The Digital Manufacturing Centre (DMC), a 3D printing service bureau, has introduced to the U.K. a process called silicone additive manufacturing (SAM). The technology, developed by a Swiss company called Spectroplast, is targeted toward the automotive, industrial, and medical sectors.
High-quality elastomeric parts are still somewhat rare in 3D printing, when compared to rigid materials. This is due to the limited range of elastomers that can be processed by the various 3D printing technologies. In extrusion 3D printing, thermoplastic polyurethane (TPU) is the go-to material. The same is true for powder bed fusion, which is even more limited in the varieties of TPU that can be printed.

A 3D printed silicone industrial part made using Spectroplast’s Silicone Additive Manufacturing technology. Image courtesy of Spectroplast.
As a thermoset, silicone is even less common in the industry, but exhibits physical properties that aren’t available with TPU and other thermoplastic elastomers. In particular, its resistance to chemicals, a wide range of temperatures, and ultraviolet light are beneficial for certain applications, such as sterilizable medical equipment.
Spectroplast is a spin-off of ETH Zurich, the renowned Swiss public research university. The exact nature of its SAM process has not been disclosed, but the firm describes it as related to stereolithography and digital light processing. In a press release, DMC did say, “SAM works by selectively exposing silicone to a light source, forming very thin solid layers that build up to create the final object.”
Additionally, the parts made with SAM are described as being isotropic, meaning that they exhibit uniform physical properties in every direction, unlike the majority of 3D printed parts. This would suggest that SAM involves a form of continuous DLP.
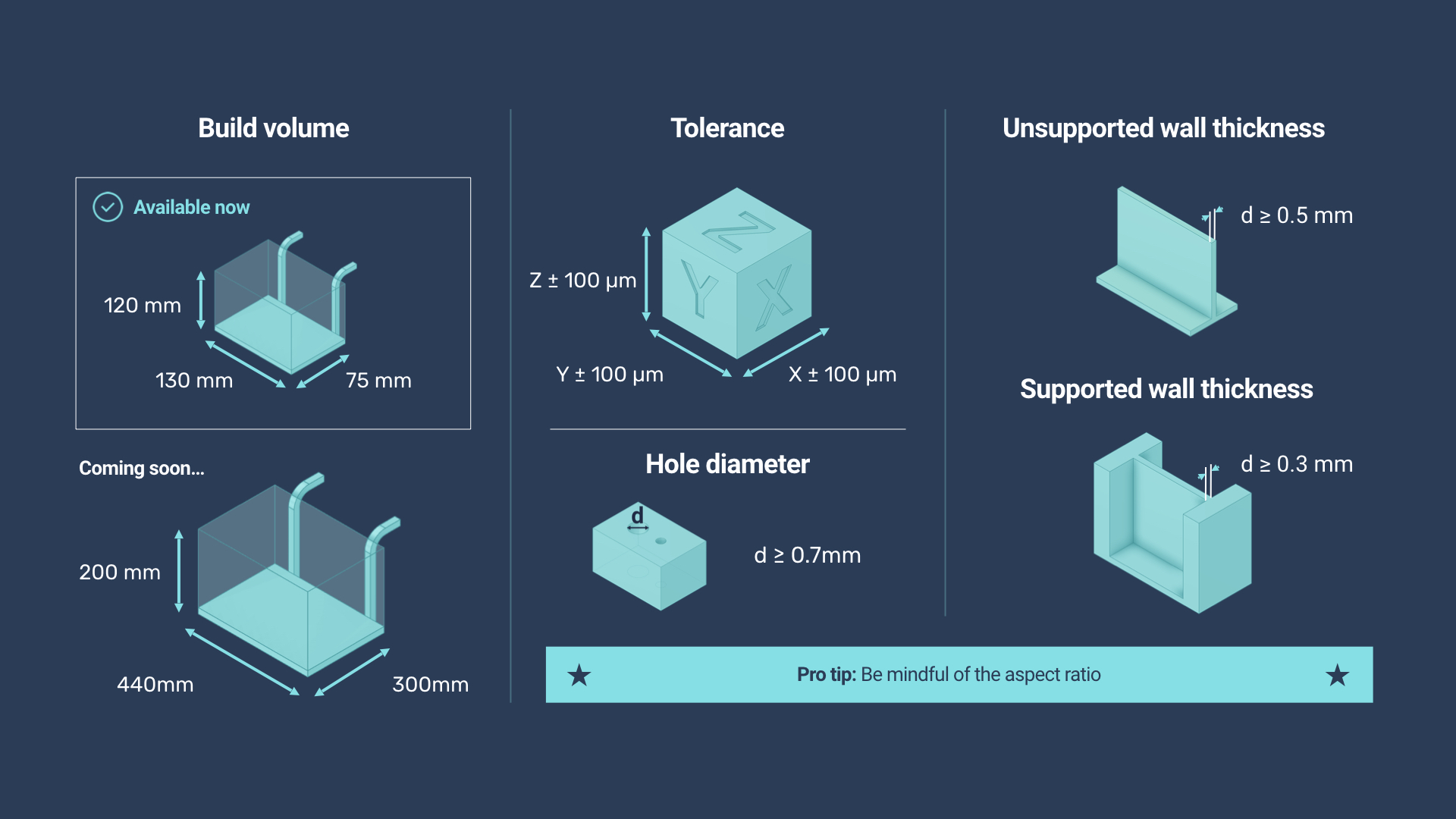
So far, the company offers four forms of silicone with shore hardness of A20, A35, A50 and A60, respectively. All four are considered biocompatible and meet ISO DIN EN10993-05/10 standards. This signifies that is not only safe for skin contact but is implantable, opening it up for use in medical devices, dental solutions, prosthetics and more. With temperature resistance in the – 50 to 200 °C range, items made with SAM can serve as gaskets, seals, grommets, dampers, and tubing for industrial applications. Achievable layer thickness at the moment is 0.1 mm and the build volume of the current platform is 120 x 130 x 75 mm, but Spectroplast is working to expand this to 200 x 440 x 300 mm.
DMC will now bring the technology to service the U.K. As a spin-out of engineering firm KW Special Projects, the DMC features a 2,000 sqm production facility that, starting in Q1 of this year, offers 3D printing services in the region. The company will provide 3D printing services, including serial production parts.
It’s worth noting that, though DMC is claiming to be the first to offer silicone 3D printing in the U.K., Picsima did conceive of a method for 3D printing silicone in roughly 2014. Unfortunately, the company announced that it would be selling its patents in 2020 as the founder began a teaching career.
Meanwhile, multinational silicone company Wacker Chemie developed its own method for 3D printing silicone. Using an inkjet technology, Wacker’s ACEO 3D printing service continues to operate. It seems as though it is possible for customers in the U.K. to order parts from the service, but how it compares to what Spectroplast and DMC are offering is limited.
ACEO allows for the printing of parts up to 200 cm3. It does seem as though it only offers one type of material, with a shore of A 50. Medical device parts are possible with ISO10993 compliance up to Class 2a. Aside from that, it’s difficult to know how the two processes compare.
Now that silicone 3D printing has multiple players involved, maybe it’s time for Picsima to get back into the game?
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Johns Hopkins University Researchers Develop HyFAM Technology
Two scientists from Johns Hopkins University, Nathan C. Brown and Jochen Mueller, have developed a hybrid manufacturing technology they call HyFam, or Hybrid Formative Additive Manufacturing. Their work on this technology...
3D Printing G-Code Gets an Upgrade: T-Code
Good old G-Code still manages many 3D printers, great and small. Just like the STL, it’s a standard that enables collaboration while also holding the additive manufacturing (AM) industry back....
AM Rewind: The Biggest News and Trends of 2024
After a sluggish 2023, driven by persistent inflation and geopolitical tensions, 2024 has seen some recovery. Economic growth climbed from about 2.8 percent in 2023 to a modest 3.2 percent...
Metal Wire 3D Printer OEM ValCUN Announces Plans for 2025 Expansion
ValCUN, a Belgian original equipment manufacturer (OEM) of wire-based metal additive manufacturing (AM) hardware, has announced that the company has entered the next phase of its growth trajectory, making key...