New EU and US Regulations Greenlight the Refinement of Global Non-Animal Testing Strategies – AMS Speaker Spotlight

The Modernization Act 2.0, set in motion by the FDA and signed by President Joe Biden, together with the Directive 2010/63/EU regulations, increases awareness about the use of animal testing in research and preclinical studies, aiming to exhaustively reduce all animal research with non-animal testing strategies.
Grasping the scope of non-animal testing strategies
Animal testing has been the gold-standard in scientific research and is vastly integral for the development, production, and quality control of biologicals such as vaccines, enzymes, and hormones (1). Conversely, they constitute copious drawbacks in their application for in vitro models of cell systems, including high economic expenses, ethical concerns related to bestial discomfort and distress, and uncontrollable variables that result in experimental inaccuracy. In June 2021, the US Food and Drug Administration (FDA) published proposals for the New Alternative Methods Program that revolves around replacing, reducing, and refining (3Rs) the use of laboratory animals through the utilization of cutting-edge alternative techniques.
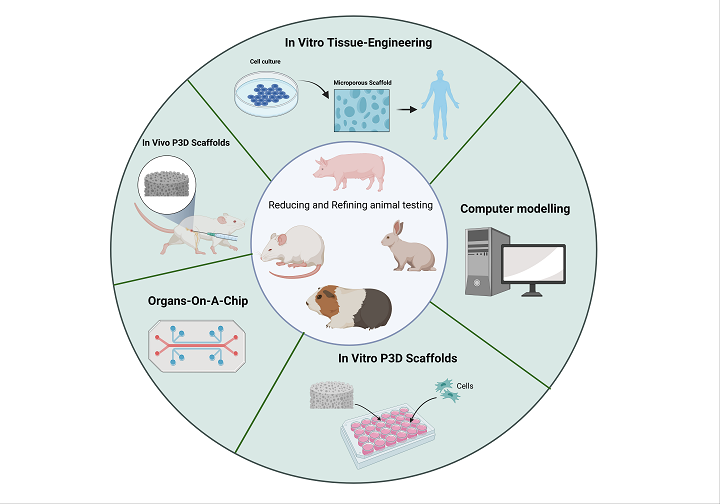
Figure 1. A wide panel of existing techniques aimed at reducing and refining the use of animals in preclinical studies.
Innovative technologies, such as the introduction of microphysiological systems (MPS), establishes in vitro models that recapitulate the highly dynamic microenvironment of living tissues and organs. The purpose of the MPS is to taper the gap between in vitro and in vivo models, hence refining the translation from preclinical animal studies to clinical trials. Conversely, despite the ignition of interest in this area in recent years, and the evident rewards for developing such models that could reduce and refine animal testing in the clinic, the full potential of this technology remains unexplored within the global market (2).

Figure 2. P3D Scaffolds produced using Ossiform’s technology to 3D print bioceramics made of beta-tricalcium phosphate. The P3D Scaffolds allow researchers to study different aspects of bone development and disease in a structurally and physiologically relevant setup.
Unveiling the extensive applications of Ossiform’s technology
With current applications within 3D cell culturing and MPS, spanning from hydrogels to printed organs, flow dynamic chambers, and organs-on-chips, one might easily assume that most of the basics are covered by now. Yet, one important tissue appears to have been forgotten – the mineralized part of our bones!
Currently, very few systems provide researchers with a stiff, porous, tricalcium phosphate-rich, and bone mimicking 3D cell culture system, leaving researchers with the options of traditional cell culturing, animal derived bone discs, or various types of polymer-based scaffolding techniques.
Ossiform’s technology is used to produce P3D Scaffolds; a natural bioceramic 3D printed structure designed to provide a clinically relevant cell culture system based on natural materials and customized structures, that enable the formation of predictive non-animal research models. Collectively, an MPS should be viewed as an estimation of reality and not as an accurate reconstruction. Typical flaws of static, one-dimensional cell cultures, such as the inability for open-surface cultures to yield significant interstitial flow and the associated signaling cascade, highlights the appetite for realistic cell cultures grown in 3D cell culture systems with tissue-like perfusion and proper dynamic cues, which is indeed provided by the P3D Scaffolds. P3D Scaffolds are highly applicable for both in vitro– and in vivo preclinical research, 2D analysis methods, and maintain relevant cell signaling, which underlines their potential for establishing an efficient MPS.

Figure 3. Confocal microscopy image of bone-forming cells (osteoblasts, blue) and their extracellular matrix (green) cultured on P3D Scaffolds. Due to the bone mimicking environment, the osteoblasts maintain their normal properties, and initial bone formation is observed.
Although innovative strategies for refining animal testing are blooming, they are relatively unprecedented and the global market size was valued at about $1.110 billion in 2019 according to The Business Research Company. With the rise in initiatives to refine and reduce animal testing, the market is expected to grow at a compound annual growth rate of 10.40% during 2019-2025, which further emphasizes the haste for developing non-animal testing strategies for preclinical studies.
By entering this emerging market, Ossiform turns its technology full circle by providing and developing products for all aspects of bone-related diseases and the treatment hereof – from fundamental research and preclinical studies to clinical use in operating theaters across the globe.
Based on Ossiform’s journey throughout the years, having been engaged with additive manufacturing in healthcare since 2014, Casper Slots, COO at Ossiform, will speak about the emergence and commercialization of new bioprinting technologies in Session 1, Panel 1: Bioprinting: New Technology and Commercialization, on February 8th at Additive Manufacturing Strategies 2023 in New York City. Register for your ticket to attend here.
About Ossiform:
Ossiform uses proprietary technology to produce patient specific, natural, and resorbable bone implants with a mission to reduce complications, improve functional and aesthetic outcomes, and obtain faster recovery. The technology is further used to provide a product line of 3D printed research products for enhancing bone and cancer research. The bone-like scaffolds provide a clinically relevant cell culture system of natural materials and customized structures that enable the creation of predictive research models of human physiology and pathology.
Website: Ossiform – We Print Bone™
References
- Lilley E, Isbrucker R, Ragan I, Holmes A. Integrating 3Rs approaches in WHO guidelines for the batch release testing of biologicals. Biologicals. 2021 Nov;74:24-27.
- Peel S, Jackman M. Imaging microphysiological systems: a review. Am J Physiol Cell Physiol. 2021 May 1;320(5):C669-C680.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: 3D Systems Returns to Growth in Q1 2026
3D Systems (NYSE: DDD) reported one of its strongest quarters in recent years, showing signs that the company may finally be moving past the tough slowdown that has weighed on...
3D People Case Study Details Development of 3D Printed POV Camera Rig
A POV, or Point of View, camera rig, is a wearable support system that helps filmographers capture first-person footage, making the images more immersive. Some good examples of movies shot...
3D Printing News Briefs, May 2, 2026: Soft Robots, Agricultural Waste, & More
In this weekend’s 3D Printing News Briefs, we’ll start off with a multi-laser metal powder bed fusion 3D printer and post-processing news. We’ll end with research into soft robotics and...
Industrial Applications on Display at RAPID 2026: CERATIZIT & 3D Systems
Applications are where it’s at in the additive manufacturing (AM) industry. At the recent RAPID+TCT in Boston, I met with a few companies to learn about some of their very...















































