Chuck Hull on 3D Systems’ Journey into Bioprinting
After 35 years of creating innovative 3D printing technologies, pioneering company 3D Systems (NYSE: DDD) is expanding its bioprinting efforts. Following a successful collaboration with United Therapeutics Corporation dedicated to developing 3D printing systems for solid-organ scaffolds, 3D Systems intends to invest, develop, and commercialize targeted applications for regenerative medicine for non-solid organ applications.
Co-founded by Charles “Chuck” Hull, known as the father of 3D printing, 3D Systems has grown into a global AM solutions company with a strong foothold in many industries. This includes healthcare, where it has delivered solutions for more than 35 years. 3D Systems’ expertise in 3D printing in healthcare has helped make the expansion into bioprinting an intuitive next step for the company.
Hull’s team and United Therapeutics’ organ manufacturing and transplantation-focused subsidiary, Lung Biotechnology PBC, have made significant progress toward developing a next-generation AM platform solution for lung scaffolds capable of full size, vascularized, rapid, micron-level printing. The ultimate goal is to address the acute national shortage of transplantable lungs and other organs with various technologies that either delay the need for such organs or expand the supply.
Focusing on future commercialization efforts and expanding into the bioprinting space, Hull shared some of his expectations about 3D Systems’ new undertaking. Currently serving as Chief Technology Officer for the company, the 81-year old inventor is laying down the foundations to help patients in critical need, and he is using 3D technology to do it.
“This is certainly a big step for our company. We have been following bioprinting for years and it has been characteristically a research applications-space, not yet focused on commercial healthcare solutions,” Hull told 3DPrint.com. “That said, 3D Systems has been focused on building a sizeable healthcare business, and I believe we all envisioned sometime in the future that we would get directly involved in bioprinting. It is still a somewhat nascent industry, but the decision was: ‘when do we start?’ And now that we have, the question becomes: `when will the sector make the transition between where it is now and the larger reality of what it could be?’ We don’t see bioprinting as a big part of 3D Systems for a while because all these applications take time to develop, mature, and go through a regulatory path. Still, we knew that we had to begin developing this technology.”
Although the timeline for an actual transplantable human lung may be years away, Hull says we may see many opportunities for a range of tissue applications that can come out of the technology much quicker, including laboratory testing methods that will accelerate the development of new drug therapies.

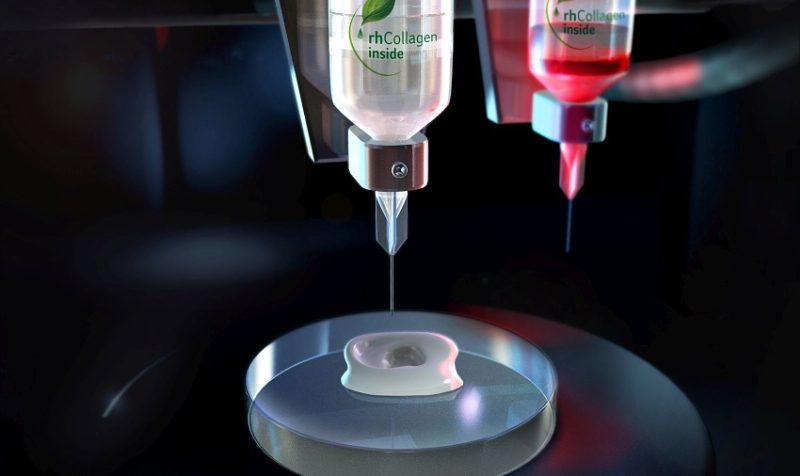
Human vasculature model created using Print to Perfusion process. Image courtesy of United Therapeutics.
The partnership with Lung Biotechnology has been making breakthrough advances in lung modeling, 3D printing, material formulation, and material handling to yield significant capabilities in bioprinters and biomaterials for lung production. However, 3D System’s journey into bioprinting began over six years ago. Hull remembers when United Therapeutics CEO Martine Rothblatt discussed with him the vision of 3D printing lung scaffolds for producing lungs for implants, which are in high demand. “That wasn’t possible,” thought Hull at the time, “It’s too difficult a job for 3D printing.” However, after researching the technology, Hull decided it could be doable.
“Not that Lung Biotechnology is ready to launch lung production, but we are on that path. We learned what it takes to support life. 3D Systems has developed the printing capability, and has helped develop the materials, and software to make this possible. We can provide the vascularization path for the complex network of airways and blood paths. We can print fully vascularized hydrogel scaffolds so that the cells have blood and nutrient supply,” said Hull. The newly developed Print to Perfusion process enables 3D printing of high-resolution scaffolds, which can be perfused with living cells to create tissues.
To advance these efforts, 3D Systems is expanding its high-speed Figure 4 technology through innovation tailored to bioprinting and regenerative medicine. Building upon these capabilities, the company will be able to make advancements into numerous applications within the human body in collaboration with its partners. This includes the potential to enable novel laboratory testing methods to accelerate the development of new drug therapies while reducing the need for animal testing.
Eager to collaborate with other companies, 3D Systems is working on providing a complete solution for regenerative medicine researchers. Hull envisions tissues will be a powerful medical device segment for better healthcare in the future. However, there are still hurdles to overcome: “The 3D printing part is easy, the cell biology is the hard part.”
3D Systems is leveraging bioprinting technology so that researchers can move the field forward. Building on the progress the company has made to date, 3D Systems is infusing additional resources into its regenerative medicine R&D efforts to accelerate development programs that expand on the scope of potential applications. 3D Systems previously announced collaborations with CollPlant Biotechnologies and Antleron that expanded its capabilities in regenerative medicine. Hull also anticipated that we could expect to hear about more partnerships throughout 2021, as it broadens its portfolio of collaborations.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: Velo3D Revenue Up Fueled by Defense Momentum
Velo3D (Nasdaq: VELO) reported a strong start to 2026, with revenue rising as defense and aerospace customers continued shifting from pilot programs into full-scale additive manufacturing (AM) production. The company...
AM & the Military’s Self-Infliction of Rapid Change
I’ve noted before that the additive manufacturing (AM) market for defense has started to evolve so quickly that it’s impossible to even keep track of all the updates in real...
ROBOZE Buys Dimanex Assets to Build “Physical AI” Platform
Dutch firm Dimanex got its start as an MRO platform for the railways. The company had a contract with the Dutch Army in 2018, and later that year signed one...
DMG Mori Joins $10M Defense 3D Printing Program
To look at the Biden administration and the Trump administration that succeeded it and find areas of policy overlap is obviously a bit of a challenge. But such areas certainly...