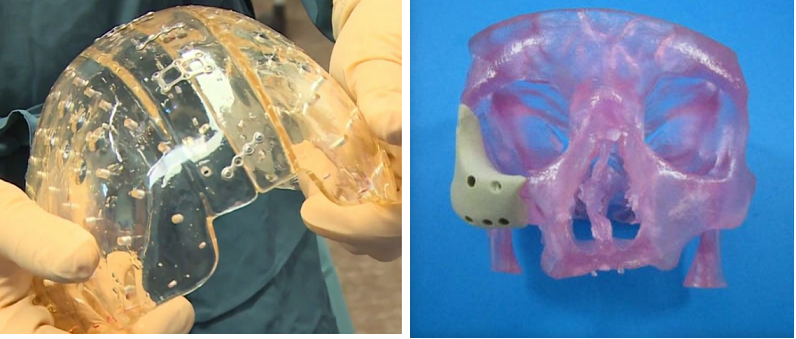
Oxford Performance Materials (OPM) has long been a leader in the area of 3D printing and high performance additive manufacturing. Their accomplishments include the development of a number of advanced materials technologies centered on poly-ether-ketone-ketone (PEKK), a high performance polymer used to deliver functional products to aerospace, industrial, and biomedical markets. In February, OPM became the first company to receive FDA clearance for the manufacturing of patient-specific 3D printed polymeric implants for a line of cranial prosthesis.
Oxford Performance Materials (OPM) has long been a leader in the area of 3D printing and high performance additive manufacturing. Their accomplishments include the development of a number of advanced materials technologies centered on poly-ether-ketone-ketone (PEKK), a high performance polymer used to deliver functional products to aerospace, industrial, and biomedical markets. In February, OPM became the first company to receive FDA clearance for the manufacturing of patient-specific 3D printed polymeric implants for a line of cranial prosthesis.
Building upon this successful track record, OPM announced yesterday they have received the FDA’s 510(k) clearance for its latest implant, the 3D printed OsteoFab® Patient-Specific Facial Device (OPSFD). This makes it the holder of the only FDA cleared 3D printed polymeric implant for facial indications. Scott DeFelice, Chairman and CEO of OPM discussed the significance of this latest development:
“There has been a substantial unmet need in personalized medicine for truly individualized – yet economical – solutions for facial reconstruction, and the FDA’s clearance of OPM’s latest orthopedic implant marks a new era in the standard of care for facial reconstruction. Until now, a technology did not exist that could treat the highly complex anatomy of these demanding cases. With the clearance of our 3D printed facial device, we now have the ability to treat these extremely complex cases in a highly effective and economical way, printing patient-specific maxillofacial implants from individualized MRI or CT digital image files from the surgeon. This is a classic example of a paradigm shift in which technology advances to meet both the patient’s needs and the cost realities of the overall healthcare system.”
 For the printing of the device, OPM Biomedical will utilize the company’s OsteoFab® process. This process is a combination of laser sintering additive manufacturing technology and the proprietary powder formulation OXPEKK. The advantage of this combination is that it creates implants that are mechanically similar to bone and therefore support bone attachment.
For the printing of the device, OPM Biomedical will utilize the company’s OsteoFab® process. This process is a combination of laser sintering additive manufacturing technology and the proprietary powder formulation OXPEKK. The advantage of this combination is that it creates implants that are mechanically similar to bone and therefore support bone attachment.
It is hoped that this technology will also reduce the cost to the patient as a result of reductions in operating room time and the minimization of procedural complications. Severine Zygmont, president of OPM Biomedical shared her excitement about the new implant:
“An exciting aspect of our technology is that additional complexity does not increase manufacturing cost, and having both cranial and facial devices cleared now enables us to answer ever more complex cases where upper facial structures can be incorporated with cranial implants as a single device. As a result, additive manufacturing has the potential to not only improve patient outcomes, but fundamentally improve the economics of orthopedics on a global scale – for developed and developing countries. These are disruptive changes that will allow the industry to provide the finest levels healthcare to more people at a lower cost.”
The OPSFD will be distributed by Biomet, Inc. the same organization that also is the exclusive global distributor of OPM’s OsteoFab Patient-Specific Cranial Device. This could be the first step towards a widespread approval of many devices within the medical field which have been 3D printed. Let’s hear your thoughts in the FDA approval forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Further Understanding of 3D Printing Design at ADDITIV Design World
ADDITIV is back once again! This time, the virtual platform for additive manufacturing will be holding the first-ever edition of ADDITIV Design World on May 23rd from 9:00 AM –...
3D Printer Maker EVO-tech Reborn as NEVO3D — Once More With Feeling
EVO-tech was a 3D printing service and original equipment manufacturer established in 2013 and based in Schörfling am Attersee, Austria. The company produced high-quality material extrusion systems featuring linear bearings,...
3D Systems Brings 3D Printed PEEK Cranial Implant to the U.S. with FDA Clearance
For more than 10 years, 3D Systems (NYSE:DDD) has worked hand-in-hand with surgeons to plan over 150,000 patient-specific cases, and develop more than two million instruments and implants from its...
CDFAM Returns to Berlin for Second Annual Symposium
The second CDFAM Computational Design Symposium is scheduled for May 7-8, 2024, in Berlin, and will convene leading experts in computational design across all scales. Building upon the first event...