3D Printing Used to Predict Behavior of Replacement Heart Valves
3D printing has been studied by several different institutions as a method for aiding in a process called transcatheter aortic valve replacement, or TAVR. More than one in eight people aged 75 and older in the United States develop moderate to severe blockage of the aortic valve, often caused by calcified deposits that build up on the valve’s leaflets and prevent them from fully opening and closing. Many of these patients are not healthy enough to undergo open heart surgery, so TAVR is an alternative that involves deploying an artificial valve via a catheter inserted into the aorta.
Inserting the properly sized valve is critical; if the valve is too small, it can dislodge or leak around the edges, and if it’s too large it can rip through the heart, which can be fatal. It’s a challenge to select the correct size without directly examining the patient’s heart, however. But researchers at the Wyss Institute for Biologically Inspired Engineering at Harvard University have come up with a 3D printing workflow that creates models of individual patients’ aortic valves using CT scan data, in addition to a “sizer” device that helps cardiologists determine the proper valve size. The work is documented in a paper entitled “Pre-procedural fit-testing of TAVR valves using parametric modeling and 3D printing.” The research was carried out in collaboration with researchers and physicians from Brigham and Women’s Hospital, The University of Washington, Massachusetts General Hospital, and the Max Planck Institute of Colloids and Interfaces.
“If you buy a pair of shoes online without trying them on first, there’s a good chance they’re not going to fit properly. Sizing replacement TAVR valves poses a similar problem, in that doctors don’t get the opportunity to evaluate how a specific valve size will fit with a patient’s anatomy before surgery,” said James Weaver, Ph.D., a Senior Research Scientist at the Wyss Institute who is a corresponding author of the paper. “Our integrative 3D printing and valve sizing system provides a customized report of every patient’s unique aortic valve shape, removing a lot of the guesswork and helping each patient receive a more accurately sized valve.”
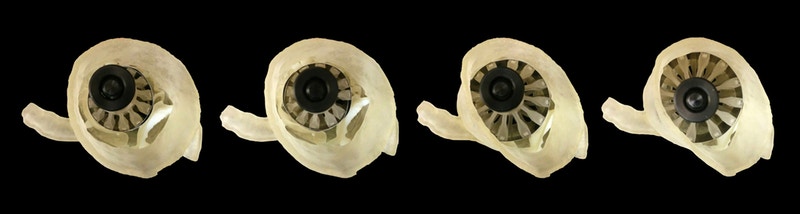
When a patient needs a new heart valve, they typically get a CT scan, but while the outer wall of the aorta and any calcified deposits are easily seen on a scan, the leaflets that open and close the valve are often too thin to show up clearly.
“After a 3D reconstruction of the heart anatomy is performed, it often looks like the calcified deposits are simply floating around inside the valve, providing little or no insight as to how a deployed TAVR valve would interact with them,” Dr. Weaver said.
To address this issue, Ahmed Hosny, who was a Research Fellow at the Wyss Institute at the time, created a software program that uses parametric modeling to generate virtual 3D models of the leaflets using seven coordinates on each patient’s valve that are visible on CT scans. The 3D models were then merged with the CT data and adjusted so that they fit into the valve correctly. The resulting model, which incorporates the leaflets and their calcified deposits, was then 3D printed in multiple materials.
The researchers also 3D printed a custom sizer device that fits inside the 3D printed valve and expands and contracts to determine what size artificial valve would best fit each patient. They then wrapped the sizer with a thin layer of pressure-sensing film to map the pressure between the sizer and the 3D-printed valves and their associated calcified deposits, while gradually expanding the sizer.
“We discovered that the size and the location of the calcified deposits on the leaflets have a big impact on how well an artificial valve will fit into a calcified one,” said Hosny, who is currently at the Dana-Farber Cancer Institute. “Sometimes, there was just no way a TAVR valve would fully seal a calcified valve, and those patients could actually be better off getting open-heart surgery to obtain a better-fitting result.”
The multi-material 3D printed valve models could also more accurately mimic the behavior of real heart valves during artificial valve deployment, as well as provide haptic feedback as the sizer is expanded. The researchers tested the system against data from 30 patients who had already undergone TAVR procedures. 15 of those patients had developed leaks from too-small valves. The researchers predicted, based on how well the sizer fit into the 3D printed models of their aortic valves, what size valve each patient should have received, and whether they would experience leaks after the procedure. The system successfully predicted leak outcome in 60 to 73% of the patients, depending on the type of valve each patient had received, and determined that 60% of the patients had received the correctly sized valve.
“Being able to identify intermediate- and low-risk patients whose heart valve anatomy gives them a higher probability of complications from TAVR is critical, and we’ve never had a non-invasive way to accurately determine that before,” said co-author Beth Ripley, M.D., Ph.D, an Assistant Professor in the Department of Radiology at the University of Washington who was a Cardiovascular Imaging Fellow at Brigham and Women’s Hospital when the study was done. “Those patients might be better served by surgery, as the risks of an imperfect TAVR result might outweigh its benefits.”
The researchers have made their leaflet modeling software and 3D printing protocol available online for free.
“At the core of the personalized medicine challenge is the realization that one medical treatment will not serve all patients equally well, and that therapies should be tailored to the individual, said Wyss Institute Founding Director Donald Ingber, M.D., Ph.D., who is also the Judah Folkman Professor of Vascular Biology at Harvard Med’ical School and the Vascular Biology Program at Boston Children’s Hospital, as well as Professor of Bioengineering at Harvard’s School of Engineering and Applied Sciences. T’his principle applies to medical devices as well as drugs, and it is exciting to see how our community is innovating in this space and attempting to translate new personalized approaches from the lab and into the clinic.”
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
[Source/Images: Wyss Institute]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: Velo3D Revenue Up Fueled by Defense Momentum
Velo3D (Nasdaq: VELO) reported a strong start to 2026, with revenue rising as defense and aerospace customers continued shifting from pilot programs into full-scale additive manufacturing (AM) production. The company...
AMS 2027: The AM Industry’s Biggest Business Conversations Return to New York
Additive Manufacturing Strategies (AMS) is heading back to New York next February for what has become one of the industry’s most important business gatherings. The 10th annual AMS conference (or...
AM & the Military’s Self-Infliction of Rapid Change
I’ve noted before that the additive manufacturing (AM) market for defense has started to evolve so quickly that it’s impossible to even keep track of all the updates in real...
ROBOZE Buys Dimanex Assets to Build “Physical AI” Platform
Dutch firm Dimanex got its start as an MRO platform for the railways. The company had a contract with the Dutch Army in 2018, and later that year signed one...