In Vivo Bioprinting Made Possible with Digital Near-infrared Photopolymerization
Researchers from Belgium, China, and the US have come together to study 3D bioprinting, working to expand noninvasive, in vivo applications further. Releasing the findings of their study in the recently published ‘Noninvasive in vivo 3D bioprinting,’ the scientists are concerned with finding better ways to treat patients without causing additional trauma during regenerative practices, surgical implantation, and plastic surgery.
The scientists chose near-infrared (NIR) light technology for this project, due to its versatility (used in drug release systems, photodynamic therapy, photothermal therapy, in vivo imaging, 3D imaging, and more) and the ability to prompt photopolymerization. Hoping to open a new avenue in 3D printing, the researchers created a noninvasive 3D printing system, bioprinting directly into tissue constructs without surgical implantation.
The new process is centered around digital near infrared (NIR) photopolymerization (DNP)–based 3D printing technology, featuring patterned light that is able to penetrate through skin—thus causing subcutaneous injection of the cell solution.
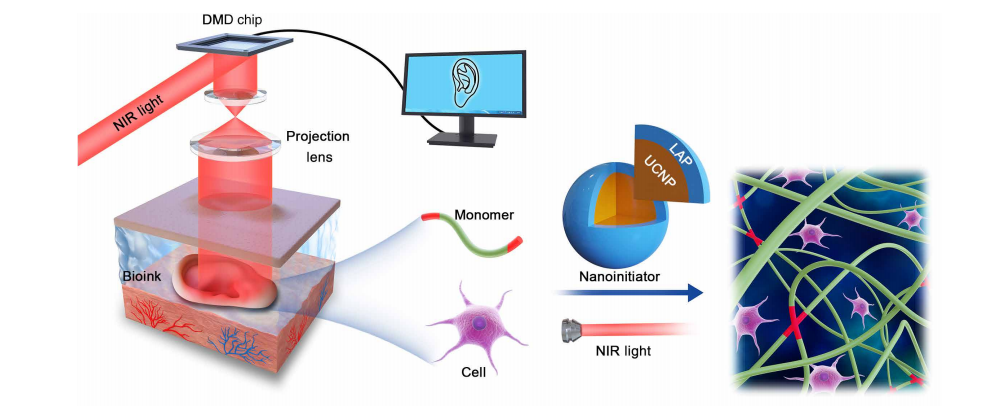
Schematic diagram of DNP-based noninvasive 3D bioprinting. The data of a customized CAD model were sent into the DMD chip through a control computer. The 980-nm NIR light with an optical pattern was casted across optical lens, and tissue onto the bioink, which was injected into the body to noninvasively fabricate a living tissue in vivo. The bioink contains UCNP@LAP nanoinitiators that can convert the NIR light to 365-nm light and then initiate the optical pattern–controlled polymerization of monomers.
The researchers began by assessing the process in vitro:
“Because of the excellent capacity of supporting cells for survival or proliferation, biocompatible hydrogels are commonly used as the matrix materials for bioprinting. Here, we found that the DNP process could fast print the GelMA-derived hydrogel obstacles by a layer-by-layer manner, and the time of printing a 200-mm-thick layer is about 15 s. Then, the capacity of the DNP process in 3D printing of complex hydrogel constructs was evaluated.”
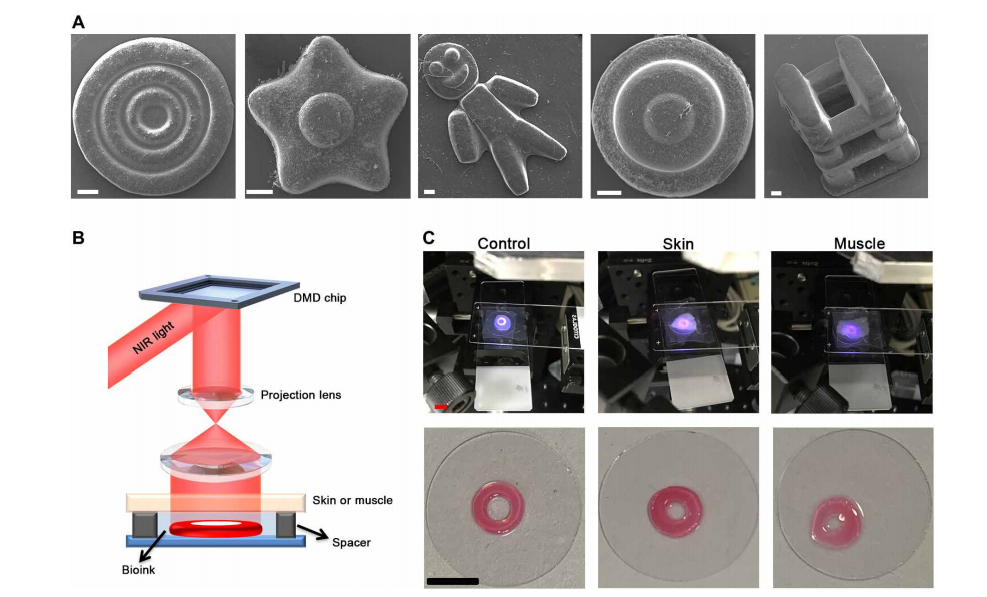
3D bioprinting acellular constructs using DNP-based process in vitro. (A) Scanning electron microscopy (SEM) images of fabricated constructs including three-ring microconstructs with decreasing widths, flower-like, Danboard-like, round cake-like, and a type of truss constructs. Scale bars, 200 m. (B) Schematic diagram of printing setup used to estimate the tissue-penetration capacity. The bioink was deposited under the skin or muscle. (C) The images of ring constructs printed from bioink (control) or bioink covered over by skin or 0.5-mm-thick muscle by DNP process. Scale bars, 0.5 cm. Photo credit: Yuwen Chen, State Key Laboratory of Biotherapy and Cancer Center.
Subsequently, the team was able to print three-ring structures at decreasing widths—from 200 to 100 mm. A variety of other complex geometries were fabricated ex vivo, as well, demonstrating the necessary precision.
Using both pig tissue and mice skin, the researchers were able to imitate noninvasive in vivo 3D bioprinting. Circular structures were created, and the team noted that the use of either the skin or tissue did not cause any changes to the print quality, with further calculations and measurements showing success in terms of using DNP for noninvasive bioprinting; in fact, they showed that in one or seven days after printing, tissues were free of ‘significant inflammation’ or abnormalities.
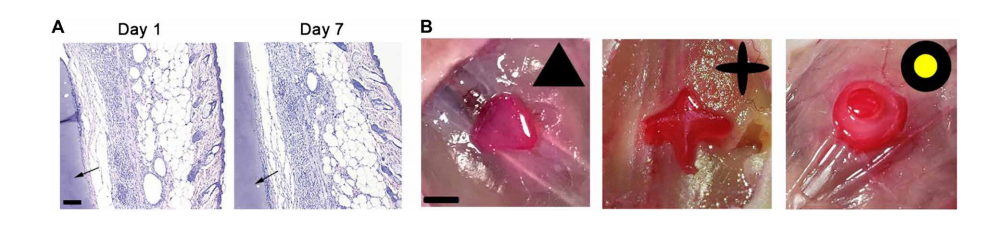
3D bioprinting acellular constructs by DNP-based process in vivo. (A) H&E of the surrounding tissue of construct after DNP printing in vivo for 1 and 7 days. The arrow represents the printed construct. Scale bar, 100 m. (B) CAD models and vitamin B12–stained triangle, cross, and two-layer cake-like constructs fabricated by noninvasive DNP-based process in vivo. Scale bar, 0.5 cm. Photo credit: Jiumeng Zhang, State Key Laboratory of Biotherapy and Cancer Center.
“This indicates that the DNP-based noninvasive bioprinting process did not cause obvious side effect in situ,” stated the researchers.
As they continued evaluations, they 3D printed a variety of samples, to include a triangle, cross, and ‘cake-like hydrogel constructs.’
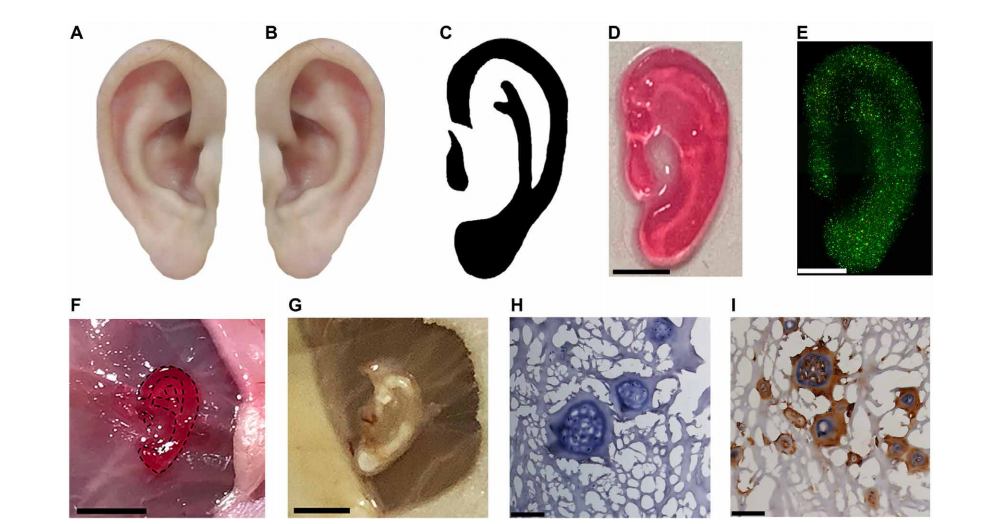
Noninvasive 3D bioprinting ear-like tissue by the DNP-based process. (A) Representative image of the normal ear. (B) Mirror image of (A). (C) Optimized ear-outline image of (B). (D) Image of printed ear-like construct from the bioink covered over by skin by DNP process. Scale bar, 2 mm. (E) The Live/Dead staining for ear constructs encapsulated with chondrocytes bioprinted from bioink covered by skin after culture for 7 days. Scale bar, 2 mm. (F) Noninvasive 3D bioprinting of ear-shaped construct in vivo by DNP-based process. The ear-shaped construct was printed subcutaneously in BALB/c nude mice. Scale bar, 5 mm. (G) Representative image of bioprinted ear-shaped construct at 1 month. Scale bar, 5 mm. (H) H&E and (I) collagen type II immunostaining of retrieved ear-shaped construct at 1 month. Photo credit: Yuwen Chen, State Key Laboratory of Biotherapy and Cancer Center.
In printing a customized ear-shaped tissue structure, the researchers realized success—and potential for future treatment of auricle defects or microtia.

Noninvasive 3D bioprinting conformal ASC-laden scaffold for muscle defect repair by the DNP-based process. (A) Schematic illustration of the conformal ASC-laden scaffold for muscle defect repair. (B) Representative images exhibit acceleration of the wound healing of the DNP group compared with control. Scale bar, 5 mm. (C) Percent closure of muscle wounds evaluated at day 10. **P<0.01, n=5. (D) H&E histological analysis of muscle wound healing at day 10 after treatments. Photo credit: Yuwen Chen, State Key Laboratory of Biotherapy and Cancer Center.
“Meanwhile, the noninvasively printed conformal ASC-laden scaffold can promote the healing of the muscle defects, showing potential clinical application. Therefore, the demonstrated noninvasive in vivo 3D bioprinting technology could provide a novel tool to advance the minimally invasive or noninvasive medicine, showing potential clinical applications,” concluded the researchers.
What do you think of this news? Let us know your thoughts; join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘Noninvasive in vivo 3D bioprinting’]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
You May Also Like
3D Printing Financials: Prodways Ends 2024 with a Profit
After a tough couple of years, Prodways (EPA: PWG) is starting to bounce back. The French 3D printing company finally made a profit in 2024, improved its operating performance, and...
Blue Origin & Auburn University Use EOS M290 to Study Copper 3D Printing
Blue Origin, the commercial space company built off of investments from Amazon founder Jeff Bezos, has donated two EOS M290 powder bed fusion (PBF) printers to Auburn University’s National Center...
Rocket Lab to Acquire Restructured Laser Communications Provider Mynaric AG
Rocket Lab USA, the Long Beach-based, end-to-end space services company that specializes in producing rockets with additive manufacturing (AM), has announced plans to acquire Mynaric AG, a German provider laser...
3D Printing Financials: Stratasys Ends 2024 with Cost Cuts and Growth Plans
Stratasys (Nasdaq: SSYS) has wrapped up 2024 with stronger margins but a full-year net loss. The polymer 3D printing leader navigated a year of economic headwinds, restructuring efforts, and shifting...



























