This week at the American Association of Neurological Surgeons (AANS) Annual Scientific Meeting, which is being held in Chicago until May 4, medical technology company Stryker will debut their newest Tritanium innovation. Stryker’s proprietary Tritanium technology has now been applied to spinal implants designed to encourage healthy regrowth of the bone tissue, and to reduce any strain or damage caused by being implanted. The Tritanium Posterior Lumbar (PL) Cage is a 3D printed intervertebral body fusion device that was developed to help patients dealing with lumbar spinal fixation due to ongoing back problems brought on by degenerative disc disease.
This week at the American Association of Neurological Surgeons (AANS) Annual Scientific Meeting, which is being held in Chicago until May 4, medical technology company Stryker will debut their newest Tritanium innovation. Stryker’s proprietary Tritanium technology has now been applied to spinal implants designed to encourage healthy regrowth of the bone tissue, and to reduce any strain or damage caused by being implanted. The Tritanium Posterior Lumbar (PL) Cage is a 3D printed intervertebral body fusion device that was developed to help patients dealing with lumbar spinal fixation due to ongoing back problems brought on by degenerative disc disease.
Despite its name, degenerative disc disease (DDD) is neither a disease nor is it degenerative in the classic sense. While the condition may be referred to as a disease, it is actually simply a degenerative condition of one or more of the discs in the spine, typically related to simple aging. And despite being classified as degenerative, the condition won’t progress with age – in fact the symptoms actually tend to lessen with age. In this case degenerative is simply a description of the condition of the disc, not a prognosis. That being said, severe or persistent cases of DDD that cannot be solved with medication or physical therapy may need to be corrected surgically.
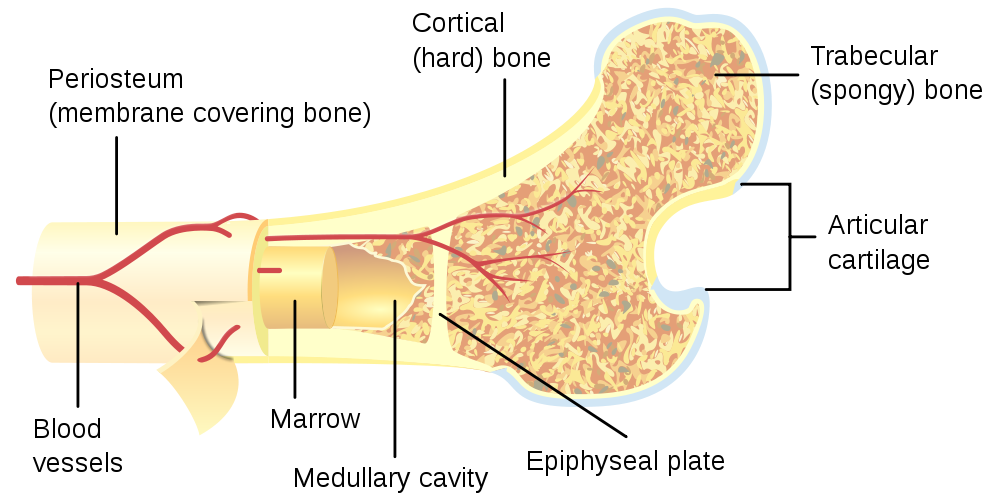
There are actually several surgical options available to those suffering from DDD, and one of the more recently developed is the new Tritanium Posterior Lumbar Cage from Stryker. This implant is made using Stryker Spine Division’s Tritanium technology, which is a proprietary process of 3D printing using a specially developed material. Tritanium is a extremely porous titanium alloy material that will encourage bone in-growth and biological fixation. This is due to the material’s porous internal structures that mimic cancellous bone, or the spongy trabecular bone material found inside of human bones.
“We are pleased to bring this technology advancement to spine surgeons and their patients. Stryker is a pioneer in 3D additive manufacturing, investing nearly 15 years in research and development. Unlike traditional manufacturing techniques, the flexibility of our 3D additive manufacturing capabilities allows us to precisely engineer and produce porous Tritanium devices. The Tritanium PL Cage is an exciting addition to our growing suite of unique spinal products,” Stryker Spine Division President Brad Paddock explained.
The Tritanium Posterior Lumbar Cage is implanted in what is called a posterior approach, which will not damage any muscle tissue around the spine. The cage will be available in several different sizes, widths, lengths, heights, and lordotic angles that will make it easy to adapt to a wide variety of different patients’ anatomies. Also, the geometry contains large lateral windows and open architecture that will allow doctors to easily visualize bone fusion and regrowth on CT scans and X-rays, so recovery can be easily monitored. The design also maximizes the surface area for endplate (the top and bottom parts of the vertebrae) contact with the spinal cage implant, and will minimize any damage to them.
“Spine surgeons need a cage that has the capability of bony integration or bony in-growth, as well as radiolucency so that we can evaluate the fusion long term. Because Tritanium has favorable radiographic capabilities, as well as the integrative surface technology, that really in my opinion is what I would ask for from an interbody cage,” said Dr. Wellington Hsu, M.D., Orthopaedic Surgeon at Northwestern Medical Group.
Stryker’s Tritanium Posterior Lumbar Cage is the just latest medical implant to take advantage of advanced medical-grade 3D printing to create porous geometries that encourage full integration with the human body. The purpose of using porous structures is to attempt to simulate the mechanical properties of bone, while still offering the strength and durability of a medical implant. The implant has received FDA approval and has been cleared for use in the lumbosacral spine. It is expected to be made available to surgeons mid-2016.
Stryker currently develops several Tritanium devices, mostly for knee and joint replacement, however they have also developed skull plates and other less common implants. Stryker offers their medical products to more than one hundred countries, in areas as diverse as Orthopaedics, Medical and Surgical, and Neurotechnology and the Spine. You can find out more about the company and their 3D printed medical devices over on their website. Discuss in the Stryker 3D Printed Titanium Implant forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Precision at the Microscale: UK Researchers Advance Medical Devices with BMF’s 3D Printing Tech
University of Nottingham researchers are using Boston Micro Fabrication‘s (BMF) 3D printing technology to develop medical devices that improve compatibility with human tissue. Funded by a UK grant, this project...
3D Printing Webinar and Event Roundup: April 21, 2024
It’s another busy week of webinars and events, starting with Hannover Messe in Germany and continuing with Metalcasting Congress, Chinaplas, TechBlick’s Innovation Festival, and more. Stratasys continues its advanced training...
3D Printing Webinar and Event Roundup: March 17, 2024
It’s another busy week of webinars and events, including SALMED 2024 and AM Forum in Berlin. Stratasys continues its in-person training and is offering two webinars, ASTM is holding a...
3D Printed Micro Antenna is 15% Smaller and 6X Lighter
Horizon Microtechnologies has achieved success in creating a high-frequency D-Band horn antenna through micro 3D printing. However, this achievement did not rely solely on 3D printing; it involved a combination...