
ROKIT Healthcare, in cooperation with a Massachusetts-based U.S. hospital, has completed pre-clinical studies for cartilage regeneration using the 3D bioprinting technology and is in the process of clinical studies with patients of age-related osteoarthritis in Egypt.
The pre-clinical study, performed together with the Charles Bragdon group at Massachusetts General Hospital, lasted about six months and followed up with the progress of hyaline cartilage regeneration in beagles.
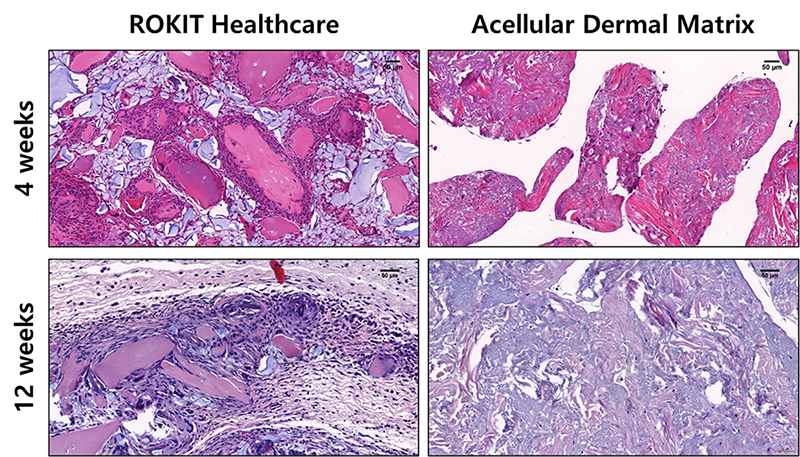
H&E staining result shows a cluster of cells, surrounding the particle which gradually induce regeneration of tissue. Unlike the comparison group, cells gradually surround the particles of ROKIT bio filler and induce regeneration of tissue.
Existing cartilage repair therapies, based on stem cell injections and seeding and culturing of cells onto alternative membranes, have limitations in treating cartilage which has complex 3D structures and compositions. Combining 3D bioprinting with optimized bioinks composed of minced cartilage and autologous solutions from the patient, ROKIT Healthcare seeks to offer a novel therapy that is founded on providing a stable environment for the recruitment of endogenous stem cells and the differentiation of 3D structurally positioned stem cells into chondrocytes in vivo.
“There has yet to be any truly regenerative method for cartilage repair despite osteoarthritis being one of the top public health burdens in this era of aging populations,” said Seok Hwan You, CEO of ROKIT Healthcare. ”Recently on the news there have been reports of recalls of artificial knee cartilage replacements, including products that have for long comprised the majority of cartilage repair markets. This testifies there are intrinsic limitations of synthetic and allogenic tissue engineering methods, and we need to find autologous alternatives that rely on bioprinting of autologous solutions to the defect site and creation of an endogenous regeneration niche. All of this needs to be done in the safest way that minimizes risk and long-term treatments.”
ROKIT Healthcare has completed applying for domestic and international patents for printing the cartilage regenerative 3D constructs using the MRI/CT scan data of the patient and inserting them back into the patient knee while minimizing the incision. Following the initial rounds of pre-clinical study in the U.S. and clinical study in Egypt, the clinical studies are planned to continue in Korea and Singapore.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Making 3D Printing Personal: How Faraz Faruqi Is Rethinking Digital Design at MIT CSAIL
What if your 3D printer could think more like an intelligent assistant, able to reason through a design idea, ask questions, and deliver something that works exactly the way the...
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Heating Up: 3D Systems’ Scott Green Discusses 3D Printing’s Potential in the Data Center Industry
The relentless rise of NVIDIA, the steadily increasing pledges of major private and public investments in national infrastructure projects around the world, and the general cultural obsession with AI have...
Formlabs Teams Up with DMG MORI in Japan
In late June, Nick Graham, Chief Revenue Officer at Formlabs, announced on LinkedIn that the company had partnered with DMG MORI, one of the world’s leading machine tool companies, to...

































