OPM Licensed for Marketing & Manufacturing 3D Printed Medical Devices in Asia
 Oxford Performance Materials Inc. is a Connecticut-based company we have followed for years as they continue to forge ahead in the realm of 3D printed implants. Their proprietary OsteoFab technology has propelled them into the industry of medical devices in both materials sciences and manufacturing. Now, this will also include the Japanese market as well.
Oxford Performance Materials Inc. is a Connecticut-based company we have followed for years as they continue to forge ahead in the realm of 3D printed implants. Their proprietary OsteoFab technology has propelled them into the industry of medical devices in both materials sciences and manufacturing. Now, this will also include the Japanese market as well.
OPM was just recently accredited as a foreign medical device manufacturer by the Japanese Ministry of Health, Labour, and Welfare. With this international development, they will also be creating OPM Asia in partnership with JSR Corp. of Tokyo. JSR is known for their strength in the life sciences industry in Asia, including a personalized medicine ecosystem, 3D software, drug discovery, and more.
The license will allow the new partners to market and manufacture medical devices in the following countries, with products expected to be on the Asian market by 2019 in:
- Japan
- China
- Taiwan
- Korea
- Brunei Darussalam
- Cambodia
- Lao PDR
- Malaysia
- Myanmar
- Philippines
- Singapore
- Thailand
- Vietnam
- India
“OPM has a broad technology platform built on PEKK polymer,” Scott DeFelice, the CEO and Chairman of Oxford Performance Materials, told 3DPrint.com. “With our establishment of OPM Japan we can now exploit our technology in a highly attractive market.”
“PEKK is a true high-performance polymer with highly unique attributes such as biocompatibility, purity, osteoconductivity, antibacterial properties and of course we have developed a proprietary method to 3D print with it. In our view as a materials company everything starts with having a polymer with the right attributes and PEKK does this uniquely.”
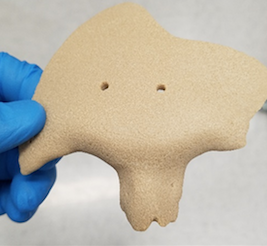
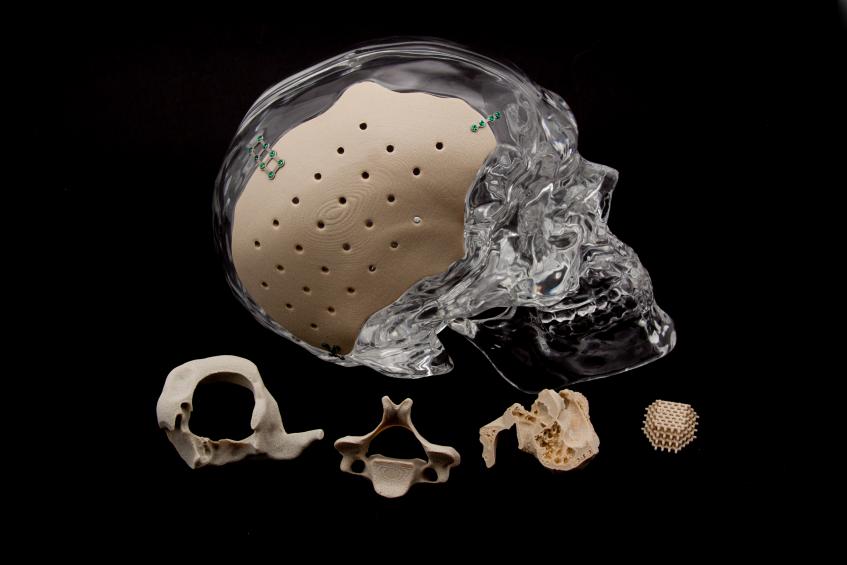
With their ongoing use of PEKK, the OPM team produces implants very similar to bone that are osteoconductive and encourage the growth of natural bone once implanted. It is also radiolucent—meaning that it works well for patients having MRIs as well.
“If a material has the same modulus as bone, it does less damage and is healthier for the bone. For instance, metal can damage the bony interface,” said DeFelice, who will also be OPM Asia’s CEO, as well as one of their board members.
OPM offers comprehensive services around the world, also serving as an OEM and regulatory filer. When PEKK was originally created by DuPont, OPM was approved to make their own OXPEKK material, and since then they have patented OXPEKK LTS for 3D printing medical implants.
“We can go from a proprietary polymer to device to distribution,” continued DeFelice. “It’s sort of what you have to do with something so fundamentally different, something disruptive. In medtech, there’s so much vested cost in development, so it’s really hard to buy in to new materials and processes. You have to do a lot of it yourself.”
“Companies don’t always have the capability and infrastructure to innovate. We have to develop proof and go to companies with a good package.”
With the OPM and JSR partnership in place, they also intend to expand into other areas:
“Independent surgical centers are growing, and we are building a business model around those models,” said DeFelice. “They offer clinical efficacy at a lower cost, which is core to our business model.”
“You’re catching us as we are internationalizing our technology,” he said. “We’ve established a global best-in-class technology that made it through the rigorous U.S. FDA regulatory system. We are already selling implants globally, and now we are ready to expand.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source: MDDI; Images: OPM]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Flashforge Bets on Meshy AI as Desktop 3D Printing Battle Intensifies
Competition in desktop 3D printing is brutal. Whereas before, firms competed through value engineering, Prusa clones now have an integrated hardware, sensor, and software setup that is making all the...
Asia AM Watch: Advantages to the Chinese Way of Doing Business
Timo Göbel, the Head of Additive Manufacturing at the BMW Group, spoke at the AM Forum in Berlin about industrializing additive. He had wise words to share, including that we...
3DPOD 300: Celebrating 300 Episodes with a Look at the Next Year in 3D Printing
In the 300th episode of the 3DPOD, we take a look at what we think will happen over the next 12 months; for instance, what will happen with Bambu’s dominance,...
3D Printing Financials: 3D Systems Returns to Growth in Q1 2026
3D Systems (NYSE: DDD) reported one of its strongest quarters in recent years, showing signs that the company may finally be moving past the tough slowdown that has weighed on...