Safety and 3D-Printed COVID-19 Medical Devices — An Interview with Veterans Affairs
In our previous article on the topic, we mentioned some broad guidelines that seem to have coalesced related to 3D printing medical devices in the face of the supply shortages caused by the COVID-19 pandemic. We have since spoken to Dr. Beth Ripley, a radiologist in the Veterans Affairs (VA) Puget Sound Health Care System and Director of the VA 3D Printing Network in the VHA Innovation Ecosystem.
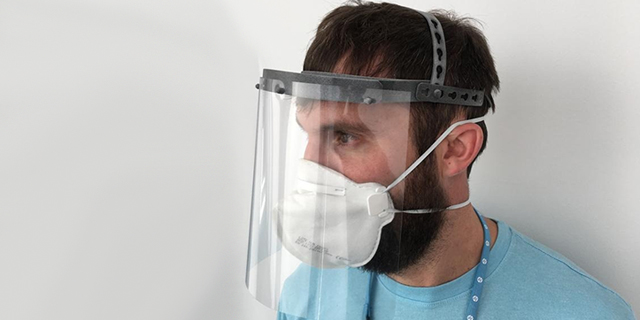
Face Shields vs Face Masks
Dr. Ripley began by underscoring the difference between face shields (Class I devices) and face masks (Class II devices), terms that are easily mixed up for obvious reasons. The former is made up of a 3D printed headband and plastic film that blocks the entire face of the wearer to protect against flecks of moisture. A face mask is more varied, depends on who is wearing them and their applications, and include surgical masks, N95 respirators and general-use, non-medical masks.
Both face shields and face masks are considered medical devices and, outside of these irregular pandemic circumstances, must be manufactured in accordance to Current Good Manufacturing Practice (CGMP/GMP) regulations in the U.S. However, due to the coronavirus outbreak, the FDA is processing Emergency Use Authorization Applications that will expedite the approval of some 3D printed devices and loosen the guidelines under which devices that are less highly regulated can be made, such as face shields.
Face shields are where the broader 3D printing community should focus its activities, according to Dr. Ripley. This is because the device isn’t in direct contact with the mucous membranes of the wearers and they do not have to breathe through it, which removes concerns related to what material they’re made of. Dr. Ripley concurred that face shields do seem to be fulfilling a supply need facing medical staff confronting the virus on the ground.
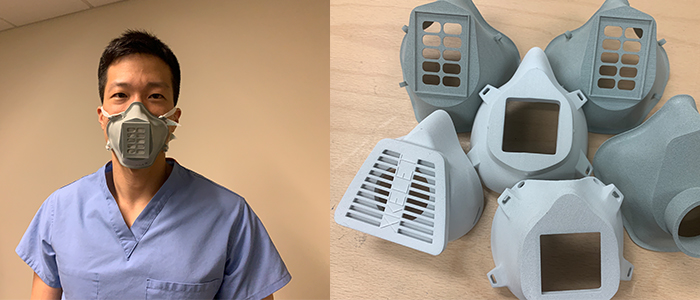
In contrast, even in the current pandemic situation, medical face masks like N95 respirators must still be manufactured in accordance to CGMP regulations in the U.S. Therefore, businesses with existing CGMP facilities and workflows will be able to more easily obtain Emergency Use Authorization for 3D printed devices. Industrial manufacturers and businesses with more substantial resources will also more able to meet the necessary regulations in order to produce emergency medical devices.
Dr. Ripley described the importance of CGMP certification for manufacturing these items:
“[CGMP] certification communicates that the facility or the manufacturer has a quality management system in place. This means that they have a proven method to make sure that the parts that they are creating meet all of the safety standards and quality standards that are needed. It’s an involved process to achieve that level of certification. And that’s what you want to see when you’re talking about medical device manufacturing. They are also registered with the FDA and those manufacturing facilities are subject to inspection and reporting. In this case right now, in which we’re turning to non-traditional manufacturing methods, we want to do so in, in a way that weighs the needs to get a product out with the benefits that we could still have from trying to make sure that we put out high quality products.”
General-Use Masks
Dr. Ripley pointed out that, while face masks for medical settings must still be made with CGMP regulations, the same isn’t true for general-use masks. The Centers for Disease Control (CDC) has just released guidance recommending the use of cloth masks when interacting with the public is necessary, particularly in high-traffic areas and regions where COVID-19 is active.
The CDC does not discuss 3D printed masks specifically and individuals can turn to 3D printing to make masks. However, we currently lack any guidance related to the types of materials might be safest, outside of the FDA’s broad guidance related to 3D printed medical devices in general.
Dr. Ripley pointed out that the VA is focused on the medical community, but that it is working providing guidance on safety for general use face masks to educate the community about safety.
Though not yet official, the VA is bearing in mind the following safety considerations to take into account with regard to public-use masks:
- How does the general-use mask fit, how does it feel? Is it actually wearable?
- What materials are being used to make the mask? Three concerns that should be considered are
- Whether or not the material is safe for skin contact.
- The effects of off-gassing of volatile organic compounds from 3D printed parts and during the printing process
- Whether or not there are any toxicities from the materials being used.
The VA and NIH teams are currently looking at several designs for community- or general-use masks that incorporate all 3D printed parts, some 3D printed and some cloth components, or are made entirely out of cloth to give users some options to choose from.
3D Printing as a Stopgap Measure
As we’ve seen from numerous stories related to the COVID-19 outbreak, 3D printing has proven extremely helpful as a rapid design and manufacturing tool. For the VA system in particular, Dr. Ripley’s colleagues were able to design a 3D-printable surgical face mask with 30 design iterations over the course of 12 days. Beyond that, the technology has enabled such companies as Lonati SpA in Italy and ŠKODA in the Czech Republic to produce large batches of medical goods in a short period of time before traditional mass manufacturing technologies can take over.
However, as powerful as these capabilities have been for the current medical supply crisis, Dr. Ripley underscored the fact that 3D printing is a stopgap measure to get us through the next weeks or months while traditional manufacturing operations are shifted toward medical supplies. Dr. Ripley explained,
“We’re trying to flatten the curve with respect to viruses, but with 3D printing we’re trying to bump up the production curve because we need to scale up production. And some of the more traditional manufacturing technologies may take a couple weeks or even months to get up and running. If you’re retooling an entire factory to make something else, or you have a new design in your injection molding, it still takes weeks. What are we going to do during these crucial one to two weeks where we haven’t scaled up? 3D printing allows us to bump up that production curve or that product curve. I think it’s totally worth it for face shields and medical supplies, getting the medical supplies into the community or into the frontline staff’s hands with a strong caveat that we should not rest on that as the final solution. It really is a stopgap, a temporary measure while we shore up more robust solutions.”
The doctor was able to bring some reality to the situation with regard to the use of 3D printing as a method of addressing COVID-19 supply issues and provided an important reminder about even the FDA’s existing emergency approval process:
“Today’s April 6, 2020. So, we’re talking about how the regulations stand as of today. As of today, you can make face shields, anyone can make face shields, you don’t have to have GMP certification. If you wanted to make a general community mask, not for healthcare providers, someone could make that. But if you want to make anything that’s a surgical mask, an N95 respirator or any type of ventilator parts, all of those have to be made in a GMP certified facility.”
As the situation progresses, we will try our best to keep on top of announcements from regulatory bodies and appreciate any and all feedback from experts in the community to help inform us and guide us in the right direction as we do so.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Creality Marks 12 Years with KliTek and AI-Powered Ecosystem Expansion
For 12 years, Creality has advanced accessible 3D printing technologies, enabling global users to turn ideas into tangible creations. What began as a desktop 3D printer manufacturer has evolved into...
Asia AM Watch: Creality Launches $177M Hong Kong IPO as HKEX Trading Debut Nears
Creality has formally launched its Hong Kong initial public offering (IPO), marking one of the biggest public market moves by a 3D printing company in 2026 and offering a new...
How AtomForm’s 12-Nozzle System Cuts Multi-Color FDM Transition Waste by Up to 90%
Pull up the print stats on multi-color FDM jobs. The number that stings isn’t time; it’s material efficiency. On six-color models, single-nozzle systems consume significant filament during transition flushing, expelling...
3D Printed Weapons Keep Showing Up in Crime
In the past few weeks, activity around 3D printed weapons in the U.S. has increased across several fronts. States including California, Colorado, New York, and Washington are moving forward with...