3D printed medical implants are typically stiffer and stronger than the surrounding tissue, and while they don’t always adapt well to biological and physical stimuli, that stiffness is important. The biological processes of our bodies are naturally dynamic, and many researchers are working to ensure that 3D printed implants are of the highest quality and will work well within the human body.
Living tissues constantly remodel in response to changes. In particular, changes in cellular and extracellular matrix (ECM) stiffness are important to many pathological and physiological processes, like migration, cell motility, and stem cell differentiation. But scientists don’t quite understand the biomechanical factors that are instrumental in soft tissue development and maintenance. That’s why we hear so often about tissue engineering – combining cells with scaffolds, 3D printed or otherwise, to fabricate a self-sustaining tissue replacement.
In a new paper, titled “Stiffness memory nanohybrid scaffolds generated by indirect 3D printing for biologically responsive soft implants,” a collaborative team of researchers from University College London, Brunel University London, and the Royal Free London NHS Foundation Trust developed thermoresponsive poly(urea-urethane) (PUU) nanohybrid scaffolds, with stiffness memory, using a 3D printing-guided, thermally induced phase separation (3D-TIPS) method.
The abstract reads, “Cell and tissue stiffness is an important biomechanical signalling parameter for dynamic biological processes; responsive polymeric materials conferring responsive functionality are therefore appealing for in vivo implants. We have developed thermoresponsive poly(urea-urethane) nanohybrid scaffolds with ‘stiffness memory’ through a versatile 3D printing-guided thermally induced phase separation (3D-TIPS) technique. 3D-TIPS, a combination of 3D printing with phase separation, allows uniform phase-separation and phase transition of the polymer solution at a large interface of network within the printed sacrificial preform, leading to the creation of full-scale scaffolds with bespoke anatomical complex geometry. A wide range of hyperelastic mechanical properties of the soft elastomer scaffolds with interconnected pores at multi-scale, controlled porosity and crystallinity have been manufactured, not previously achievable via direct printing techniques or phase-separation alone. Semi-crystalline polymeric reverse self-assembly to a ground-stated quasi-random nanophase structure, throughout a hierarchical structure of internal pores, contributes to gradual stiffness relaxation during in vitro cell culture with minimal changes to shape. This ‘stiffness memory’ provides initial mechanical support to surrounding tissues before gradually softening to a better mechanical match, raising hopes for personalized and biologically responsive soft tissue implants which promote human fibroblast cells growth as model and potential scaffold tissue integration.”
Schematic outlining the multi-micro-channel-controlled phase separation of 3D PUU-POSS scaffolds with hierarchical interconnected porous structure.
While the controlled elastic modulus of several responsive polymeric materials for tissue engineering have been used as a model for studying stiffness effects in cell cultures, their combined elasticity and molecular structural change coupling effect does not have a wide range of tuneable stiffness. But block-copolymers, like polyurethane (PU) elastomers, have many tuneable properties without having to change their molecular structure – the method of self-assembly just needs to be switched up. When used in long-term implantable cardiovascular devices, PUU has previously demonstrated excellent biocompatability, biostability, compliance, and fatigue resistance, and some PUs even have shape memory.
By using materials that are responsive to stimuli, it’s possible to print dynamic 3D structures that can transform their shapes or behavior – like stiffness.
“This work has contributed to the development of a range of thermoresponsive nanohybrid elastomer scaffolds, with tuneable stiffness and hierarchically interconnected porous structure, manufactured by a versatile indirect 3D printing technique,” the researchers wrote. “For the first time, stiffness memory of the scaffold was observed to be driven by phase transition and a reverse self-assembly from a semicrystalline phase to a quasi-random nanostructured rubber phase. Early insight into cell response during the stiffness relaxation of the scaffolds in vitro holds promise for personalized biologically responsive soft implants.”
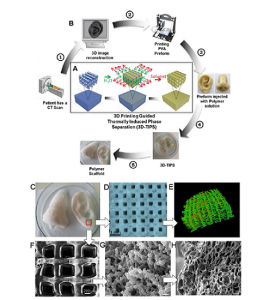
The team used AutoCAD 2014 to design 3D printable polyvinyl alcohol (PVA) preforms for the manufacturing and characterization of PUU-POSS scaffolds, before exporting the STL files into Slic3r software, “where they were sliced into an array of consecutive 200μm layers” for versatile, cost-effective 3D-TIPS indirect printing.
The researchers wrote, “Reconstructed digital .stl file images of an ear and nose were obtained from CT-scan images, sliced with 50% infill density, and printed as PVA preform. PUU-POSS solution was injected into each preform, and the preform washed as outlined for the RTC+H groups above, to produce anatomically shaped polymer scaffolds.”

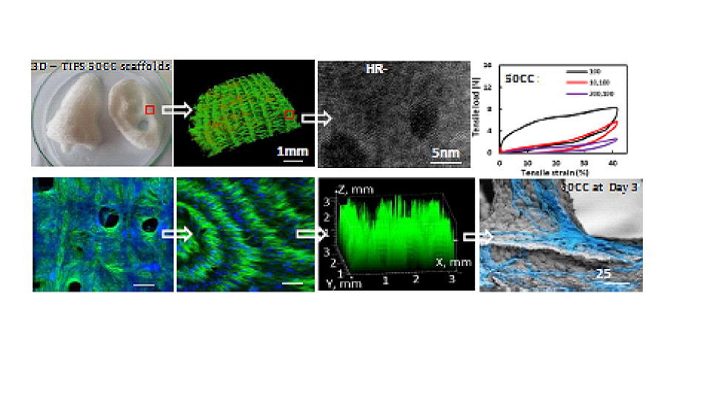
Phase transitions, crystalline structure, pore size distribution, morphology and mechanical properties of 3D PUU-POSS scaffolds (50% infill density) with varying processing temperatures.
The team used its 3D-TIPS indirect printing technique to manufacture a body temperature-responsive, bespoke tissue scaffold out of a PUU-POSS nanohybrid elastomer solution, which was confined inside a scalable, 3D printed, interconnected PVA preform network. The team produced 3D scaffolds with uniform, identical macroscopic dimensions and polymer content, but variable cellular and biomechanical properties, by thermally controlling PUU solution coagulation and micro-phase separation of polymer chains within the network.
“The most intriguing property of these scaffolds, that of ‘stiffness memory’, is driven by the reverse self-assembly of phase transition of 3D ordered crystalline structure into quasi-random nanostructures of soft and hard segments at body temperature, which imparts the unique and desirable dynamic mechanical properties of stiffness relaxation with unnoticeable shape change,” the researchers explained. “The small volume changes, due to the relaxation of ordered chain packing at the melting point of soft segments within the uniform interconnected porous network, contribute to the stability of the scaffold shape with little deformation.”
No matter what the initial stiffness was at various thermal process conditions, the PUU-POSS scaffolds made with the team’s 3D-TIPS method would ‘remember’ to relax into their hyperelastic rubber phase once they’d reached the melting temperature of the soft segments.
“3D-TIPS technique opens up the use of a wide range of biofunctional and previously unprintable polymers and their nanocomposites and permits tunability in the architecture and stiffness of fabricated constructs not previously achievable via direct printing techniques or phase-separation alone,” the team concluded. “The method is low-cost with a short-lead time, even for intricate and bespoke patient-specific implants. The potential for improved cell growth and bio-responsiveness of scaffolds in the postoperative healing period, due to stiffness relaxation, represents a promising technique platform for the development of biological responsive tissue-engineered implants, devices and surgical robotics, with matched dynamic mechanical properties to suit dynamic cell-lines, tissues and organs.”
Co-authors of the paper are Linxiao wu, Jatinder Virdee, Elizabeth Maughan, Arnold Darbyshire, Gavin Jell, Marilena Loizidou, Mark Emberton, Peter Butler, Ashley Howkins, Alan Reynolds, Ian W. Boyd, Martin Birchall, and Wenhui Song.
Discuss this research and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the comments below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Financials: Fathom Struggles in Financial Quicksand During Critical Transition
Facing a year of key transitions and financial pressures, Fathom (Nasdaq: FTHM) has filed its annual report for 2023 with the U.S. Securities and Exchange Commission (SEC). The document outlines...
Latest Earnings Overview for Australian 3D Printing Firms Titomic and AML3D
Australian 3D printing manufacturing firms Titomic (ASX: TTT) and AML3D (ASX: AL3) reported their financial results for the period from July to December 2023, marking the first half of their...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...
3D Printing Financials: Unpacking Farsoon and BLT’s 2023 Performance
In the Chinese 3D printing industry, two companies, Farsoon (SHA: 688433) and Bright Laser Technologies, or BLT (SHA: 688333), have recently unveiled their full-year earnings for 2023. Farsoon reported increases...