Researchers at the Swiss Federal Institute of Technology Lausanne (EPFL) have developed an approach to print centimeter-scale tissues with high physiological relevance, that look and function almost like their full-sized in-vivo counterpart. The engineered mini-tissues could allow scientists to study biological processes and even test new treatment approaches in ways that were previously not possible, opening new avenues for drug discovery, diagnostics, and regenerative medicine.
In a study published in September 2020 in the journal Nature Materials, the researchers reported the design of a customized extrusion-mode bioprinting set-up consisting of a microscope and a device that can aspirate and deposit cells through a thin nozzle coupled to a syringe pump. The approach, called bioprinting-assisted tissue emergence (BATE), uses organoid-forming stem cells as building blocks that can be deposited directly into extracellular matrices conducive to spontaneous self-organization. By moving the microscope stage and monitoring constantly, the process through the microscope lens, the researchers were able to deposit into the gel a line of intestinal stem cells that measured a few centimeters in length.
To date, researchers have been able to engineer organoids that resemble the brain, stomach, kidney, lung, and liver, facilitating the development of new therapeutic strategies as well as the advancement of personalized medicine. However, EPFL reported that conventional approaches to growing organoids result in stem cells assembling into micro- to millimeter-sized, hollow spheres.
Furthermore, according to Matthias Lütolf, a professor at EPFL’s Institute of Bioengineering and leading author of the study, this approach is “non-physiological,” because many organs, such as the intestine or the airway, are tube-shaped and much larger. To develop larger organoids that resemble their normal counterparts, Lütolf and his team turned to bioprinting.
Although scientists have produced lab-grown organoids for decades, advances in biotechnology areas, such as tissue engineering, biomaterials, and biofabrication have accelerated organoid research, enabling the development of complex biological systems.
“Bioprinting is very compelling because it allows you to deposit cells anywhere in 3D space, so you could think of arranging cells into an organ-like configuration such as a tube,” Lütolf said. “With traditional methods for growing organoids, you can grow either stomach organoids or intestinal organoids—whereas, with bioprinting, you can combine different cell types and arrange them in different ways.”
Moreover, what makes the newly developed approach different from other methods to grow organoids is that it combines the flexibility and precision of 3D printing with the ability of stem cells to grow and organize themselves. In what Lütolf described as “allowing the biology to happen,” something he considers “absolutely crucial.”
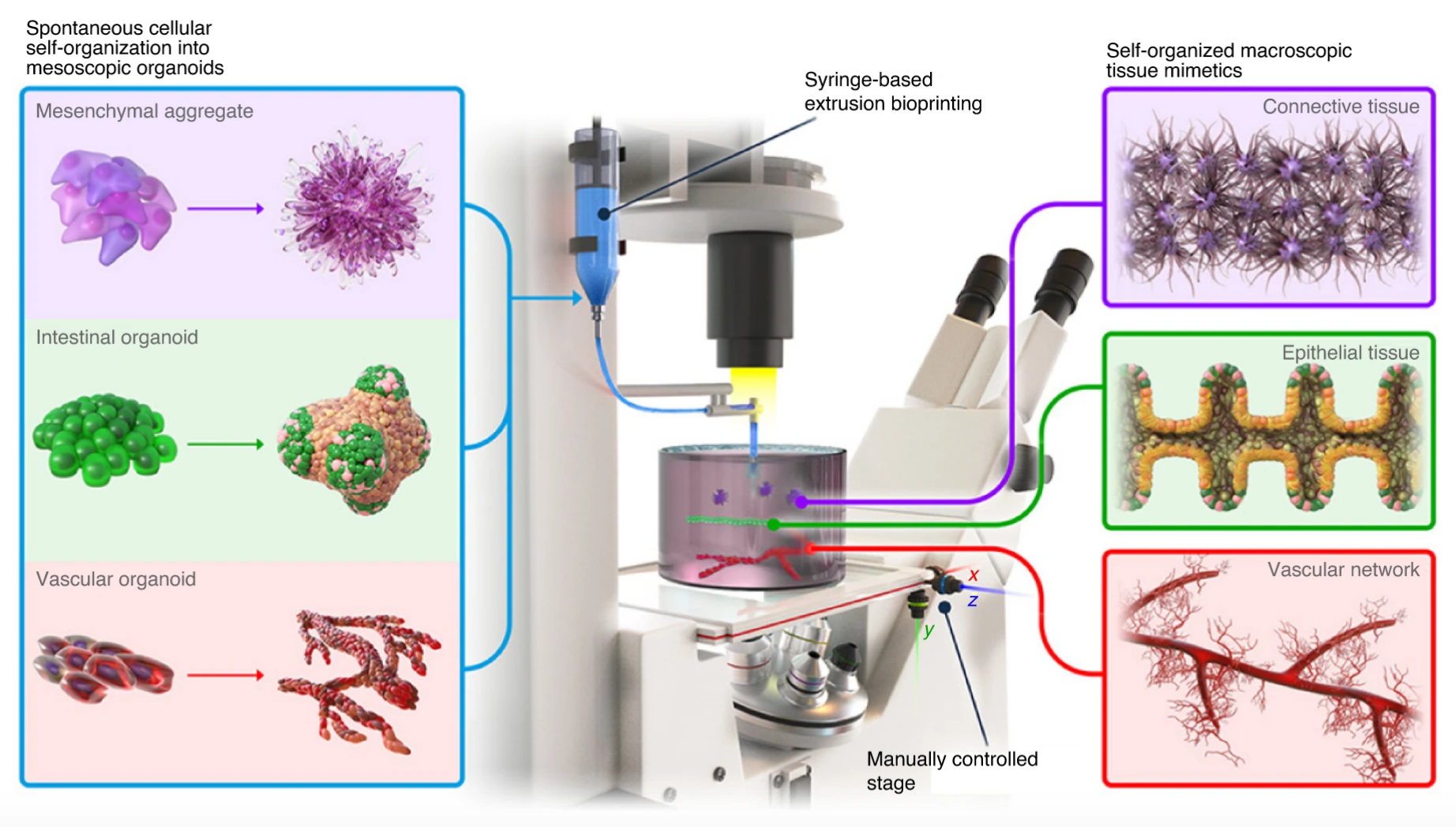
BATE bioprinting method using spontaneously self-organizing building blocks to create large-scale tissues. Image courtesy of EPFL.
Also, the researchers found that relying on microscope-based bioprinting lowered the need for in-depth expertise on hydrogel rheology and bioink formulation as it facilitated printout optimization by providing direct user feedback for visually controlling and modulating the printing process in real-time. The versatility of BATE is also seen in its ability to control the sequential deposition of supportive cells spatially and temporally, since the integration of the bioprinter into an automated microscope makes it possible to track tissue emergence in real-time and, if desired, return to specific locations to precisely place other cell types, described the scientists.
“In other bioprinting approaches, you don’t see what’s going on. The cool thing about using a microscope is that you can always see what you’re doing and you can watch what cells do – you’re not blind,” Lütolf went on.
Once stem cells were seeded, the cells started to grow and interact with each other, forming a continuous, tube-shaped tissue that mimicked many of the anatomical and functional features of a regular gut, suggested the study. Similar results were obtained with primary mouse colon and stomach stem cells, as well as with human colon stem cells, indicating that BATE may be broadly applicable to epithelial stem cell-derived organoids.
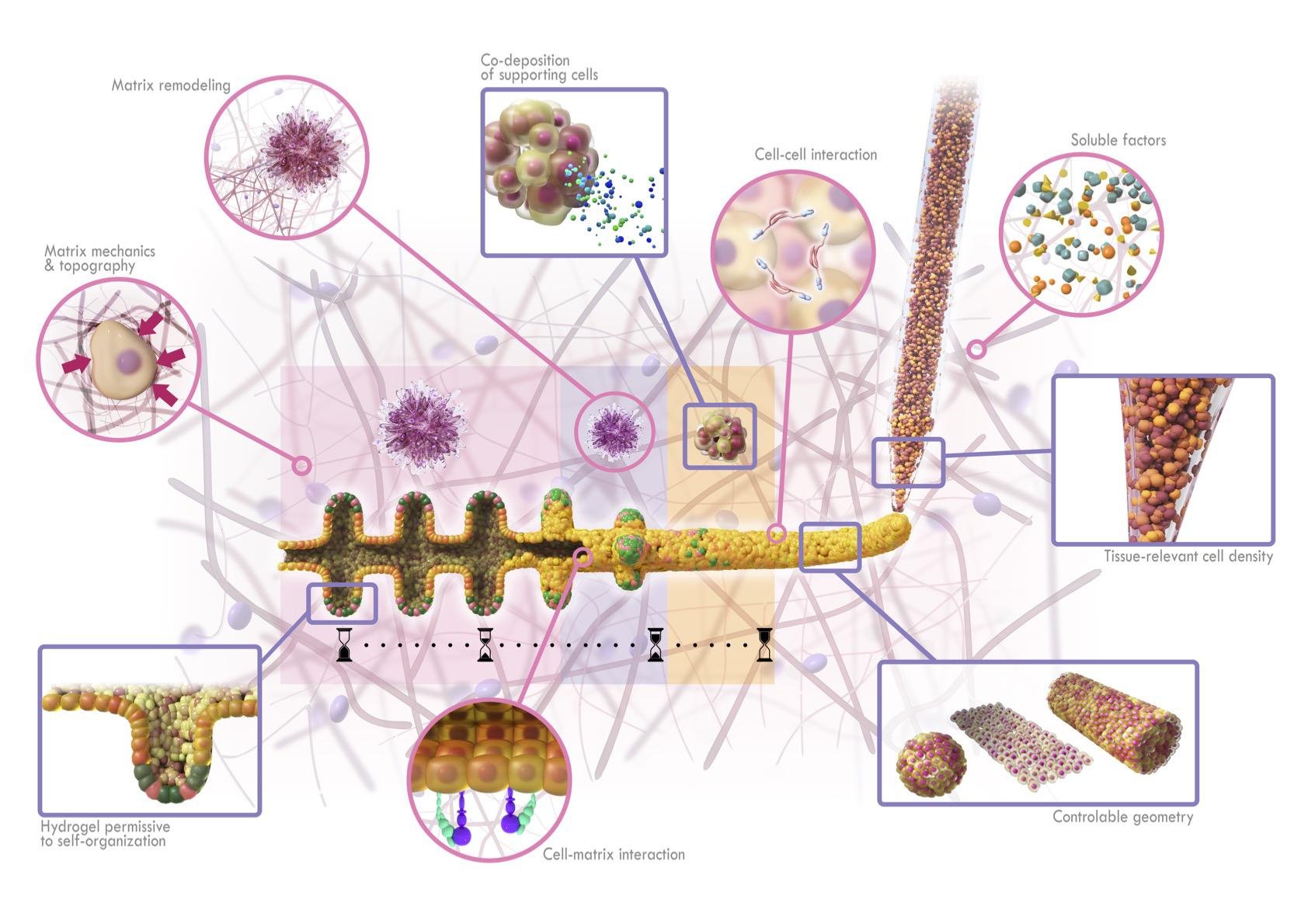
BATE applied to intestinal tissue engineering. Robust control over the cellular density and tissue geometry can be achieved directly inside environments permissive to multicellular self-organization. Image courtesy of EPFL.
The lab-grown guts, which reached up to three centimeters in size, were composed of crypt-shaped pockets with stem cells and they contained the same specialized absorptive and secretory cells as those found in a full-sized intestine. The mini-guts’ secretory cells were also able to secrete antimicrobial molecules in response to specific stimuli.
Lütolf also noted that with traditional methods for growing organoids, researchers can grow either stomach organoids or intestinal organoids, whereas, with bioprinting, they were able to combine different cell types and arrange them in different ways. In particular, BATE enabled the sequential printing of multiple cell types to form complex geometries and cell-type arrangements with good spatial resolution.
In the study, the investigators were able to show how this concept of organoid fusion can be used to produce relatively large-scale tissues using identical building blocks, as well as mimic tissue boundaries by using building blocks from related organs. Moreover, the authors considered that their original cell-printing approach to guide tissue morphogenesis at different scales has several advantages compared to other existing bioprinting technologies.
Results from the study showed that BATE allowed fragile cells, such as primary stem cells to organize into a complex geometry directly within the most potent 3D culture matrices, such as the one used in the study, Matrigel. It also reduced printing time and geometrical complexity because the microscopic architecture of the final constructs was created by the cells during subsequent remodeling and self-organization.
Although the study provides a new tool for engineering self-organized tissues and mimicking organ boundaries, Lütolf believed that their use in regenerative medicine, including for replacing human tissues and organs, is still years away. But he noted that the newly developed approach could be used to build tissue models for human diseases, including cancer, and for testing how drug candidates act on specific cell types within a tissue.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Inside The Barnes Global Advisors’ Vision for a Stronger AM Ecosystem
As additive manufacturing (AM) continues to revolutionize the industrial landscape, Pittsburgh-based consultancy The Barnes Global Advisors (TBGA) is helping shape what that future looks like. As the largest independent AM...
Ruggedized: How USMC Innovation Officer Matt Pine Navigates 3D Printing in the Military
Disclaimer: Matt Pine’s views are not the views of the Department of Defense nor the U.S. Marine Corps Throughout this decade thus far, the military’s adoption of additive manufacturing (AM)...
U.S. Congress Calls Out 3D Printing in Proposal for Commercial Reserve Manufacturing Network
Last week, the U.S. House of Representatives’ Appropriations Committee moved the FY 2026 defense bill forward to the House floor. Included in the legislation is a $131 million proposal for...


































