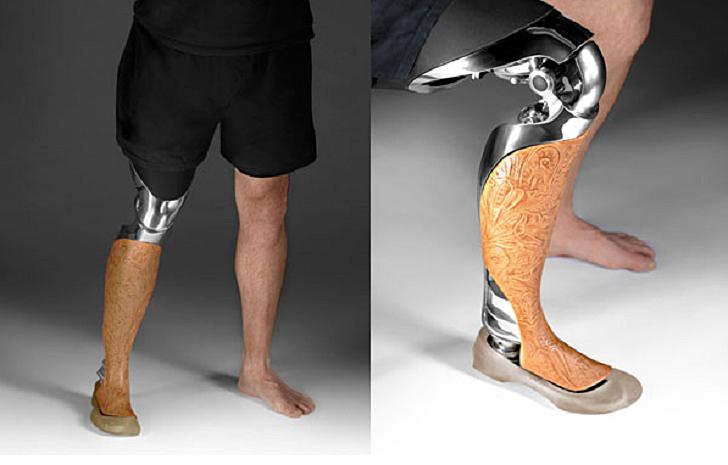
The 3D Printed Medical Devices Market is Expected to Show Steady Gains Through 2026
 According to a new market report from London-based industrial research firm Future Market Insights, the unexpected growth of the 3D printed medical devices market is likely to be a fixture for some time to come. According to the report, medical communities worldwide are adopting 3D printing technology and the use of 3D printed devices at a consistent and rapid pace. The technology is leading to significantly improved quality of care for patients, and is capable of reducing the average procedure time for most surgical applications. This benefits both doctors and hospitals by lowing the strain placed on already overworked staff and leads to lowered costs all around.
According to a new market report from London-based industrial research firm Future Market Insights, the unexpected growth of the 3D printed medical devices market is likely to be a fixture for some time to come. According to the report, medical communities worldwide are adopting 3D printing technology and the use of 3D printed devices at a consistent and rapid pace. The technology is leading to significantly improved quality of care for patients, and is capable of reducing the average procedure time for most surgical applications. This benefits both doctors and hospitals by lowing the strain placed on already overworked staff and leads to lowered costs all around.
The only restraining factor to the growth of the market is the tight regulations on 3D printed devices, implants and tooling. Whenever anything will be used inside of the human body the United States FDA and analogous organizations in other countries stringently review, monitor and test them. They are subjected to a rigorous verification and validation process that insures that they have been designed with consideration to the mechanical and thermal properties and their biocompatibility. However 3D printing is still emergent technology, so these regulations will eventually ease as better guidelines and requirements are formalized, which will only cause the market to expand faster than it already is.
The 3D Printed Medical Devices Market: Global Industry Analysis and Opportunity Assessment 2016-2026 is a comprehensive examination of the complex global market and all of the related products, companies and additive manufacturing processes. Not only does the report include market predictions and forecasts, but it supports it with historical, statistical and industry-validated data. According to the report’s authors, the 3D printed medical devices market is expected to grow at a compounded annual growth rate of 5% year over year until 2026. Additionally, 3D printing technology will also help transform a wide range of industries in addition to healthcare, including aerospace, food and beverages, and the manufacturing industry.
The report includes analysis of individual market segments and dynamics, supply and demand, market trends, market challenges, the various technologies and applications employed, and the major companies driving the industry. It also includes an overview of the parent market, changing market dynamics and a breakdown of geographical regions and any promising growth in those areas. Most regional growth is expected in the United States, which currently has the largest demand for 3D printed medical devices, followed closely by Europe. Combined, both markets are expected to continue to hold more than half of the global market over the next ten years. You can get your copy of the report here. Discuss further in the 3D Printed Medical Devices Future forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
RAPID Roundup 2026: New Machines and Market Moves
RAPID + TCT 2026 wrapped up yesterday, but the show floor proved there’s a lot happening across the additive manufacturing (AM) space, especially when it comes to new hardware and...
Portal Space Systems Raises $50M Series A, Hits $250M Valuation
Portal Space Systems has raised a $50 million Series A round, valuing the company at $250 million. The startup is developing maneuverable spacecraft designed to operate more dynamically in orbit,...
Eplus3D and Hankook Push Metal 3D Printing Into Tire Production
One of the hidden gems in 3D printing is tire production. Several companies, most notably Michelin, have used 3D printing extensively for tire molds and other parts of tire production...
Harvard Engineering Students 3D Print VTOL Drone to Improve Marine Biology Research
With all the current focus on the boom in drones used for national security, it’s easy to forget that the civilian drone market is growing, too. In addition to the...