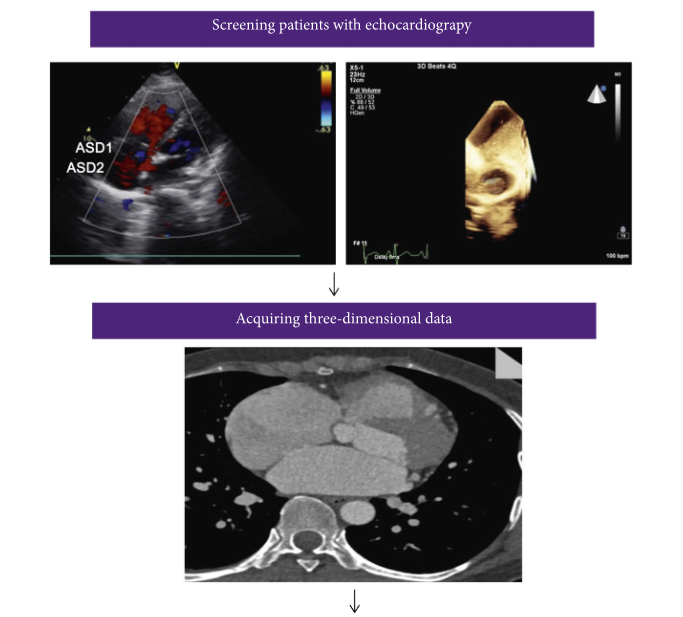
Researchers Evaluate Feasibility of Closing Multiple Atrial Septal Defects Guided by 3D Printed Model
We’ve often seen physicians use 3D printed heart models to help during surgeries, but a group of researchers from China published a paper on using them to help with an alternative to surgery for repairing secundum atrial septal defect (ASD), a rare congenital defect characterized by a hole in the wall between the atria. Their goal was to evaluate how feasible it was to use a single device to close several ASDs guided by the 3D printed heart model and transthoracic echocardiography (TTE).
Due to interference between devices and threat of repeat intervention, it’s difficult to use multiple devices simultaneously, or in staged device closure, in percutaneous transcatheter closure of an ASD. But using an over-sized device can tear the atrial septum. So the best plan is to use single device closure for patients with multiple ASDs, as it preserves the anatomical structure.
“However, this strategy is technically challenging because of inability to determine the target defect for catheter passage and occluder selection, warranting careful interventional planning with comprehensive anatomical information for successful device closure,” the team wrote.
That’s where the 3D printed heart model comes in. The researchers used the single-device strategy, assisted by 3D printing, to perform multiple ASDs closure, and compared their results of “3D printing-based and transthoracic echocardiography (TTE)-guided percutaneous transcatheter closure with those of traditional fluoroscopy-guided closure.”
Simple working flowchart in patients with multiple ASDs, from image acquisition to 3D printed solid and hollow model.
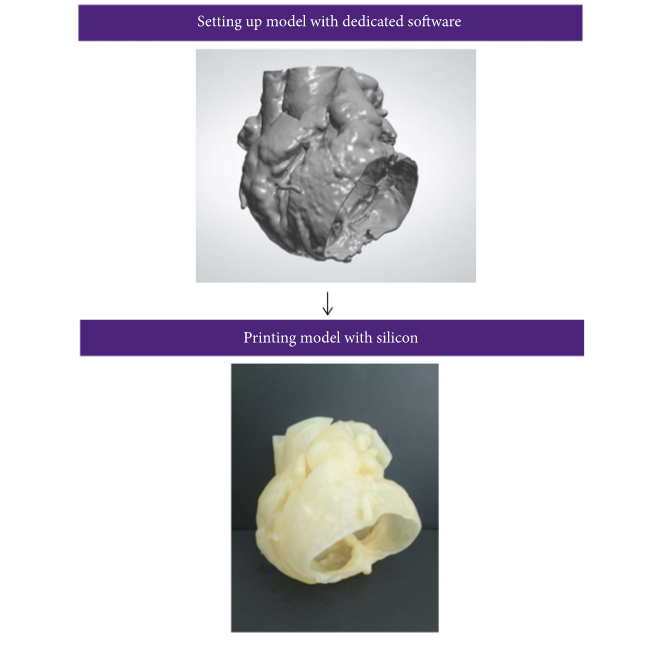
62 patients diagnosed by TTE with two or more ASDs with a 5mm or more diameter were enrolled in their non-randomized study for analysis. 30 had cardiac computed tomography angiography (CTA) ahead of surgery in order to get data to create their 3D printed heart models. The CTA images were reconstructed and saved in DICOM format, before being imported to Materialise Mimics software. Cardiac masks were generated for 3D models, and 3-matic software was used to hollow them. The STL files were 3D printed, in hollow fashion, at 1:1 scale on a ProJet MJP 2500 Plus 3D printer out of silicone.

3D printed model of a patient with multiple ASDs. (a) and (b) show the model from left and right atrial sides, respectively. The arrows depict the position of the ASDs. (c) and (d) illustrate the status after occluder deployment in the model.
The surgeons performed in vitro simulated occlusion with the 3D printed models as a pre-op evaluation. Then, while the other 32 patients underwent ASD closure with fluoroscopic guidance, this group had TTE-guided closure procedures.
“The apical four-chamber view and parasternal short-axis view were used for guidance, and the multipurpose catheter was passed through the targeted defect, which was determined using the 3D printing model and intraoperative TTE,” the researchers explained.
“Then, a single septal occluder was inserted for ASD closure under TTE guidance. An ASD occluder or PFO occluder was selected based on the in vitro simulated occlusion in a 3D printing model.”
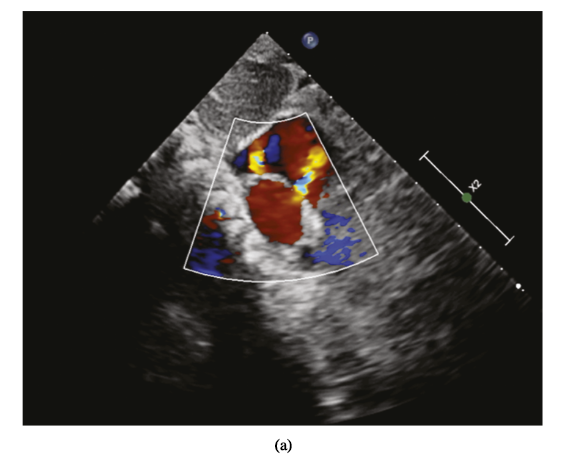
After implantation, the device position was evaluated through subcostal, apical four-chamber, and parasternal short-axis views, and they also performed Color Doppler assessment to detect any issues, like coronary sinus return or residual shunting. Once they determined that the occluder had been implanted correctly, “it was released by rotating the cable counterclockwise under TTE guidance,” and a reassessment was then performed in echo views, below.
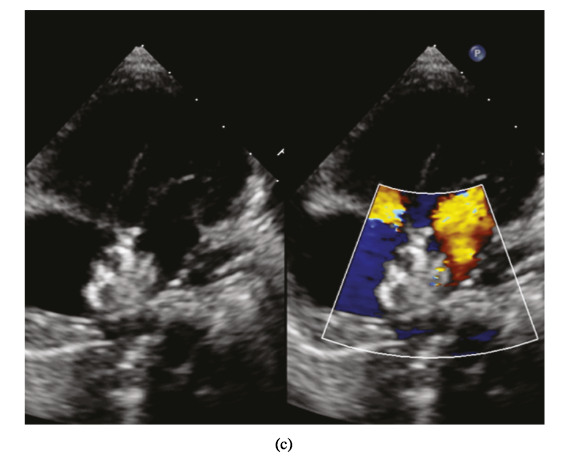
Percutaneous closure of multiple ASDs under TTE guidance. (a) Multiple ASDs image displayed in subcostal view. (b) The left disc was released (parasternal short-axis view). (c) The ASDs were closed (four-chamber view).
“In the conventional group, multiple ASDs occlusion was performed under fluoroscopic guidance using the single occlusion device,” they wrote. “Based on TTE measurements, the single device was selected, equal to or up to 4 mm larger than the main defect [10]. According to experience [10, 20, 21], the device was usually implanted into the largest defect. The occluder was replaced if echography found more than two residual shunts, the residual shunt was >5 mm in diameter, or the device compressed the mitral valve.”
Immediately post-op, and 6 months after the device closure, all 62 patients were evaluated via TTE and electrocardiogram, with the researchers noting the presence of any arrhythmia, residual shunt, or valve dysfunction. A Brand-Altman analysis was used to evaluate the agreement “between device size of 3D printed model and traditional estimation,” and the data was analyzed with SPSS software.
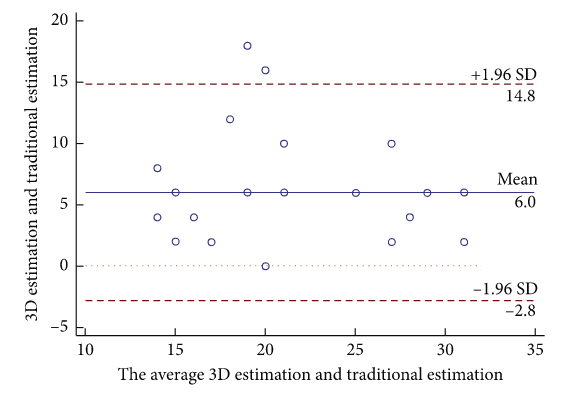
Bland–Altman plot analysis. Bland–Altman plot of empirical estimation versus 3D printed model estimation of occluder size.
They found that 26 patients in the 3D printing/TTE group, and 27 patients in the conventional group, achieved successful transcatheter closure with a single device. The prevalence of residual shunts was lower in the first group immediately and 6 months post-op, and there were no complications in either group during the procedure or the two follow-ups.
“Gender, age [18.8 ± 15.9 (3–51) years in the 3D printing and TTE group; 14.0 ± 11.6 (3–50) years in the conventional group], mean maximum distance between defects, prevalence of 3 atrial defects and large defect distance (defined as distance ≥7 mm), and occluder size used were similarly distributed between groups,” the team wrote. “However, the 3D printing and TTE group had lower frequency of occluder replacement (3.8% vs 59.3%, ), prevalence of mild residual shunts (defined as <5 mm) immediately (19.2% vs 44.4%, ) and at 6 months (7.7% vs 29.6%, ) after the procedure, and cost (32960.8 ± 2018.7 CNY vs 41019.9 ± 13758.2 CNY, ).”
They did note that the occluder on the 3D printed model was “consistently larger than in the empirical estimation but similar to final clinical selection,” which indicates a higher level of accuracy. Even in patients with a large defect distance, the results of the study suggest that “interventional therapy with a single occluder for multiple ASDs is feasible,” especially as technical difficulties and complex anatomy make successful single device closure tricky to achieve. It’s important to remember that the accuracy of the 3D printed anatomic model is paramount in attaining single device closure in patients with multiple ASDs.
“Occluders’ sizes preestimated by the 3D printed model were similar to the size actually used for patients and larger than the size from conventional empirical estimation. These results indicate that preevaluation using the 3D printed model can avoid unnecessary interventions, the possibility of enlarging ASD by changing occluders and the financial waste of replacing occluders,” they explained.
The researchers ultimately determined that it’s feasible to use a 3D printed model to help achieve successful device closure for patients with multiple ASDs with a defect distance of ≥7 mm. The model can also help screen patients who may not be well-suited for the closure route, and should instead seek direct surgical repair.
“The combination of the 3D printing technology and ultrasound-guided interventional procedure provides a new approach for individualized therapeutic strategy of structural heart disease and in particular a reliable therapeutic method for multiple ASDs, especially for challenging cases with large defect distance,” they concluded.
Discuss this research and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
The Dental Additive Manufacturing Market Could Nearly Double by 2033, According to AM Research
According to an AM Research report from 2024, the medical device industry, specifically in dentistry, prosthetics, and audiology, is expected to see significant growth as these segments continue to benefit from...
Heating Up: 3D Systems’ Scott Green Discusses 3D Printing’s Potential in the Data Center Industry
The relentless rise of NVIDIA, the steadily increasing pledges of major private and public investments in national infrastructure projects around the world, and the general cultural obsession with AI have...
AM Research Webinar Explores Continuum’s Sustainable Metal Additive Manufacturing Powders
Metal additive manufacturing (AM) powder supplier Continuum Powders is working to develop solutions that empower industries to reduce waste and optimize their resources. An independent life cycle assessment (LCA) of...
3D Printed Footwear Startup Koobz Lands $7.2M in Seed Round
California-based Koobz is focused on reshoring the U.S. footwear supply chain with advanced manufacturing processes, including 3D printing. The startup just announced that it has added $6 million to its...