 There is tremendous progress being made within the area of 3D bioprinting. In fact, there are companies working to print human organs as we speak, and within the next decade such organs may, if we are lucky, be available for human transplantation. With that said we still are a ways away from such an accomplishment. There are multiple obstacles researchers must first overcome. When considering the organ printing space in general, the printing of complicated vascular networks is the main obstacle currently preventing progress. On the other hand, when printing cellular musculoskeletal tissues the main obstacle in this space is actually the lack of strength inherent in hydrogels.
There is tremendous progress being made within the area of 3D bioprinting. In fact, there are companies working to print human organs as we speak, and within the next decade such organs may, if we are lucky, be available for human transplantation. With that said we still are a ways away from such an accomplishment. There are multiple obstacles researchers must first overcome. When considering the organ printing space in general, the printing of complicated vascular networks is the main obstacle currently preventing progress. On the other hand, when printing cellular musculoskeletal tissues the main obstacle in this space is actually the lack of strength inherent in hydrogels.
Hydrogels currently in use are unable to meet the biological and mechanical requirements for the printing of numerous human tissues. That’s until recently. In a recent article published on Nature Communications on April 28, called ‘Reinforcement of hydrogels using three-dimensionally printed microfibers,‘ researchers at Queensland University of Technology, in Brisbane, Australia, figured out a viable method of creating far more robust and stiff hydrogels.
The researchers — Jetze Visser, Ferry P.W. Melchels, June E. Jeon, Erik M. van Bussel, Laura S. Kimpton, Helen M. Byrne, Wouter J.A. Dhert, Paul D. Dalton, Dietmar W. Hutmacher, and Jos Malda — found that by using a new 3D printed high-porosity microfiber technique, they could increase the stiffness of a hydrogel/scaffold composite by up to 54x, while maintaining an elasticity similar to human tissue.
While nature often uses fiber to strengthen structures, when trying to reproduce such structures in the lab, scientists do not have any way of mimicking this process.
“Such is the case with articular cartilage tissue, which is formed by stiff and strong collagen fibres intertwined within a very weak gel matrix of proteoglycans,” explained Professor Dietmar W. Hutmacher, from QUT’s Institute of Health and Biomedical Innovation. “By bringing this natural design perspective of fibre reinforcement into the field of tissue engineering (TE), we can learn a lot about how to choose an effective combination of matrix and reinforcement structure in order to achieve composite materials with enhanced mechanical properties for engineering body parts.”
In order to successfully harvest human cells, the hydrogel needs to act as a fully hydrated 3D environment which is “comparable to the extracellular matrix of the native tissue,” so that when cells are embedded they are able to repair and regenerate the damaged tissue. The typical hydrogels which are currently being used are oftentimes too weak, or do not present the most conducive environments for the cellular growth.
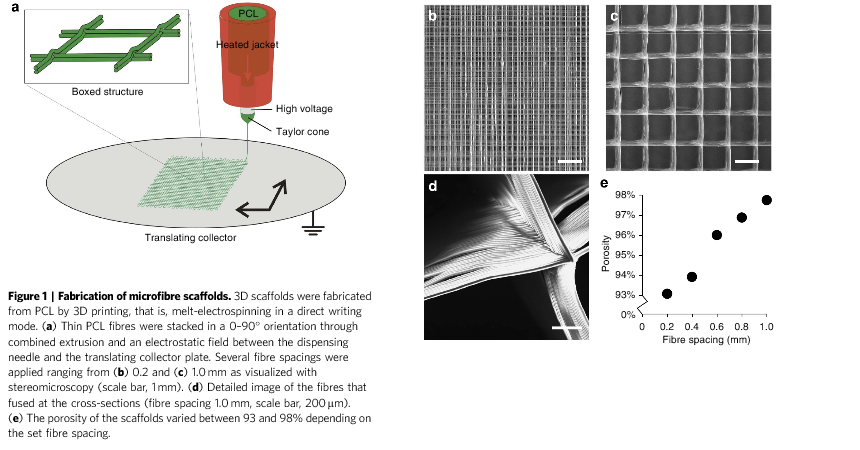
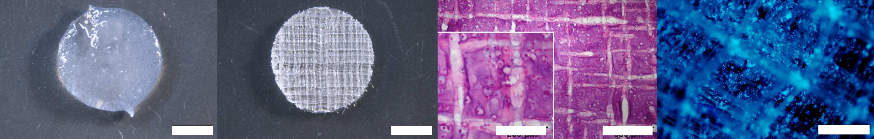
Using a technique called melt electrospinning direct writing (a form of 3D printing which stacks layers of fibers in a given orientation), researchers have been able to reinforce gelatin metha-crylamide (GelMA) hydrogels with poly(e-caprolactone) (PCL) fiber scaffolds, which have a very high porosity. GelMA, which is currently a hydrogel being put to use within many medical fields, because it allows for matrix deposition of cells, is also rather weak, making it not ideal for bone or musculoskeletal tissue. By reinforcing the GelMA with the PCL, however, fibrous hydrogel composites that are incredibly stiff, resembling that of articular cartilage, or the cartilage often found on the surface of bones, can be created. This composite material also is able to remain relatively elastic.
“We found that the stiffness of the gel/scaffold composites increased synergistically up to 54 times, compared with hydrogels or microfiber scaffolds alone,” explained Hutmacher. “Computational modelling has shown that we can use these 3D-printed microfibres in different hydrogels and a large range of tissue engineering applications.”
 By utilizing this melt-electrospinning writing 3D printing process, researchers were able to construct the PCL fibers using micrometer-scale fibers, which would have been impossible to create using other extrusion methods of 3D printing. The filament diameter was able to be as small as 5 microns, which is 1/20th the size of filament used with traditional extrusion processes on FDM/FFF 3D printers today.
By utilizing this melt-electrospinning writing 3D printing process, researchers were able to construct the PCL fibers using micrometer-scale fibers, which would have been impossible to create using other extrusion methods of 3D printing. The filament diameter was able to be as small as 5 microns, which is 1/20th the size of filament used with traditional extrusion processes on FDM/FFF 3D printers today.
In conclusion, this research will likely lead to new approaches to bioprinting numerous human tissues, especially that of cartilage and other musculoskeletal tissues. When human chondrocytes cells (healthy cartilage cells) were encapsulated in the GelMA/PCL composites they were found to be much more responsive to mechanical loading, “which led to significant changes in gene expression in vitro.”
It will be interesting to see what additional progress is made within this area of research, and just how the 3D bioprinting of human tissue may be influenced. Let’s hear your thoughts on this research in the 3D Printed Hydrogel Composite forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Making 3D Printing Personal: How Faraz Faruqi Is Rethinking Digital Design at MIT CSAIL
What if your 3D printer could think more like an intelligent assistant, able to reason through a design idea, ask questions, and deliver something that works exactly the way the...
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Heating Up: 3D Systems’ Scott Green Discusses 3D Printing’s Potential in the Data Center Industry
The relentless rise of NVIDIA, the steadily increasing pledges of major private and public investments in national infrastructure projects around the world, and the general cultural obsession with AI have...
Formlabs Teams Up with DMG MORI in Japan
In late June, Nick Graham, Chief Revenue Officer at Formlabs, announced on LinkedIn that the company had partnered with DMG MORI, one of the world’s leading machine tool companies, to...


































