with our strategic wins across a number of diverse industries over the last two years,” said Bernard Charlès, Dassault Systèmes’ Vice Chairman and Chief Executive Officer.
“Our plan to acquire Medidata, announced in June, fits perfectly into our strategy in that regard. As the Life Sciences industry shifts to science-based experience, we believe scientific innovation 2 and industrial performance call for a unified new approach and this is what we have been working towards with our investments in Life Sciences over the past years. The acquisition of Medidata, with its clinical and commercial solutions, reinforces our position as a science-based company by providing the Life Sciences industry with an integrated business experience platform for an end to-end approach to research and discovery, development, clinical testing, manufacturing and commercialization of new therapies and health technologies.
“Thanks to our platform strategy, more companies view us as a strategic partner to help them transform. We were very honored and pleased that Groupe PSA has named Dassault Systèmes a key supplier, and its preferred digital partner for its digital transformation. With Dassault Systèmes as Groupe PSA’s preferred digital partner, the two companies are engaging in a long term strategy with the intent to deploy the 3DEXPERIENCE platform as a key innovation enabler across the group’s activities.”
Dassault Systèmes continues to be a dynamic force also with recent collaborations, acquisitions, and a long and impressive client base continually signing on to adopt the 3DEXPERIENCE Platform. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: Dassault Systèmes press release]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
ARC & ORNL Form Partnership to Accelerate AI-Enabled Manufacturing for Defense
Last year, Autonomous Resource Corporation (ARC) became the surprising owner of Desktop Metal’s (DM’s) assets following the bankruptcy of the one-time additive manufacturing (AM) unicorn, an acquisition that cost ARC...
RAPID 2026 in Pictures: The Coolest & Craziest Things on the Show Floor
Last week, North America’s premier 3D printing trade show, RAPID+TCT 2026, came to Boston. I spent two days trekking the show floor, trying to see as much as I could,...
Euler Viewer for Metal LPBF 3D Printing Released
Icelandic software startup Euler has released Euler Viewer, a real time build viewer for metal LPBF. The product does not need to be installed, and doesn’t require hardware to be...
3D Printing News Briefs, April 8, 2026: LiDAR Scanning, Vapor Smoothing, FDM Optimization, & More
We’ll kick off today’s 3D Printing News Briefs with some 3D scanning news from Artec 3D, and then move on to new America Makes Project Calls. Then, Raise3D and AMT...