Xometry: On-Demand Manufacturing Marketplace Completes $50MM Funding Round
Led by Greenspring Associates, Xometry has just announced $50MM raised in equity funding. Also funding the on-demand manufacturing marketplace are:
- Dell Technologies Capital
- BMW i Ventures
- Foundry Group
- GE Ventures
- Highland Capital Partners
- Maryland Venture Fund
- Almaz Capital
Founded in 2014 as a manufacturing startup and headquartered in Maryland, Xometry has now raised a total of $113MM, leading us to wonder how they will designate these funds in relation to research and development—a question answered succinctly by Randy Altschuler, co-founder and CEO of Xometry:
“Xometry’s vast network, massive data sets and AI breakthroughs make it easier for engineers and designers to buy custom manufacturing and help manufacturers more efficiently operate their businesses. We’re planning to invest these funds behind growth initiatives, product development and global expansion.”
Allowing for everyone to benefit from custom manufacturing, Xometry has created a successful building model for being able to produce smaller, detailed numbers of parts for consumers rather than requiring mass orders due to profitability requirements like most product manufacturers today—and especially those still using more conventional methods aside from 3D printing and additive manufacturing. The Xometry team is able to meet on-demand needs for parts to a customer base spanning from startups to Fortune 100 companies.
“Xometry has quickly become a key player in the $80B custom manufacturing market,” said Hunter Somerville, General Partner for Greenspring Associates, who will also join Xometry’s Board of Directors.. “We’re thrilled to invest in helping them expand the reach and breadth of their marketplace and the services it offers.”
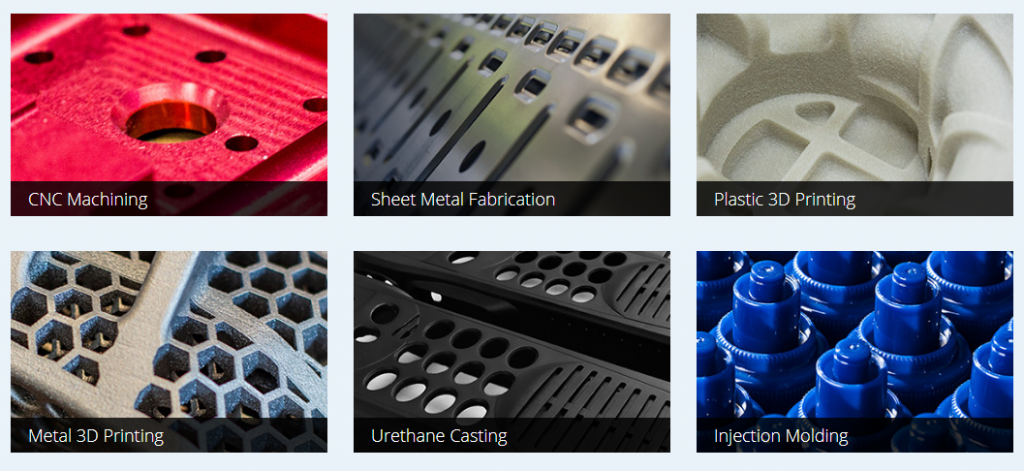
Xometry maintains a network of over 3,000 partnering facilities for manufacturing, allowing them to produce parts for companies like BMW, Dell, NASA, and GE through:
- 3D printing services
- Traditional CNC machining processes
- Sheet metal fabrication
- Injection molding
- Die casting
- Stamping
- Extrusion
- Urethane casting
“Xometry is yet another example of how machine learning based intelligent software is driving huge efficiency gains across many different industries,” said Daniel Docter, Managing Director Dell Technologies Capital. “We look forward to helping Xometry expand into new verticals that leverage the expertise of Dell Technologies.”
Their growth has continued at rapid speed, with revenue doubling in just the past year. The Xometry team has also created a spin-off with Xometry Supplies—meant to offer Xometry customers with materials and tools for manufacturing more rapidly, and affordably.
We have been following Xometry’s 3D printing progress for years, since they transformed from NextLine Manufacturing into their current and dynamic state, as the largest custom manufacturing marketplace available to consumers today. From streamlining the way engineers order 3D printed parts to changing the rules in manufacturing and even guiding us in choosing the best 3D printing materials, Xometry is geared toward making life easier for engineers and designers, catering to their specific needs.
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: Xometry]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Srini Kaza Discusses Strategically Scaling Align’s “Smile-Changing” 3D Printed Aligners
Align Technology‘s Invisalign is a revolutionary method to get you the smile you want through 3D printing. It is also a hugely popular process to go through, a $4 billion...
Analysis: Additive Manufacturing Strategies Spotlights Vertical Value Creation
A slowdown in the industry and particularly a tightening of access to capital following the additive manufacturing (AM) industry’s peak special purpose acquisition company (SPAC) phase in early 2021 is...
Department of Defense Awards PROTECT3D $1.3M for Custom 3D Printed Braces
Custom prosthetics company PROTECT3D has secured a $1.3M Department of Defense (DoD) Small Business Innovation Research contract to develop a platform for creating custom braces for soldiers. The PROTECT3D Platform...
AMS 2025 Highlights Big Changes in 3D Printing with Pivotal Speakers and Panels
2023 was filled with excitement around the potential mergers being pursued by industry stalwart Stratasys (Nasdaq: SSYS), leading to some of the most insightful conversations imaginable at Additive Manufacturing Strategies...