When it comes to utilizing 3D printing technology to produce medical devices and tools, there is usually a comprehensive trial that these printed materials must undergo before they can be applied in the healthcare sector. These pre-clinical studies help to unearth the benefits and disadvantages of these potential medical solutions, comparing them to other biocompatible materials that are commonly used in similar applications. The medial technology company Stryker is one of the pioneers of using 3D printing technology to enhance the production of medical devices and implants.
When it comes to utilizing 3D printing technology to produce medical devices and tools, there is usually a comprehensive trial that these printed materials must undergo before they can be applied in the healthcare sector. These pre-clinical studies help to unearth the benefits and disadvantages of these potential medical solutions, comparing them to other biocompatible materials that are commonly used in similar applications. The medial technology company Stryker is one of the pioneers of using 3D printing technology to enhance the production of medical devices and implants.
After announcing plans to start construction on a multimillion-dollar 3D printing manufacturing facility earlier this year, Stryker went on to develop their 3D printed Tritanium Posterior Lumbar (PL) Cage Spinal Implant, which was debuted at the American Association of Neurological Surgeons (AANS) Annual Scientific Meeting back in May. Last week, the Spine division of the medical technology company announced the completion of a pre-clinical study for this Tritanium PL Interbody Cage.
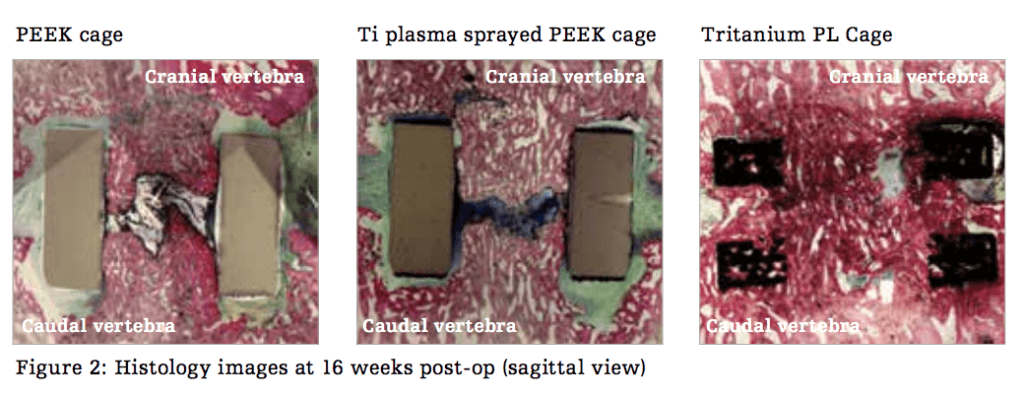
The results of the study compare their highly porous titanium alloy with the biomechanical, radiographic, and histological performance of commonly used implants with different surface technologies in an ovine lumbar interbody fusion model. The materials involved in the comparative study include traditional PEEK cages and plasma-sprayed titanium-coated PEEK cages. Using their proprietary Tritanium Technology, Stryker was able to produce a material with porous structures that resembles spongy bone tissue.
“Stryker’s proprietary Tritanium Technology, a novel, highly porous titanium alloy material designed for bone in-growth and biological fixation, is based on new additive manufacturing techniques pioneered by Stryker over nearly 15 years,” said Bradley Paddock, president of Stryker’s Spine division. “Tritanium, and this important new study, demonstrate Stryker’s commitment to bringing the latest technologies to our customers, and a growing number of surgeons are turning to our new Tritanium PL Cage.”
The pre-clinical study demonstrated that Stryker’s Tritanium PL cages promoted reductions in segmental motion, as well as increases in construct stiffness across all three loading directions between the prepared 8- and 16-week post-implant time points. Neither of these advantages were found when utilizing PEEK and plasma-sprayed titanium-coated PEEK cage groups (p<0.02). In addition, micro–CT and histological data showcased much greater total bone volume within the Tritanium PL Cage at the same 8 and 16 week periods when compared the other treatment groups.
The study, entitled “Biomechanical and Histologic Comparison of a Novel 3D-Printed Porous Titanium Interbody Cage to PEEK”, was recently published in The Spine Journal. Stryker also made the results of this pre-clinical interbody fusion study is also available on their website. The results of this study were presented at the North American Spine Society (NASS) Meeting, which took place in Boston from October 26-29.
All in all, the results show their Tritanium PL cage implant to have superior range of motion, quality bone in-growth profile, and greater construct stiffness when compared to the other cages. Still, these findings have yet to demonstrate this correlation to human clinical outcomes, which will likely be the next step for Stryker to make this implantable material useful to the medical sector. Discuss in the 3D Printed Spinal Implant forum at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Inside The Barnes Global Advisors’ Vision for a Stronger AM Ecosystem
As additive manufacturing (AM) continues to revolutionize the industrial landscape, Pittsburgh-based consultancy The Barnes Global Advisors (TBGA) is helping shape what that future looks like. As the largest independent AM...
Ruggedized: How USMC Innovation Officer Matt Pine Navigates 3D Printing in the Military
Disclaimer: Matt Pine’s views are not the views of the Department of Defense nor the U.S. Marine Corps Throughout this decade thus far, the military’s adoption of additive manufacturing (AM)...
U.S. Congress Calls Out 3D Printing in Proposal for Commercial Reserve Manufacturing Network
Last week, the U.S. House of Representatives’ Appropriations Committee moved the FY 2026 defense bill forward to the House floor. Included in the legislation is a $131 million proposal for...