Organovo Presents Promising Preclinical Data on 3D Bioprinted Human Liver Tissue, Announces Distributor Agreement with Japanese Supplier Cosmo Bio
 California-based biology company Organovo, dedicated to delivering scientific and medical breakthroughs with its innovative and celebrated 3D bioprinting technology, is presenting the first preclinical data on its 3D bioprinted human liver tissue, and the results are promising. The data, being presented at the TERMIS-Americas Meeting in San Diego, shows survival and sustained functionality of the tissue when implanted into animal models. This comes just months after the company announced its plans to develop its 3D bioprinted human liver for transplant. The company, which reported a 209% revenue growth for Q1 FY2016 over Q1 FY2015 thanks to its bioprinted liver tissue, also partnered with Uniquest last year to work on patenting and bioprinting kidney tissue. So it’s exciting to see what will happen next, as ambitious Organovo is bringing us ever closer to the real possibility of functional 3D printed human tissue restoration that appears almost indistinguishable from natural tissue.
California-based biology company Organovo, dedicated to delivering scientific and medical breakthroughs with its innovative and celebrated 3D bioprinting technology, is presenting the first preclinical data on its 3D bioprinted human liver tissue, and the results are promising. The data, being presented at the TERMIS-Americas Meeting in San Diego, shows survival and sustained functionality of the tissue when implanted into animal models. This comes just months after the company announced its plans to develop its 3D bioprinted human liver for transplant. The company, which reported a 209% revenue growth for Q1 FY2016 over Q1 FY2015 thanks to its bioprinted liver tissue, also partnered with Uniquest last year to work on patenting and bioprinting kidney tissue. So it’s exciting to see what will happen next, as ambitious Organovo is bringing us ever closer to the real possibility of functional 3D printed human tissue restoration that appears almost indistinguishable from natural tissue.
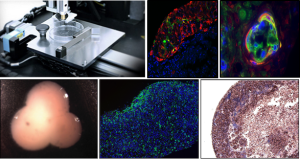 Organovo implanted 3D bioprinted human liver tissue patches, composed of human hepatocytes and select non-parenchymal cells, onto the livers of NOD/SCID (immunodeficient) mice. Scientists and researchers were able to tell that the tissue patches were functioning in the mice through the detection of proteins like human albumin and fibrinogen, as well as protease inhibitor alpha-1-anti-trypsin, in their circulating blood in as early as seven days post-implantation. Microscopic examination and evaluation of the implanted tissue showed that the bioprinted cellular organization was retained for at least 28 days, along with staining for enzymes that are associated with inborn metabolism errors, such as Ornithine Transcarbamylase (OTC) deficiency, which can cause ammonia to accumulate in the blood.
Organovo implanted 3D bioprinted human liver tissue patches, composed of human hepatocytes and select non-parenchymal cells, onto the livers of NOD/SCID (immunodeficient) mice. Scientists and researchers were able to tell that the tissue patches were functioning in the mice through the detection of proteins like human albumin and fibrinogen, as well as protease inhibitor alpha-1-anti-trypsin, in their circulating blood in as early as seven days post-implantation. Microscopic examination and evaluation of the implanted tissue showed that the bioprinted cellular organization was retained for at least 28 days, along with staining for enzymes that are associated with inborn metabolism errors, such as Ornithine Transcarbamylase (OTC) deficiency, which can cause ammonia to accumulate in the blood.
Eric Michael David, M.D., J.D., chief strategy officer, and executive vice president of Organovo’s preclinical development, said, “With a critical shortage of donor organs and few alternatives to transplantation, Organovo is using its 3D bioprinting technology to develop novel therapeutic tissues for direct surgical implantation. Our preclinical data show rapid vascularization and tissue engraftment, and evidence of function and durability of our 3D bioprinted human liver tissue over several weeks. Most importantly, we see evidence of stable production of key human liver proteins in the animal bloodstream, and tissue staining for key human metabolic enzymes. The presence of these enzymes provides an important first step in demonstrating the capability of this tissue to treat inborn errors of metabolism, a key indication we are targeting.”
The bioprinted tissues were able to stay intact on the animal livers, and the mice tolerated them well. Both of these facts support continuing preclinical development of Organovo’s 3D bioprinted liver tissue for therapeutic use, and Organovo plans to submit an Investigational New Drug application to the US Food and Drug Administration (FDA) for its therapeutic liver tissue in a few years. The application will focus on acute-on-chronic liver failure and pediatric inborn errors of metabolism, both of which could could be helped using the therapeutic benefits from a bioprinted liver patch. Organovo will first pursue other opportunities, like breakthrough therapy designation and clinical development outside of the United States, to help accelerate the time to market for its valuable 3D bioprinted tissue. This makes sense, as the total market opportunity for the initial technology in the US exceeds $3 billion.
![]() Speaking of development outside of the US, Organovo also recently announced an exclusive distributor agreement with Cosmo Bio Co., Ltd. for its NovoView Preclinical Services in Japan. Headquartered in Tokyo, Cosmo Bio is one of the country’s largest and most established suppliers in the life science industry, with a vast network of over 200 contracted dealers in Japan.
Speaking of development outside of the US, Organovo also recently announced an exclusive distributor agreement with Cosmo Bio Co., Ltd. for its NovoView Preclinical Services in Japan. Headquartered in Tokyo, Cosmo Bio is one of the country’s largest and most established suppliers in the life science industry, with a vast network of over 200 contracted dealers in Japan.
Organovo’s general manager, Paul Gallant, said, “We’re committed to expanding our customer footprint in Asia, as four of the world’s Top 25 global pharmaceutical companies are located in Japan. Cosmo Bio is an ideal distribution partner for our liver and kidney tissue testing services because of their deep network, strong customer relationships and excellent track record selling the products and services of leading research tool companies.”
This is different than Organovo’s research agreement with Merck Sharp & Dohme Corp. Cosmo Bio was established to sell and distribute biological reagents and services for the purposes of medical research. In order to get important, direct access to customers and other support projects in Japan’s biopharmaceutical industry, Organovo will be working with their Drug Discovery Services team. Discuss in the Organovo forum at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: Velo3D Revenue Up Fueled by Defense Momentum
Velo3D (Nasdaq: VELO) reported a strong start to 2026, with revenue rising as defense and aerospace customers continued shifting from pilot programs into full-scale additive manufacturing (AM) production. The company...
AM & the Military’s Self-Infliction of Rapid Change
I’ve noted before that the additive manufacturing (AM) market for defense has started to evolve so quickly that it’s impossible to even keep track of all the updates in real...
ROBOZE Buys Dimanex Assets to Build “Physical AI” Platform
Dutch firm Dimanex got its start as an MRO platform for the railways. The company had a contract with the Dutch Army in 2018, and later that year signed one...
DMG Mori Joins $10M Defense 3D Printing Program
To look at the Biden administration and the Trump administration that succeeded it and find areas of policy overlap is obviously a bit of a challenge. But such areas certainly...
















































