According to Johns Hopkins researchers, every year due to birth defect, trauma or cancer, more than 200,000 people will need replacement bones in their face or skull. Typically, doctors would remove a part of the patient’s fibula and try to carve it into the required shape and implant the bone back into the patient’s face. While the procedure typically results in the bone regrowing and healing the damage in the face, it isn’t the ideal solution. Depending on the damage being corrected, the bone fragment often can’t be shaped to fit the face very well, which leaves the patient with significant scarring. The removal of part the fibula also creates trauma in the patient’s leg which, when combined with the ongoing trauma in their face or skull, can be quite stressful.
 As detailed in a recently published paper from the associate professor of biomedical engineering at Johns Hopkins University School of Medicine, Warren Grayson, Ph.D. has developed a groundbreaking recipe for natural, 3D printable bone scaffolds. They are made with a combination of man-made, biodegradable plastics and pulverized natural bone material. Using this mix of ingredients, doctors could potentially 3D print replacement bones that could be implanted in the patient’s body. The 3D printed bone would encourage natural regrowth, while slowly allowing the plastic scaffold to be naturally broken down and absorbed by the body. The paper detailing the 3D printable bone material was published online by ACS Biomaterials Science & Engineering.
As detailed in a recently published paper from the associate professor of biomedical engineering at Johns Hopkins University School of Medicine, Warren Grayson, Ph.D. has developed a groundbreaking recipe for natural, 3D printable bone scaffolds. They are made with a combination of man-made, biodegradable plastics and pulverized natural bone material. Using this mix of ingredients, doctors could potentially 3D print replacement bones that could be implanted in the patient’s body. The 3D printed bone would encourage natural regrowth, while slowly allowing the plastic scaffold to be naturally broken down and absorbed by the body. The paper detailing the 3D printable bone material was published online by ACS Biomaterials Science & Engineering.
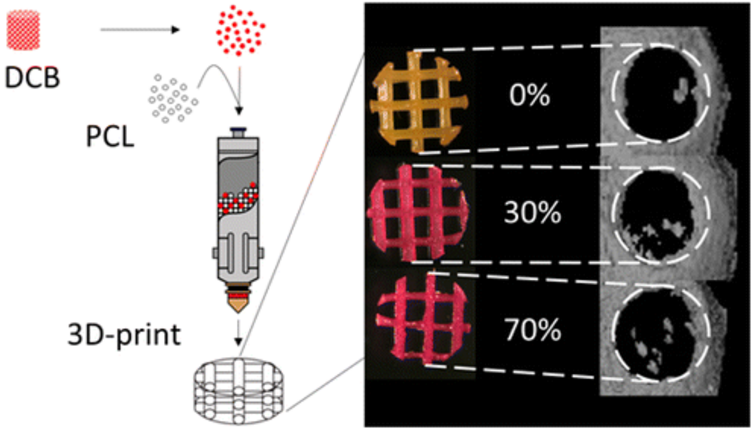
Grayson and his team of researchers created a composite material that combines the durability and 3D printability of a plastic material with the biological “information” that is contained within natural bone. They chose a biodegradable polyester material called polycaprolactone (PCL) that had already received FDA approval for several clinical applications. PCL is a very strong material that has a low melting point and breaks down inside of the body well. Structurally, it would be well suited as a bone replacement, however on its own it doesn’t encourage the regrowth of bone material very effectively. The team mixed it with a natural “bone powder” additive that was made from pulverizing the porous internal structure of cow knees that had been stripped of its cells.

3D printed bone scaffold made of natural bone powder and biodegradable polyester material polycaprolactone.
“Bone powder contains structural proteins native to the body plus pro-bone growth factors that help immature stem cells mature into bone cells. It also adds roughness to the PCL, which helps the cells grip and reinforces the message of the growth factors,” Grayson says.
In order to create the ideal 3D printed bone framework, Grayson experimented with several different mixtures of PCL and the bone powder. The team discovered that the minimum amount of bone powder that would allow the bone to work as intended was at least 30% pulverized natural bone mixed with 70% PCL. The maximum amount of bone powder that could be used was 70% mixed with 30% PCL. The team also attempted a mixture with 85% bone powder, however there wasn’t enough PCL in the mix to properly hold its shape and maintain the necessary lattice structure. Both the 30% mixture and the 70% mixture were tested by introducing them to a nutritional broth material that included fat-derived stem cells harvested from liposuction procedures.
Within three weeks, the cells mixed with 70% bone powder scaffolds displayed gene activity hundreds of times higher in the genes that induce bone formation when compared to cells grown on scaffolds made entirely of PCL. The 30% bone powder scaffolds offered less impressive gene activity, however the bone cells were still showing signs of regrowth. When the team added beta-glycerophosphate to the cell material, it enabled the cell enzymes to begin depositing natural calcium on the scaffolds. When compared to those made using pure PCL, the scaffolds made from 30% bone powder showed a 30% increase in calcium, while those made from 70% bone powder produced twice as much calcium. The team also experimented with implanting bone scaffolds into large holes in rat skulls and discovered that the mice showed at least 50% more bone growth in the scaffolds that contained bone powder than the scaffolds made entirely of PCL.
“In the broth experiments, the 70 percent scaffold encouraged bone formation much better than the 30 percent scaffold, but the 30 percent scaffold is stronger. Since there wasn’t a difference between the two scaffolds in healing the mouse skulls, we are investigating further to figure out which blend is best overall,” Grayson explained.
Grayson and the rest of the Johns Hopkins research team are still experimenting with the ideal bone powder to PCL ratio. While the 70% mixture grew new bone faster, it wasn’t as strong or durable as the 30% mixture, so a trade off is going to have to be made. The team is also going to start experiments with bone powder made from human bone, primarily because it is more commonly available. They also plan to create more natural scaffold structures that closer match the geometries of real bones. And the team is also planning tests for additives that would encourage new blood vessel growth to within the scaffolds, which is necessary for thicker bone replacements to survive. Discuss these new developments further in the 3D Printed Scaffolds forum over at 3DPB.com.
[Source: Science Blog]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Financials: Steakholder Foods Balances Losses and 3D Printed Tech Advances in 2023
A pioneer in the field of 3D printed meat and fish, Steakholder Foods (Nasdaq: STKH) disclosed its 2023 financial results, a year highlighted by technological progress but overshadowed by financial...
3D Printing Financials: Protolabs’ Q1 3D Printing Revenue is Flat, Company Advances in Technology Push
Protolabs (NYSE: PRLB) has kicked off 2024 with a mild boost in revenue, revealing how the Minnesota-based company manages to adapt and thrive even in uncertain market conditions. While the...
Supply Chain Management and the Role of 3D Printing Digital Inventories
As the additive manufacturing (AM) industry grows beyond its humble roots as a rapid prototyping technology, it has been adopted by some of the world’s leading companies to produce not...
3D Printing Financials: Materialise’s Profitability Amid Revenue Dip in Q1 2024
Materialise (Nasdaq: MTLS) has released its first-quarter earnings for 2024, highlighting a challenging quarter with some key advances despite a dip in revenue. While navigating a mixed performance across its...