3D Printed Drug Delivery Implant Achieves Successful Clinical Trial for Parkinson’s Disease
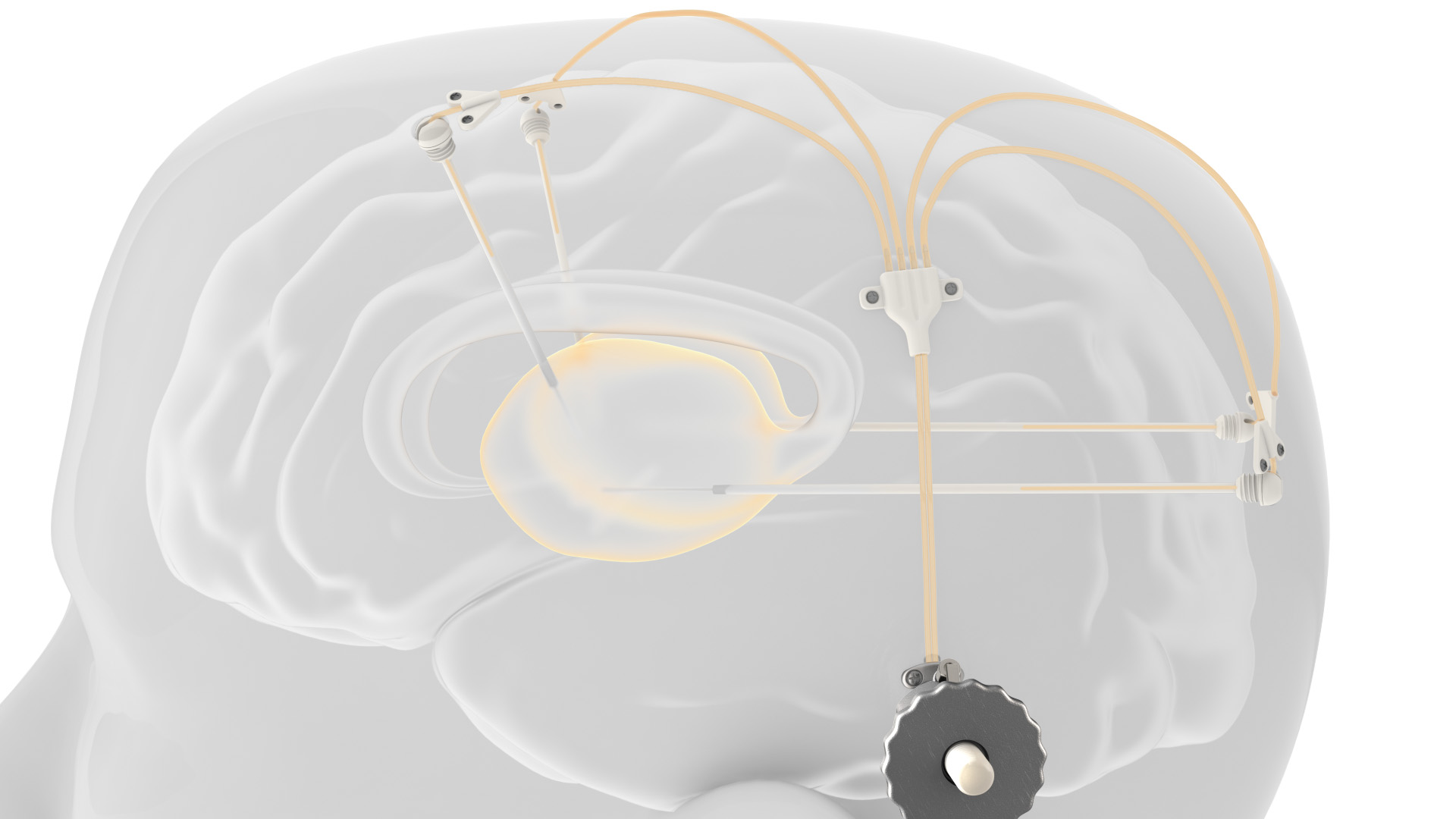
A novel drug delivery system by the global engineering technologies company, Renishaw, was successfully used as part of an extension study to a first-in-human clinical trial for the treatment of Parkinson’s disease. Renishaw, designed and manufactured the chronic drug delivery system called neuroinfuse, which is composed of up to four catheters that can be accessed via a 3D printed titanium port implanted behind the patient’s ear to aid in safely delivering a novel drug through brain infusion to patients with the neurodegenerative disorder.
Originally created to enable pharmaceuticals to bypass the blood-brain barrier for the ongoing treatment of neurological disorders and brain tumors, neuroinfuse acts as a mechanism to deliver drugs directly into the brain, so that new therapeutic regimes can be developed to treat neurological disorders, using minimal doses for maximum effect, claimed Renishaw.

The transcutaneous bone-anchored 3D printed titanium port that is implanted behind the patient’s ear. The port connects the internal catheters to the external administration kit. (Image courtesy of Renishaw)
The innovative, patented design is expected to allow patients to receive infusions in an out-patient setting, rather than requiring the implantation of new catheters for each infusion, which has been the only option for many patients to date. According to experts, infusion therapies are usually more effective than long-acting pills and patches to treat Parkinson’s disease, especially as the neurodegenerative disorder continues to progress and patient’s suffer from so-called “wearing-off” symptoms when the benefits of medicines don’t last to the next dose. Instead, infusion therapies provide a continuous flow of drugs throughout the day, address the lack of dopamine in the brain, and treat the motor and non-motor symptoms of the patient.
The neuroinfuse chronic drug delivery system is composed of up to four catheters, which are implanted into a target area within the brain. The catheters can be accessed via a 3D printed titanium transcutaneous bone-anchored port implanted behind the patient’s ear. The port then connects the internal catheters to the external administration kit and is designed to allow intermittent chronic re-administration of therapeutics without the need for further surgery. Drug-filled infusion lines are connected using an MRI compatible application set, which repeatably locates onto the port. While retractable needles extend through a septum in the port to enable therapeutics in the external infusion lines to be infused through the implanted catheters.
Manufactured on Renishaw’s own metal 3D printing systems, the port features a roughened surface below the skin to encourage bone integration and device anchorage, whereas, above the skin, it is polished to discourage bacterial settlement. Immediately following the implantation surgery, a silicone cap is pressed over the port along with sterile dressings, to prevent thickening of the skin around the port for easy application set connection.
“This device has the potential to completely change how neurological diseases are treated. The port allows repeated delivery of pharmaceuticals over long periods of time, without the need for further surgery — patients can be admitted as outpatients for the infusions,” described Max Woolley, Technical Fellow and Head of Drug Delivery Device Research and Development at Renishaw in 2019. “If we produced the device using traditional subtractive machining, we estimate it would have required up to ten parts. By designing for metal additive manufacturing, it was possible to produce it as one component, reducing the time needed for manufacture and the potential for error from unnecessary assembly operations.”
The company recently announced that its intraparenchymal drug delivery system played a critical role in a joint Phase 1/2 trial clinical study with Herantis Pharma, a Finland-based pharmaceutical company, that enrolled 17 people with moderate Parkinson’s disease, each with 15 years of evident motor symptoms. The study explored the safety, performance, and tolerability of Renishaw’s neuroinfuse device and an investigational therapy called cerebral dopamine neurotrophic factor (CDNF), which is based on a protein naturally found in the blood and liquid surrounding the brain and spinal cord, as a treatment for Parkinson’s disease. As well as the surgical accuracy of the device implantation procedure.
During the trial, CDNF was administered directly into patients’ brains through the delivery system provided by Renishaw Neuro Solutions. The drug delivery device, which can only be implanted by a neurosurgeon, is currently considered the only platform to allow for repeated, intermittent infusions into the functional tissue in the brain, known as parenchyma. Initial results from the main study indicated the predictable and accurate placement of the device as well as its positive performance and safety.
Moreover, as part of the extension study, neuroinfuse has continued to safely and effectively deliver infusions to the brain, proving that this potential Parkinson’s treatment continues to be well-tolerated by patients with moderate disease showing a promising potential to ease motor symptoms and slow disease progression, as reported by Herantis Pharma. In fact, Renishaw claimed that the results from the 15 patients who progressed into the extension study demonstrated that the device was safe and presented no serious adverse events considered to have device or drug causality. Additionally, the system’s unique ability to deliver repeated infusions continued to facilitate the assessment of CDNF’s safety and early efficacy in patients.

The port attachment device and four needle drug application set designed to kinematically locate and lock onto the transcutaneous port. (Image courtesy of Renishaw)
“I’m delighted to see Renishaw’s drug delivery system continuing to facilitate repeated infusions over an extended period of time for such a complex condition as Parkinson’s Disease,” said Rupert Jones, Managing Director of Renishaw Neuro Solutions. “The device’s performance demonstrates what a powerful delivery platform it is for the treatment of many, currently incurable, neurological conditions, opening new possibilities in the field of neurosurgery and neuroscience. I see this as a hugely positive step forward and believe all involved in the study should be proud of their achievements.”
As one of the novel infusion regimes, neuroinfuse is currently only used in approved clinical trial settings. Yet, in order to eventually make the device generally available to patients, Renishaw is seeking academic, clinical, and industrial partners across a wide range of indications, from oncology to neurodegenerative diseases. However, the company expects the drug delivery system to have potential applications in a comprehensive suite of therapy treatments, like gene, cell, drug, protein, and enzyme therapies, and particularly enhance the treatment of neurodegenerative, neuro-oncology, and other debilitating neurological conditions. More importantly, in the short term, Renishaw is looking to open a pathway from laboratory to clinic for effective and cost-efficient neurological therapies.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
DMG Mori Joins $10M Defense 3D Printing Program
To look at the Biden administration and the Trump administration that succeeded it and find areas of policy overlap is obviously a bit of a challenge. But such areas certainly...
Where the Money Is Going: The New Infrastructure Landscape
This is Part 1 of a two-part PRO series examining where infrastructure investment is flowing and how those trends are reshaping manufacturing, energy, logistics, and additive manufacturing. Part 2, by...
First Containerized System from AML3D Now Operational at US Navy AM CoE
Shipbuilding giant Austal USA recently announced the launch of the Digital SEA (Secure Exchange for Additive) platform, which should play a major role in expanding accessibility to the sorts of...
INDOPACOM Advanced Manufacturing Team Saves Thousands Per Week at Joint Exercise in the Philippines
In the summer of 2025, the US Indo-Pacific Command (INDOPACOM) opened a new advanced manufacturing facility at Schofield Barracks, the Hawaiian home of the US Army’s 25th Infantry Division. INDOPACOM...