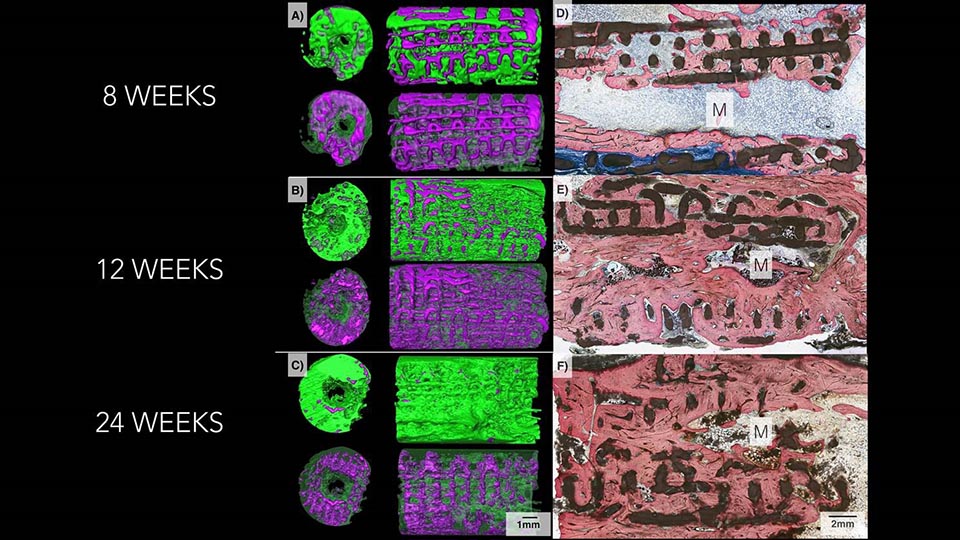
The imaging on the left shows how bone, in green, replaced the bioactive ceramic scaffold, in purple, over a six-month period. Microscope images on the right show progressively increasing degrees of bone, stained pink, and lower amount of scaffold, in black, as time goes by in the body.
Patients with missing bone due to injury or disease have a couple of different options for bone grafts to replace it – autografts, which come from the patient’s own body, or allografts, which come from a donor. Both have the potential for complications, but researchers at New York University are working on developing another option: 3D printed ceramic implants that dissolve slowly within the body, stimulating bone to grow in their place. 3D printing allows scientists to create implants that precisely fit the defects; they can also be coated with chemicals that promote bone growth.
The research is published in a paper entitled “Form and Functional Repair of Long Bone Using 3D Printed Bioactive Scaffolds,” which you can access here. According to the researchers, the animals they tested were able to absorb the implants into their natural bone, healing defects.
“Our three-dimensional scaffold represents the best implant in development because of its ability to regenerate real bone,” says study senior investigator and biomedical engineer Paulo G. Coelho, DDS, PhD, the Dr. Leonard I. Linkow Professor at NYU Dentistry and a professor of plastic surgery at NYU School of Medicine. “Our latest study results move us closer to clinical trials and potential bone implants for children living with skull deformations since birth, as well as for veterans seeking to repair damaged limbs.”
According to the NYU researchers, their ceramic 3D printed implant closely resembles the shape and composition of real bone. They contain beta tricalcium phosphate, which is similar to components in natural bone, making the implants resorbable over time. They are coated with dipyridamole, a blood thinning agent that stimulates bone growth and attracts bone cells to the implant.
“Dipyridamole has proven to be key to the implant’s success,” said study co-investigator Bruce N. Cronstein, MD, the Dr. Paul R. Esserman Professor of Medicine at NYU School of Medicine, who also serves as the director of the Clinical and Translational Science Institute, and chief of the Division of Translational Medicine at NYU Langone Health. Dr. Cronstein perfected the drug’s use during device testing. “And because the implant is gradually resorbed, the drug is released a little at a time and locally into the bone, not into the whole body, thereby minimizing risks of abnormal bone growth, bleeding, or other side effects.”
In their most recent experiments, the researchers used the test scaffolds to repair small holes surgically made in the jaws of mice, as well as missing bone pieces as much as 1.2 centimeters long in rabbit limbs and jaws. On average, 77 percent of each scaffold was resorbed by the animals’ bodies six months after implantation. New bone also grows into the lattice-like support structure of the scaffold, which then dissolves. CT scans of the implant sites showed almost no trace of beta tricalcium phospate. Weight-bearing tests also showed that the new bone was the same strength as the original undamaged bone.
Clinical trials are still several years away, according to the researchers, and the next step is to test the patent-pending scaffolds in larger animals.
Authors of the paper include Nick Tovar, Lukasz Witek, Pablo Atria, Michael Sobieraj, Michelle Bowers, Christopher Lopez, Bruce Cronstein and Paulo G. Coelho.
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
[Source: NYU]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Financials: Fathom Struggles in Financial Quicksand During Critical Transition
Facing a year of key transitions and financial pressures, Fathom (Nasdaq: FTHM) has filed its annual report for 2023 with the U.S. Securities and Exchange Commission (SEC). The document outlines...
Latest Earnings Overview for Australian 3D Printing Firms Titomic and AML3D
Australian 3D printing manufacturing firms Titomic (ASX: TTT) and AML3D (ASX: AL3) reported their financial results for the period from July to December 2023, marking the first half of their...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...
3D Printing Financials: Unpacking Farsoon and BLT’s 2023 Performance
In the Chinese 3D printing industry, two companies, Farsoon (SHA: 688433) and Bright Laser Technologies, or BLT (SHA: 688333), have recently unveiled their full-year earnings for 2023. Farsoon reported increases...

































