In this week’s 3D Printing News Briefs, we’re talking about a strategic alliance, a software launch, and FDA clearance. Sigma Labs and IN4.OS are teaming up for Factories of the Future, Raise3D is introducing the latest version of its slicing software, and PrinterPrezz has received 510(k) clearance for its new family of 3D printed spinal devices.
Sigma Labs Partnering with IN4.OS
In-process quality assurance software provider for metal 3D printing Sigma Labs (NASDAQ: SGLB) has announced a strategic alliance with Industry 4.0-focused advanced manufacturing leader IN4.OS to ensure a high standard of quality during AM by building Smart Factories of the Future, a cyber-physical system that can deliver scalable manufacturing regardless of location. Sigma Labs will bring its PrintRite3D IPQA software to ensure high quality of 3D printed mission critical parts for high technology sectors, such as aerospace, life sciences, and defense. IN4.OS will use DMG Mori’s Lasertec metal 3D printers, certified as PrintRite3D Ready, to produce these parts.
“IN4.OS’s concept of Smart Factories of the Future is a timely and courageous response to increasing the country’s self-sufficiency in times when supply chains are disrupted. We are excited to work with IN4.OS and DMG MORI, as they take on this important initiative,” said Mark K. Ruport, the President and CEO of Sigma Labs.
Raise3D Launches ideaMaker 4.0
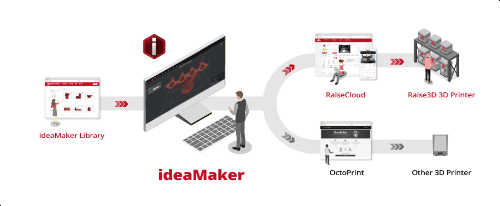
Just months after introducing its new ideaMaker Library platform, global 3D printer manufacturer Raise3D, which is also committed to software development, has announced that it’s launching the latest version of its slicing software, ideaMaker 4.0. This update is able to integrate better with ideaMaker Library, as well as RaiseCloud Print Management software and Octoprint, so third party printers can also benefit: users can easily download and share slicing templates on the library platform, upload gCode to Octoprint, and monitor the printing process remotely with RaiseCloud.
The updated software allows users to optimize models by customizing the settings of one section, selected layers, or a specific shape inside of an STL model, which reduces both print time and waste material. Additional features of ideaMaker 4.0 include a “Light Theme” option under Preferences, an “array” function to distribute models in specific ways, functions to modify a model by removing or keeping overlap regions, and some other helpful functions, such as “minimal part size” and “spiral vase mode path interpolation.” The new ideaMaker 4.0 will be available in English, French, German, Italian, Polish, Russian, and Spanish.
PrinterPrezz Announces 510(k) Clearance for Spinal Implants
California-based company PrinterPrezz, which is making waves in medical polymer and metal 3D printing, announced that it has received 510(k) clearance from the FDA for its GAIA Lumbar Interbody Fusion Device (LIFD) family of 3D printed spinal implants. The first implants in this platform included in this announcement include the Astraeus ALIF, Themis TPLIF, and Prometheus PLIF lumbar interbody systems, which are 3D printed out of titanium (Ti6Al4V-ELI) using Laser Powder Bed Fusion technology. This clearance is a reference platform for future medical device innovators, though market introduction plans for the GAIA implant platform have not been finalized just yet.
“The products announced today represent our cumulative learnings in medical device design, additive manufacturing processes, non-active implantable medical device (NAIMD) testing, cleaning, packaging & sterilization, and quality management. What makes GAIA™ a true reference platform, and PrinterPrezz a true medical device foundry, is that our customers have access to the array of technologies developed and validated during the GAIA™ program for their own product lines. Experienced customers benefit from our 3D print and design optimizations that help reduce manufacturing costs, while small, and mid-size customers may find benefit leveraging our FDA documentation,” explained Shri Shetty, the CEO of PrinterPrezz.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Divergent & GA-ASI: How Connecting at AMUG Led to an Important Industry Partnership
As I mentioned in my AMUG 2026 overview, I had a lot of delays traveling to Reno, Nevada to attend the conference. So unfortunately, I missed “From Hypercars to Defense...
BLT Touts 100,000 Copper Parts Made
With the significant volume of copper used in electronics, semiconductors, aerospace, defense, and beyond, copper additive has had immense promise from the early days of copper 3D printing at Beamit...
TCT Asia 2026: Empowering Asia · Connecting the World
TCT Asia, now in its 12th year, is more than just a 3D printing exhibition — it embodies its original concept: Time Compression Technology, a vision of making the entire...
Pete Pharma Deal with Atrium24 Signals Path for Broader 3D Printed Drug Commercialization
While I would guess we’re still probably at least several years away from 3D printed drugs being a commonplace reality, the growing number of companies with proprietary additive manufacturing (AM)...